Section 4
Inorganic Compounds
Book
Version 6
By Boundless
By Boundless
Boundless Microbiology
Microbiology
by Boundless
3 concepts
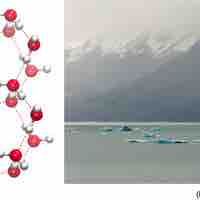
Water’s States: Gas, Liquid, and Solid
The orientation of hydrogen bonds as water changes states dictates the properties of water in its gaseous, liquid, and solid forms.
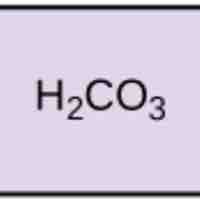
pH, Buffers, Acids, and Bases
Acids dissociate into H+ and lower pH, while bases dissociate into OH- and raise pH; buffers can absorb these excess ions to maintain pH.
Overview of the Acid-Base Properties of Salt
Some salts, such as ammonium bicarbonate (NH4HCO3), contain cations and anions that can both undergo hydrolysis.