Bacterial therapy
Bacterial therapy is the therapeutic use of bacteria to treat diseases. Bacterial therapeutics are living medicines, and may be wild type bacteria (often in the form of probiotics) or bacteria that have been genetically engineered to possess therapeutic properties that is injected into a patient.[1][2] Other examples of living medicines include cellular therapeutics (including immunotherapeutics) and phage therapeutics.
Development of bacterial therapeutics
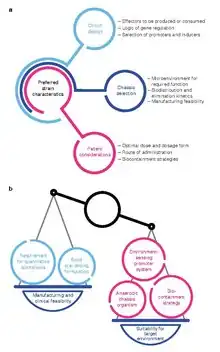
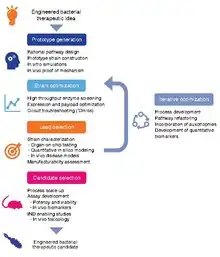
Development of bacterial therapeutics is an extremely active research area in the fields of synthetic biology and microbiology.[5][6][7][8][9][10][11][12][13] Currently, there is a large focus on: 1) identifying bacteria that naturally produce therapeutic effects (for example, probiotic bacteria), and 2) genetically programming bacteria to produce therapeutic effects.[14][15][16]
Applications
Cancer therapy


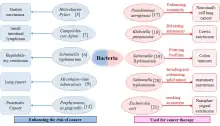
There is tremendous interest in using bacteria as a therapy to treat tumors. In particular, tumor-homing bacteria that thrive in hypoxic environments are particularly attractive for this purpose, as they will tend to migrate to, invade (through the leaky vasculature in the tumor microenvironment) and colonize tumors. This property tends to increase their residence time in the tumor, giving them longer to exert their therapeutic effects, in contrast to other bacteria that would be quickly cleared by the immune system.[18][19][20]
Microbiome engineering
.pdf.jpg.webp)
There is considerable interest in using bacterial therapeutics to alter human gastrointestinal microbiota, which would be helpful in treating diseases like small intestinal bacterial overgrowth and other forms of dysbiosis.
References
- ↑ editor, Ian Sample Science (16 January 2019). "'Living medicine' helps make toxic ammonia breakthrough". The Guardian. Retrieved 5 April 2020.
{{cite news}}:|last1=has generic name (help) - ↑ "Engineering Living Medicines for Chronic Diseases | SBE | Society for Biological Engineering". www.aiche.org.
- ↑
 This article incorporates text by Mark R. Charbonneau, Vincent M. Isabella, Ning Li & Caroline B. Kurtz available under the CC BY 4.0 license.
This article incorporates text by Mark R. Charbonneau, Vincent M. Isabella, Ning Li & Caroline B. Kurtz available under the CC BY 4.0 license. - ↑
 This article incorporates text by Mark R. Charbonneau, Vincent M. Isabella, Ning Li & Caroline B. Kurtz available under the CC BY 4.0 license.
This article incorporates text by Mark R. Charbonneau, Vincent M. Isabella, Ning Li & Caroline B. Kurtz available under the CC BY 4.0 license. - ↑ Weber, Wilfried; Fussenegger, Martin (January 2012). "Emerging biomedical applications of synthetic biology". Nature Reviews Genetics. 13 (1): 21–35. doi:10.1038/nrg3094. ISSN 1471-0056. PMC 7097403. PMID 22124480.
- ↑ Fischbach, M. A.; Bluestone, J. A.; Lim, W. A. (2013-04-03). "Cell-Based Therapeutics: The Next Pillar of Medicine". Science Translational Medicine. 5 (179): 179ps7. doi:10.1126/scitranslmed.3005568. ISSN 1946-6234. PMC 3772767. PMID 23552369.
- ↑ Kitada, Tasuku; DiAndreth, Breanna; Teague, Brian; Weiss, Ron (2018-02-09). "Programming gene and engineered-cell therapies with synthetic biology". Science. 359 (6376): eaad1067. doi:10.1126/science.aad1067. ISSN 0036-8075. PMC 7643872. PMID 29439214.
- ↑ McCarty, Niko (18 December 2018). "Why 2018 Was the Year of 'Living' Medicine". Medium. Medium. Retrieved 5 April 2020.
