Essential fatty acid interactions
The effects of the ω-3 (omega-3) and ω-6 (omega-6) essential fatty acids (EFAs) are characterized by their interactions.
Arachidonic acid (AA) is a 20-carbon ω-6 essential fatty acid.[1] It sits at the head of the "arachidonic acid cascade" that initiates 20 different signaling paths that control a wide array of biological functions, including inflammation, cell growth and the central nervous system.[2][3] Most AA in the human body is derived from dietary linoleic acid (18:2 ω-6), found in nuts, seeds, vegetable oils and animal fats.[4][5][6]
During inflammation two other groups of dietary essential fatty acids form cascades that compete with the arachidonic acid cascade. EPA (20:5 ω-3) provides the most important competing cascade. EPA is ingested from oily fish, algae oil, or alpha-linolenic acid (derived from walnuts, hemp oil and flax oil). DGLA (20:3 ω-6) provides a third, less prominent cascade. It is derived from dietary GLA (18:3 ω-6) found in borage oil. These two parallel cascades soften the inflammatory promoting effects of specific eicosanoids made from AA.
Today, the typical diet in industrial countries contains much less ω-3 fatty acids than the diet of a century ago and a much greater amount of air pollution on a daily basis that evokes the inflammatory response. The diet from a century ago had much less ω-3 than the diet of early hunter-gatherers but also much less pollution than today.[7] We can also look at the ratio of ω-3 to ω-6 in comparisons of their diets. These changes have been accompanied by increased rates of many diseases – the so-called diseases of civilization – that involve inflammatory processes. There is now very strong evidence[8] that several of these diseases are ameliorated by increasing dietary ω-3. There is also more preliminary evidence showing that dietary ω-3 can ease symptoms in several psychiatric disorders.[9]
Eicosanoid series nomenclature
Eicosanoids are signalling molecules derived from the essential fatty acids (EFA); they are a major pathway by which the EFAs act in the body. There are four classes of eicosanoid and two or three series within each class. Before discussing eicosanoid action, we will explain the series nomenclature.
The plasma membranes of cells contain phospholipids, which are composed of a hydrophilic phosphate head and two hydrophobic fatty acid tails. Some of these fatty acids are 20-carbon polyunsaturated essential fatty acids – AA, EPA or DGLA. In response to a variety of inflammatory signals, these EFAs are cleaved out of the phospholipid and released as free fatty acids. Next, the EFA is oxygenated (by either of two pathways), then further modified, yielding the eicosanoids. Cyclooxygenase (COX) oxidation removes two C=C double bonds, leading to the TX, PG and PGI series. Lipoxygenase oxidation removes no C=C double bonds, and leads to the LK.[10]
After oxidation, the eicosanoids are further modified, making a series. Members of a series are differentiated by an ABC... letter, and are numbered by the number of double bonds, which does not change within a series. For example, cyclooxygenase action upon AA (with 4 double bonds) leads to the series-2 thromboxanes (TXA2, TXB2... ) each with two double bonds. Cyclooxygenase action on EPA (with 5 double bonds) leads to the series-3 thromboxanes (TXA3, TXB3... ) each with three double bonds. There are exceptions to this pattern, some of which indicate stereochemistry (PGF2α).
Table (1) shows these sequences for AA (20:4 ω-6). The sequences for EPA (20:5 ω-3) and DGLA (20:3 ω-6) are analogous.
| Dietary Essential Fatty Acid |
Abbr | Formula carbons:double bonds ω |
Eicosanoid product series | ||
|---|---|---|---|---|---|
| TX PG PGI |
LK | Effects | |||
| Gamma-linolenic acid via Dihomo gamma linolenic acid |
GLA DGLA |
18:3ω6 20:3ω6 |
series-1 | series-3 | less inflammatory |
| Arachidonic acid | AA | 20:4ω6 | series-2 | series-4 | more inflammatory |
| Eicosapentaenoic acid | EPA | 20:5ω3 | series-3 | series-5 | less inflammatory |
All the prostenoids are substituted prostanoic acids. Cyberlipid Center's Prostenoid page[11] illustrates the parent compound and the rings associated with each series–letter.
