EMM (psychedelic)
 | |
| Names | |
|---|---|
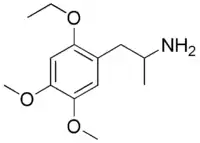
| Preferred IUPAC name
1-(2,4,5-Trimethoxyphenyl)propan-2-amine | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C13H21NO3 |
| Molar mass | 239.315 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
EMM (2-ethoxy-4,5-dimethoxyamphetamine) is a lesser-known substituted amphetamine. It is a dimethoxy-ethoxy analog of trimethoxyamphetamine (TMA-2). EMM was first synthesized by Alexander Shulgin. In his book PiHKAL, both the dosage and duration are unknown.[1] EMM produces few to no effects. Very little data exists about the pharmacological properties, metabolism, and toxicity of EMM.
See also
References
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.