HP-505
 | |
| Identifiers | |
|---|---|
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C18H19NO |
| Molar mass | 265.356 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
HP-505 is a triple reuptake inhibitor that was investigated by Hoechst-Roussel Pharmaceuticals.[1] In mice, HP-505 was a potent inhibitor of tetrabenazine-induced ptosis which may indicate antidepressant activity.[2]
Pharmacology
| 5-HT | NE | DA | NE:5-HT | DA:5-HT |
|---|---|---|---|---|
| 0.19 ± 0.04 | 0.34 ± 0.1 | 0.66 ± 0.15 | 1.8 | 3.5 |
The inhibitory effect of HP-505 on serotonin reuptake is approximately 1.8 and 3.5 times stronger than on norepinephrine and dopamine, respectively.[1] Subsequent investigations have found that HP-505 acts on presynaptic dopamine transporters and is devoid of anticholinergic effects.[3]
Synthesis
The N-methylated analog is called HP-365 [59142-29-9].
An older synthesis is available, although more modern methods exist now:[4]
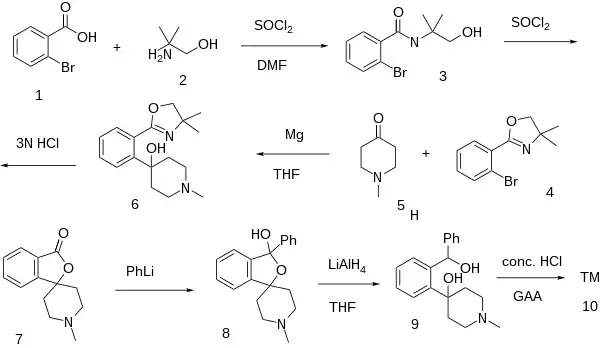
HP-365 patent:[5]
References
- 1 2 3 Meyerson LR, Ong HH, Martin LL, Ellis DB (June 1980). "Effect of antidepressant agents on beta-adrenergic receptor and neurotransmitter regulatory systems". Pharmacology, Biochemistry, and Behavior. 12 (6): 943–948. doi:10.1016/0091-3057(80)90457-8. PMID 6105676. S2CID 45400599.
- ↑ Klioze SS, Bauer VJ, Geyer HM (April 1977). "Synthesis of spiro[isobenzofuran-1(3H),4'-piperidines] as potential central nervous system agents. 2. Compounds containing a heteroatom attached to nitrogen". Journal of Medicinal Chemistry. 20 (4): 610–612. doi:10.1021/jm00214a039. PMID 850252.
- ↑ Puri SK, Mantione CR, Petko W, Ellis DB (1979). "Spiroisobenzofuran Piperidine Derivatives. Interaction with Pre- and Post-Synaptic Dopaminergic Sites". In Usdin E, Kopin IJ, Barchas J (eds.). Catecholamines: Basic and Clinical Frontiers. Pergamon. pp. 514–516. doi:10.1016/B978-1-4832-8363-0.50158-0. ISBN 978-1-4832-8363-0.
- ↑ Maier, C. A., Wünsch, B. (1 January 2002). "Novel Spiropiperidines as Highly Potent and Subtype Selective σ-Receptor Ligands. Part 1". Journal of Medicinal Chemistry. 45 (2): 438–448. doi:10.1021/jm010992z. ISSN 0022-2623.
- ↑ Victor J. Bauer & Raymond W. Kosley, Jr., U.S. Patent 3,959,475 (1976 to CNA Holdings LLC).
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.