The IUPAC and the IUBMB use the equivalent term Icosanoid.[11]
Arachidonic acid cascade in inflammation
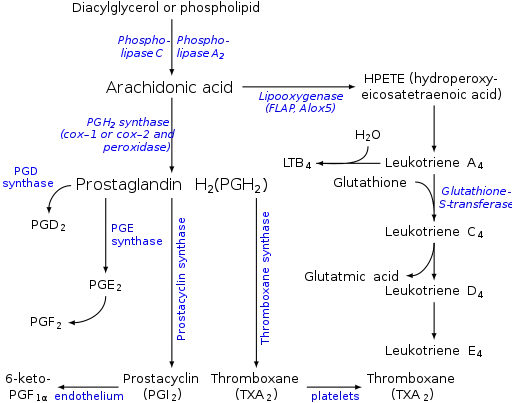
In the arachidonic acid cascade, dietary linoleic acid (18:2 ω-6) is desaturated and elongated to form arachidonic acid (and also other omega 6), esterified into a phospholipid in the cell membrane.[12] Next, in response to many inflammatory stimuli, such as air pollution, smoking, second-hand smoke, hydrogenated vegetable oils and other exogenous toxins; phospholipase is generated and cleaves this phospholipid, releasing AA as a free fatty acid. AA can then be oxygenated and then further modified to form eicosanoids – autocrine and paracrine agents that bind receptors on the cell or its neighbors to alert the immune system of the cell damage. Alternatively, AA can diffuse into the cell nucleus and interact with transcription factors to control DNA transcription for cytokines or other hormones.
Mechanisms of ω-3 eicosanoid action
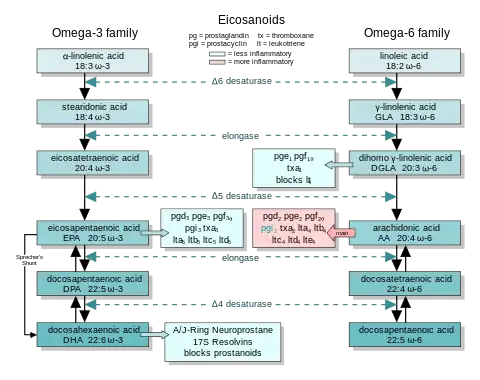
The eicosanoids from AA generally promote inflammation. Those from GLA (via DGLA) and from EPA are generally less inflammatory, or inactive, or even anti-inflammatory. (This generalization is qualified: an eicosanoid may be pro-inflammatory in one tissue and anti-inflammatory in another. See discussion of PGE2 at Calder[13] or Tilley.[14])
Figure (2) shows the ω-3 and -6 synthesis chains, along with the major eicosanoids from AA, EPA and DGLA.
Dietary ω-3 and GLA counter the inflammatory effects of AA's eicosanoids in three ways – displacement, competitive inhibition and direct counteraction.
Displacement
Dietary ω-3 decreases tissue concentrations of AA. Animal studies show that increased dietary ω-3 results in decreased AA in brain and other tissue.[15] Linolenic acid (18:3 ω-3) contributes to this by displacing linoleic acid (18:2 ω-6) from the elongase and desaturase enzymes that produce AA. EPA inhibits phospholipase A2's release of AA from cell membrane.[16] Other mechanisms involving the transport of EFAs may also play a role.
The reverse is also true – high dietary linoleic acid decreases the body's conversion of α-linolenic acid to EPA. However, the effect is not as strong; the desaturase has a higher affinity for α-linolenic acid than it has for linoleic acid.[17]
Competitive Inhibition
DGLA and EPA compete with AA for access to the cyclooxygenase and lipoxygenase enzymes. So the presence of DGLA and EPA in tissues lowers the output of AA's eicosanoids. For example, dietary GLA increases tissue DGLA and lowers TXB2.[18][19] Likewise, EPA inhibits the production of series-2 PG and TX.[13] Although DGLA forms no LTs, a DGLA derivative blocks the transformation of AA to LTs.[20]
Counteraction
Some DGLA and EPA derived eicosanoids counteract their AA derived counterparts. For example, DGLA yields PGE1, which powerfully counteracts PGE2.[21] EPA yields the antiaggregatory prostacyclin PGI3 [22] It also yields the leuokotriene LTB5 which vitiates the action of the AA-derived LTB4.[23]
The paradox of dietary GLA
Dietary oxidized linoleic acid (LA, 18:2 ω-6) is inflammatory. In the body, LA is desaturated to form GLA (18:3 ω-6), yet dietary GLA is anti-inflammatory. Some observations partially explain this paradox: LA competes with α-linolenic acid, (ALA, 18:3 ω-3) for Δ6-desaturase, and thereby eventually inhibits formation of anti-inflammatory EPA (20:5 ω-3). In contrast, GLA does not compete for Δ6-desaturase. GLA's elongation product DGLA (20:3 ω-6) competes with 20:4 ω-3 for the Δ5-desaturase, and it might be expected that this would make GLA inflammatory, but it is not, perhaps because this step isn't rate-determining. Δ6-desaturase does appear to be the rate-limiting step; 20:4 ω-3 does not significantly accumulate in bodily lipids.
