Ionic Bonds
Ionic bonding is a type of chemical bond in which valence electrons are lost from one atom and gained by another. This exchange results in a more stable, noble gas electronic configuration for both atoms involved. An ionic bond is based on attractive electrostatic forces between two ions of opposite charge.
Cations and Anions
Ionic bonds involve a cation and an anion. The bond is formed when an atom, typically a metal, loses an electron or electrons, and becomes a positive ion, or cation. Another atom, typically a non-metal, is able to acquire the electron(s) to become a negative ion, or anion.
One example of an ionic bond is the formation of sodium fluoride, NaF, from a sodium atom and a fluorine atom. In this reaction, the sodium atom loses its single valence electron to the fluorine atom, which has just enough space to accept it. The ions produced are oppositely charged and are attracted to one another due to electrostatic forces.
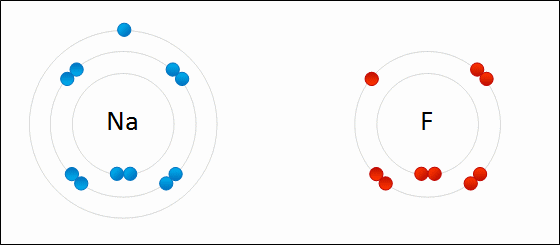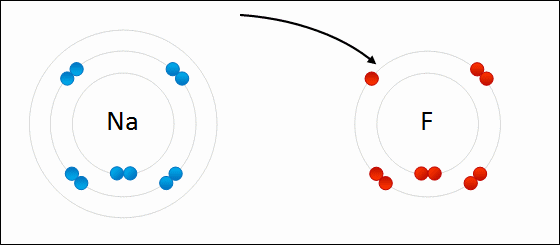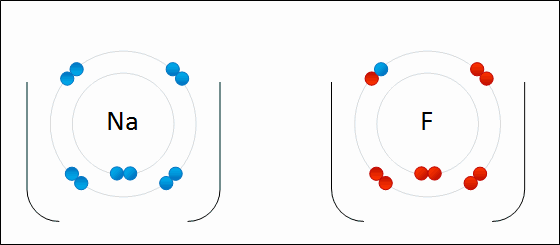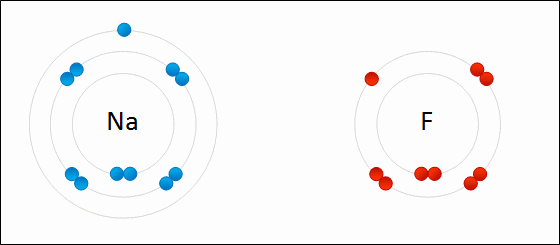
Formation of NaF
An electron is transferred from Na to F. The resulting Na+ and F- ions are electrically attracted to each other.
At the macroscopic scale, ionic compounds form lattices, are crystalline solids under normal conditions, and have high melting points. Most of these solids are soluble in H2O and conduct electricity when dissolved. The ability to conduct electricity in solution is why these substances are called electrolytes. Table salt, NaCl, is a good example of this type of compound.
Ionic bonds differ from covalent bonds. Both types result in the stable electronic states associated with the noble gases. However, in covalent bonds, the electrons are shared between the two atoms. All ionic bonds have some covalent character, but the larger the difference in electronegativity between the two atoms, the greater the ionic character of the interaction.