Section 1
Atoms, Isotopes, Ions, and Molecules
By Boundless
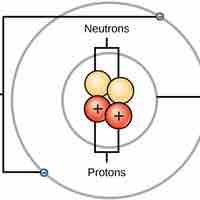
Atoms are made up of particles called protons, neutrons, and electrons, which are responsible for the mass and charge of atoms.
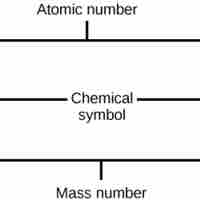
The atomic number is the number of protons in an element, while the mass number is the number of protons plus the number of neutrons.

Isotopes are various forms of an element that have the same number of protons, but a different number of neutrons.
Everything in the universe is made of one or more elements. The periodic table is a means of organizing the various elements according to similar physical and chemical properties.
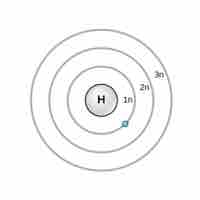
Niels Bohr proposed an early model of the atom as a central nucleus containing protons and neutrons being orbited by electrons in shells.
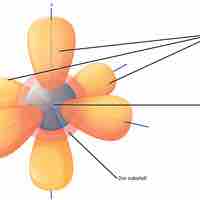
Electron orbitals are three-dimensional representations of the space in which an electron is likely to be found.
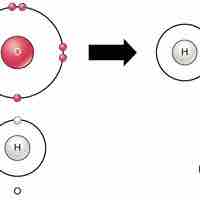
Chemical reactions occur when two or more atoms bond together to form molecules or when bonded atoms are broken apart.
Ionic bonds are attractions between oppositely charged atoms or groups of atoms where electrons are donated and accepted.
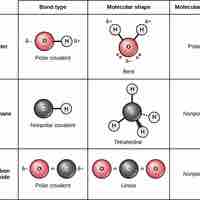
Covalent bonds result from a sharing of electrons between two atoms and hold most biomolecules together.
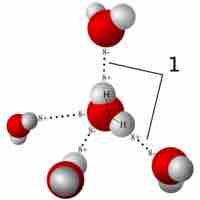
Hydrogen bonds and van der Waals interactions are two types of weak bonds that are necessary to the basic building blocks of life.