DGLA inhibits inflammation through both competitive inhibition and direct counteraction (see above.) Dietary GLA leads to sharply increased DGLA in the white blood cells' membranes, where LA does not. This may reflect white blood cells' lack of desaturase. Supplementing dietary GLA increases serum DGLA without increasing serum AA.[21][24]
It is likely that some dietary GLA eventually forms AA and contributes to inflammation. Animal studies indicate the effect is small.[19] The empirical observation of GLA's actual effects argues that DGLA's anti-inflammatory effects dominate.[25]
Complexity of pathways
Eicosanoid signaling paths are complex. It is therefore difficult to characterize the action of any particular eicosanoid. For example, PGE2 binds four receptors, dubbed EP1–4. Each is coded by a separate gene, and some exist in multiple isoforms. Each EP receptor in turn couples to a G protein. The EP2, EP4 and one isoform of the EP3 receptors couple to Gs. This increases intracellular cAMP and is anti-inflammatory. EP1 and other EP3 isoforms couple to Gq. This leads to increased intracellular calcium and is pro-inflammatory. Finally, yet another EP3 isoform couples to Gi, which both decreases cAMP and increases calcium. Many immune-system cells express multiple receptors that couple these apparently opposing pathways.[14] Presumably, EPA-derived PGE3 has a somewhat different effect of on this system, but it is not well-characterized.
The arachidonic acid cascade in the central nervous system (CNS)
The arachidonic acid cascade is arguably the most elaborate signaling system neurobiologists have to deal with.
Daniele Piomelli Arachidonic Acid[3]
The arachidonic acid cascade proceeds somewhat differently in the brain. Neurohormones, neuromodulators or neurotransmitters act as first messengers. They activate phospholipidase to release AA from neuron cell membranes as a free fatty acid. During its short lifespan, free AA may affect the activity of the neuron's ion channels and protein kinases. Or it may be metabolized to form eicosanoids, epoxyeicosatrienoic acids (EETs), neuroprotectin D or various endocannabinoids (anandamide and its analogs.)
The actions of eicosanoids within the brain are not as well characterized as they are in inflammation. It is theorized that they act within the neuron as second messengers controlling presynaptic inhibition and the activation of protein kinase C. They also act as paracrine mediators, acting across synapses to nearby cells. Although detail on the effects of these signals is scant, (Piomelli, 2000) comments
Neurons in the CNS are organized as interconnected groups of functionally related cells (e.g., in sensory systems). A diffusible factor released from a neuron into the interstitial fluid, and able to interact with membrane receptors on adjacent cells, would be ideally used to "synchronize" the activity of an ensemble of interconnected neural cells. Furthermore, during development and in certain forms of learning, postsynaptic cells may secrete regulatory factors which diffuse back to the presynaptic component, determining its survival as an active terminal, the amplitude of its sprouting, and its efficacy in secreting neurotransmitters—a phenomenon known as retrograde regulation. The participation of arachidonic acid metabolites in retrograde signaling and in other forms of local modulation of neuronal activity has been proposed.
| Arachidonic Acid Cascade | ||
|---|---|---|
| In inflammation | In the brain | |
| Major effect on | Inflammation in tissue | Neuronal excitability |
| AA released from | White blood cells | Neurons |
| Triggers for AA release | Inflammatory stimuli | Neurotransmitters, neurohormones and neuromodulators |
| Intracellular effects on | DNA transcription of cytokines and other mediators of inflammation |
Activity of ion channels and protein kinases |
| Metabolized to form | Eicosanoids, resolvins, isofurans, isoprostanes, lipoxins, epoxyeicosatrienoic acids (EETs) |
Eicosanoids, neuroprotectin D, EETs and some endocannabinoids |
The EPA and DGLA cascades are also present in the brain and their eicosanoid metabolites have been detected. The ways in which these differently affect mental and neural processes are not nearly as well characterized as are the effects in inflammation.
