Concept
Version 10
Created by Boundless
Bond Order
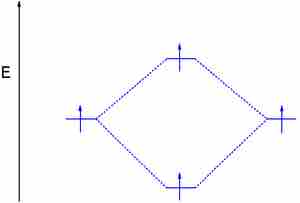
Dihydrogen with an electron in the antibonding orbital
By adding energy to an electon and pushing it to the antibonding orbital, this H2 molecule's bond order is zero, effectively showing a broken bond.
Source
Boundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources: