C3a (complement)
C3a is one of the proteins formed by the cleavage of complement component 3; the other is C3b. C3a is a 77 residue anaphylatoxin that binds to the C3a receptor (C3aR), a class A G protein-coupled receptor. It plays a large role in the immune response.
C3a molecules induce responses through the GPCR C3a receptor. Like other anaphylatoxins, C3a is regulated by cleavage of its carboxy-terminal arginine, which results in a molecule with lowered inflammatory function (C3a desarginine).[1]
C3a is an effector of the complement system with a range of functions including T cell activation and survival,[2] angiogenesis stimulation,[3] chemotaxis, mast cell degranulation, and macrophage activation.[4] It has been shown to have both proinflammatory and anti-inflammatory responses, its activity able to counteract the proinflammatory effects of C5a.[5]
Structure
C3a
C3a is a strongly basic and highly cationic 77 residue protein with a molecular mass of approximately 10 kDa.[6] Residues 17-66 are made up of three anti-parallel helices and three disulfide bonds, which confer stability to the protein. The N-terminus consists of a fourth flexible helical structure, while the C terminus is disordered.[7] C3a has a regulatory process and a structure homologous to complement component C5a, with which it shares 36% of its sequence identity.[1]
Receptor
C3a induces an immunological response through a 482 residue G-protein-coupled receptor called C3aR. The C3aR is similarly structurally homologous to C5aR, but contains an extracellular domain with more than 160 amino acids.[8] Specific binding sites for interactions between C3a and C3aR are unknown, but it has been shown that sulfation of tyrosine 174, one of the amino acids in the extracellular domain, is required for C3a binding.[9] It has also been demonstrated that the C3aR N terminus is not required for ligand binding.[10]
Formation
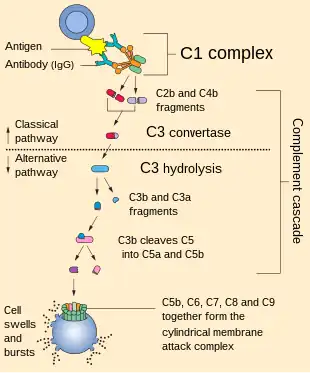
C3a formation occurs through activation and cleavage of complement component 3 in a reaction catalyzed by C3-convertase. There are three pathways of activation, each of which leads to the formation of C3a and C3b, which is involved in antigen opsonization. Other than the alternative pathway, which is constantly active, C3a formation is triggered by pathogenic infection.
Classical pathway
The classical pathway of complement activation is initiated when the C1 complex, made up of C1r and C1s serine proteases, recognizes the Fc region of IgM or IgG antibodies bound to a pathogen. C1q mediates the classical pathway by activating the C1 complex, which cleaves C4 and C2 into smaller fragments (C4a, C4b, C2a, and C2b). C4a and C2b form C4bC2b, also known as C3 convertase.[11]
Lectin pathway
The lectin pathway is activated when pattern-recognition receptors, like mannan-binding lectin or ficolins, recognize and bind to pathogen-associated molecular patterns on the antigen, including sugars.[12] These bound receptors then complex with Mannose-Binding Lectin-Associated Serine Proteases (MASPs), which have proteolytic activity similar to the C1 complex. The MASPs cleave C4 and C2, resulting in C3 convertase formation.[13]
Alternative pathway
The alternative pathway of complement activation is typically always active at low levels in blood plasma through a process called tick-over, in which C3 spontaneously hydrolyzes into its active form, C3(H2O). This activation induces a conformational change in the thioester domain of C3(H2O) that allows it to bind to a plasma protein called Factor B. This complex is then cleaved by Factor D, a serine protease, to form C3b(H2O)Bb, or fluid-phase C3-convertase. This complex has the ability to catalyze the formation of C3a and C3b after it binds properdin, a globulin protein, and is stabilized.[14]
Functions
Anaphylatoxins are small complement peptides that induce proinflammatory responses in tissues. C3a is primarily regarded for its role in the innate and adaptive immune responses as an anaphylatoxin, moderating and activating multiple inflammatory pathways.
Role in innate immunity
The roles of C3a in innate immunity, upon binding C3aR, include increased vasodilation via endothelial cell contraction, increased vascular permeability, and mast cell and basophil degranulation of histamine, induction of respiratory burst and subsequent degradation of pathogens by neutrophils, macrophages, and eosinophils, and regulation of cationic eosinophil protein migration, adhesion, and production.[15] C3a is also able to play a role in chemotaxis for mast cells and eosinophils, but C5a is a more potent chemoattractant.[16]
Traditionally thought to serve a strictly pro-inflammatory role, recent investigations have shown that C3a can also work against C5a to serve an anti-inflammatory role. In addition, migration and degranulation of neutrophils can be suppressed in the presence of C3a.[5]
Role in adaptive immunity
C3a also plays an important role in adaptive immunity, moderating leukocyte production and proliferation. C3a is able to regulate B cell and monocyte production of IL-6 and TNF-α, and human C3a has been shown to dampen the polyclonal immune response through dose-dependent regulation of B cell molecule production.[17] C3aR signaling along antigen-presenting cells' CD28 and CD40L pathways also plays a role in T cell proliferation and differentiation.[2] C3aR has been shown to be necessary for TH1 cell generation and regulates TH1 IL-10 expression, while an absence of active C3aR on dendritic cells upregulates regulatory T cell production. The absence of C3 has also been shown to decrease IL-2 receptor expression on T cells.[17]
Regulation
Regulation of complement activation
Levels of complement are regulated by moderating convertase formation and enzymatic activity. C3 convertase formation is primarily regulated by levels of active C3b and C4b. Factor I, a serine protease activated by cofactors, can cleave and C3b and C4b, thus preventing convertase formation. C3 convertase activity is also regulated without C3b inactivation, through complement control proteins, including decay-accelerating factors that function to speed up C3 convertase half-lives and avert convertase formation.[12]
Deactivation
C3a, like other anaphylatoxins, has a C-terminal arginine residue. Serum carboxypeptidase B, a protease, cleaves the arginine residue from C3a, forming the desArg derivative of C3a, also known as acylation stimulating protein (ASP). Unlike C5a desArg, this version of C3a has no proinflammatory activity.[1] However, ASP functions as a hormone in the adipose tissue, moderating fatty acid migration to adipocytes and triacylglycerol synthesis.[18] In addition, it has been shown that ASP downregulates the polyclonal immune response in the same way C3a does.[12]
References
- 1 2 3 Bajic, Goran; Yatime, Laure; Klos, Andreas; Andersen, Gregers Rom (2013-02-01). "Human C3a and C3a desArg anaphylatoxins have conserved structures, in contrast to C5a and C5a desArg". Protein Science. 22 (2): 204–212. doi:10.1002/pro.2200. ISSN 1469-896X. PMC 3588916. PMID 23184394.
- 1 2 Strainic, MG; Liu, J; Huang, D; An, F; Lalli, PN; Muqim, N; Shapiro, VS; Dubyak, GR; Heeger, PS; Medof, ME (March 2008). "Locally produced complement fragments C5a and C3a provide both costimulatory and survival signals to naive CD4+ T cells". Immunity. 28 (3): 425–35. doi:10.1016/j.immuni.2008.02.001. PMC 2646383. PMID 18328742.
- ↑ Khan, MA; Assiri, AM; Broering, DC (22 July 2015). "Complement and macrophage crosstalk during process of angiogenesis in tumor progression". Journal of Biomedical Science. 22: 58. doi:10.1186/s12929-015-0151-1. PMC 4511526. PMID 26198107.
- ↑ Mathern, DR; Heeger, PS (4 September 2015). "Molecules Great and Small: The Complement System". Clinical Journal of the American Society of Nephrology. 10 (9): 1636–50. doi:10.2215/cjn.06230614. PMC 4559511. PMID 25568220.
- 1 2 Coulthard, LG; Woodruff, TM (15 April 2015). "Is the complement activation product C3a a proinflammatory molecule? Re-evaluating the evidence and the myth". Journal of Immunology. 194 (8): 3542–8. doi:10.4049/jimmunol.1403068. PMID 25848071.
- ↑ Zhou, Wuding (2012-02-01). "The new face of anaphylatoxins in immune regulation". Immunobiology. 217 (2): 225–234. doi:10.1016/j.imbio.2011.07.016. ISSN 1878-3279. PMID 21856033.
- ↑ Chang, Jui-Yoa; Lin, Curtis C. -J.; Salamanca, Silvia; Pangburn, Michael K.; Wetsel, Rick A. (2008-12-15). "Denaturation and unfolding of human anaphylatoxin C3a: An unusually low covalent stability of its native disulfide bonds". Archives of Biochemistry and Biophysics. 480 (2): 104–110. doi:10.1016/j.abb.2008.09.013. PMC 2636726. PMID 18854167.
- ↑ Ames, R. S.; Li, Y.; Sarau, H. M.; Nuthulaganti, P.; Foley, J. J.; Ellis, C.; Zeng, Z.; Su, K.; Jurewicz, A. J. (1996-08-23). "Molecular cloning and characterization of the human anaphylatoxin C3a receptor". The Journal of Biological Chemistry. 271 (34): 20231–20234. doi:10.1074/jbc.271.34.20231. ISSN 0021-9258. PMID 8702752.
- ↑ Gao, Jinming; Choe, Hyeryun; Bota, Dalena; Wright, Paulette L.; Gerard, Craig; Gerard, Norma P. (2003-09-26). "Sulfation of tyrosine 174 in the human C3a receptor is essential for binding of C3a anaphylatoxin". The Journal of Biological Chemistry. 278 (39): 37902–37908. doi:10.1074/jbc.M306061200. ISSN 0021-9258. PMID 12871936.
- ↑ Crass, T.; Ames, R. S.; Sarau, H. M.; Tornetta, M. A.; Foley, J. J.; Köhl, J.; Klos, A.; Bautsch, W. (1999-03-26). "Chimeric receptors of the human C3a receptor and C5a receptor (CD88)". The Journal of Biological Chemistry. 274 (13): 8367–8370. doi:10.1074/jbc.274.13.8367. ISSN 0021-9258. PMID 10085065.
- ↑ Arlaud, G. J.; Gaboriaud, C.; Thielens, N. M.; Rossi, V.; Bersch, B.; Hernandez, J. F.; Fontecilla-Camps, J. C. (2001-04-01). "Structural biology of C1: dissection of a complex molecular machinery". Immunological Reviews. 180: 136–145. doi:10.1034/j.1600-065x.2001.1800112.x. ISSN 0105-2896. PMID 11414355.
- 1 2 3 Dunkelberger, Jason R.; Song, Wen-Chao (2010-01-01). "Complement and its role in innate and adaptive immune responses". Cell Research. 20 (1): 34–50. doi:10.1038/cr.2009.139. ISSN 1748-7838. PMID 20010915.
- ↑ Degn, Søren E.; Thiel, Steffen; Jensenius, Jens C. (2007-01-01). "New perspectives on mannan-binding lectin-mediated complement activation". Immunobiology. 212 (4–5): 301–311. doi:10.1016/j.imbio.2006.12.004. ISSN 0171-2985. PMID 17544815.
- ↑ Merle, Nicolas S.; Church, Sarah Elizabeth; Fremeaux-Bacchi, Veronique; Roumenina, Lubka T. (2015-01-01). "Complement System Part I - Molecular Mechanisms of Activation and Regulation". Frontiers in Immunology. 6: 262. doi:10.3389/fimmu.2015.00262. ISSN 1664-3224. PMC 4451739. PMID 26082779.
- ↑ Klos, Andreas; Tenner, Andrea J.; Johswich, Kay-Ole; Ager, Rahasson R.; Reis, Edimara S.; Köhl, Jörg (2009-09-01). "The role of the anaphylatoxins in health and disease". Molecular Immunology. 12th European Meeting on Complement in Human Disease12th European Meeting on CHD12th European Meeting on Complement in Human Disease. 46 (14): 2753–2766. doi:10.1016/j.molimm.2009.04.027. PMC 2725201. PMID 19477527.
- ↑ Schraufstatter, Ingrid U.; DiScipio, Richard G.; Zhao, Ming; Khaldoyanidi, Sophia K. (2009-03-15). "C3a and C5a Are Chemotactic Factors for Human Mesenchymal Stem Cells, Which Cause Prolonged ERK1/2 Phosphorylation". The Journal of Immunology. 182 (6): 3827–3836. doi:10.4049/jimmunol.0803055. ISSN 0022-1767. PMID 19265162.
- 1 2 Merle, Nicolas S.; Noe, Remi; Halbwachs-Mecarelli, Lise; Fremeaux-Bacchi, Veronique; Roumenina, Lubka T. (2015-05-26). "Complement System Part II: Role in Immunity". Frontiers in Immunology. 6: 257. doi:10.3389/fimmu.2015.00257. ISSN 1664-3224. PMC 4443744. PMID 26074922.
- ↑ Barbu, Andreea; Hamad, Osama A.; Lind, Lars; Ekdahl, Kristina N.; Nilsson, Bo (2015-09-01). "The role of complement factor C3 in lipid metabolism". Molecular Immunology. 15th European Meeting on Complement in Human Disease 2015, Uppsala, Sweden. 67 (1): 101–107. doi:10.1016/j.molimm.2015.02.027. PMID 25746915.
- Dinasarapu, A R; Chandrasekhar, A; Sahu, A; Subramaniam, S (2012). "Complement C3 (Human)". UCSD Molecule Pages. doi:10.6072/H0.MP.A004235.01.