- ↑ Kelly, Jason (12 June 2019). "The Era of Living Medicines". Ginkgo Bioworks. Retrieved 5 April 2020.
- ↑ ServiceFeb. 18, Robert F. (18 February 2020). "From 'living' cement to medicine-delivering biofilms, biologists remake the material world". AAAS. Retrieved 5 April 2020.
- ↑ Kurtz, Caroline B.; Millet, Yves A.; Puurunen, Marja K.; Perreault, Mylène; Charbonneau, Mark R.; Isabella, Vincent M.; Kotula, Jonathan W.; Antipov, Eugene; Dagon, Yossi; Denney, William S.; Wagner, David A. (2019-01-16). "An engineered E. coli Nissle improves hyperammonemia and survival in mice and shows dose-dependent exposure in healthy humans". Science Translational Medicine. 11 (475): eaau7975. doi:10.1126/scitranslmed.aau7975. ISSN 1946-6234. PMID 30651324. S2CID 58031579.
- ↑ Charbonneau, Mark R.; Isabella, Vincent M.; Li, Ning; Kurtz, Caroline B. (2020-04-08). "Developing a new class of engineered live bacterial therapeutics to treat human diseases". Nature Communications. 11 (1): 1738. Bibcode:2020NatCo..11.1738C. doi:10.1038/s41467-020-15508-1. ISSN 2041-1723. PMC 7142098. PMID 32269218.
- ↑ "Gene Circuits Empower Next-Generation Cell and Gene Therapies". GEN - Genetic Engineering and Biotechnology News. 1 February 2020. Retrieved 5 April 2020.
- ↑ "Why now is the time for programmable living medicines: insights from Jim Collins, Aoife Brennan, and Jason Kelly". SynBioBeta. SynBioBeta. 2 April 2019. Retrieved 5 April 2020.
- ↑ Costa, Kevin (20 February 2019). "Living medicines: Ginkgo's machine to disrupt the pharma industry". SynBioBeta. SynBioBeta. Retrieved 5 April 2020.
- ↑ Sieow, Brendan Fu-Long; Wun, Kwok Soon; Yong, Wei Peng; Hwang, In Young; Chang, Matthew Wook (December 2020). "Tweak to Treat: Reprograming Bacteria for Cancer Treatment". Trends in Cancer. 7 (5): 447–464. doi:10.1016/j.trecan.2020.11.004. ISSN 2405-8033. PMID 33303401.
- ↑
 This article incorporates text by Mai Thi-Quynh Duong, Yeshan Qin, Sung-Hwan You & Jung-Joon Min available under the CC BY 4.0 license.
This article incorporates text by Mai Thi-Quynh Duong, Yeshan Qin, Sung-Hwan You & Jung-Joon Min available under the CC BY 4.0 license. - ↑ Duong, Mai Thi-Quynh; Qin, Yeshan; You, Sung-Hwan; Min, Jung-Joon (2019-12-11). "Bacteria-cancer interactions: bacteria-based cancer therapy". Experimental & Molecular Medicine. 51 (12): 1–15. doi:10.1038/s12276-019-0297-0. ISSN 2092-6413. PMC 6906302. PMID 31827064.
- ↑ Sedighi, Mansour; Zahedi Bialvaei, Abed; Hamblin, Michael R.; Ohadi, Elnaz; Asadi, Arezoo; Halajzadeh, Masoumeh; Lohrasbi, Vahid; Mohammadzadeh, Nima; Amiriani, Taghi; Krutova, Marcela; Amini, Abolfazl (2019-04-05). "Therapeutic bacteria to combat cancer; current advances, challenges, and opportunities". Cancer Medicine. 8 (6): 3167–3181. doi:10.1002/cam4.2148. ISSN 2045-7634. PMC 6558487. PMID 30950210.
- ↑ Song, Shiyu; Vuai, Miza S.; Zhong, Mintao (2018-03-15). "The role of bacteria in cancer therapy – enemies in the past, but allies at present". Infectious Agents and Cancer. 13 (1): 9. doi:10.1186/s13027-018-0180-y. ISSN 1750-9378. PMC 5856380. PMID 29568324.