Further discussion
Figure (2) shows two pathways from EPA to DHA, including the exceptional Sprecher's shunt.
5-LO acts at the fifth carbon from the carboxyl group. Other lipoxygenases—8-LO, 12-LO and 15-LO—make other eicosanoid-like products. To act, 5-LO uses the nuclear-membrane enzyme 5-lipoxygenase-activating protein (FLAP), first to a hydroperoxyeicosatetraenoic acid (HPETE), then to the first leuokotriene, LTA.
See also
References
- ↑ Cunnane SC (November 2003). "Problems with essential fatty acids: time for a new paradigm?". Progress in Lipid Research. 42 (6): 544–568. doi:10.1016/S0163-7827(03)00038-9. PMID 14559071.
- ↑ de Melo Reis RA, Isaac AR, Freitas HR, de Almeida MM, Schuck PF, Ferreira GC, et al. (2021). "Quality of Life and a Surveillant Endocannabinoid System". Frontiers in Neuroscience. 15: 747229. doi:10.3389/fnins.2021.747229. PMC 8581450. PMID 34776851.
- 1 2 Piomelli, Daniele (2000). "Arachidonic Acid". Neuropsychopharmacology: The Fifth Generation of Progress. Archived from the original on 2006-07-15. Retrieved 2006-03-03.
- ↑ Freitas HR (2017-08-25). "Chlorella vulgaris as a Source of Essential Fatty Acids and Micronutrients: A Brief Commentary". The Open Plant Science Journal. 10 (1): 92–99. doi:10.2174/1874294701710010092. ISSN 1874-2947.
- ↑ Freitas HR, Isaac AR, Malcher-Lopes R, Diaz BL, Trevenzoli IH, De Melo Reis RA (December 2018). "Polyunsaturated fatty acids and endocannabinoids in health and disease". Nutritional Neuroscience. 21 (10): 695–714. doi:10.1080/1028415X.2017.1347373. PMID 28686542.
- ↑ Freitas HR, Ferreira GD, Trevenzoli IH, Oliveira KJ, de Melo Reis RA (November 2017). "Fatty Acids, Antioxidants and Physical Activity in Brain Aging". Nutrients. 9 (11): 1263. doi:10.3390/nu9111263. PMC 5707735. PMID 29156608.
- ↑ Simopoulos A (2001). "Evolutionary aspects of diet and essential fatty acids". Fatty Acids and Lipids - New Findings (PDF). World Rev Nutr Diet. World Review of Nutrition and Dietetics. Vol. 88. pp. 18–27. doi:10.1159/000059742. ISBN 978-3-8055-7182-1. PMID 11935953. Archived from the original (PDF) on 2006-09-26.
- ↑ National Institute of Health (2005-08-01). "Omega-3 fatty acids, fish oil, alpha-linolenic acid". Archived from the original on February 8, 2006. Retrieved August 21, 2010.
- ↑ De Caterina R, Basta G (2001). "n-3 Fatty acids and the inflammatory response — biological background". European Heart Journal Supplements. 3 (suppl D): D42. doi:10.1016/S1520-765X(01)90118-X.
- ↑ Cyberlipid Center. "Polyenoic fatty acids". Archived from the original on September 30, 2018. Retrieved February 11, 2006.
- 1 2 Cyberlipid Center. "Prostanoids". Archived from the original on February 8, 2007. Retrieved February 11, 2006.
- ↑ Whelan J, Fritsche K (May 2013). "Linoleic acid". Advances in Nutrition. 4 (3): 311–312. doi:10.3945/an.113.003772. PMC 3650500. PMID 23674797.
- 1 2 Calder, Philip C. (September 2004). "n-3 Fatty Acids and Inflammation – New Twists in an Old Tale". Archived from the original on March 16, 2006. Retrieved February 8, 2006.
- Invited review article, PUFA Newsletter.
- 1 2 Tilley SL, Coffman TM, Koller BH (July 2001). "Mixed messages: modulation of inflammation and immune responses by prostaglandins and thromboxanes". The Journal of Clinical Investigation. 108 (1): 15–23. doi:10.1172/JCI13416. PMC 209346. PMID 11435451.
- ↑ Medical Study News (25 May 2005). "Brain fatty acid levels linked to depression". Retrieved February 10, 2006.
- Who were in turn citing Green P, Gispan-Herman I, Yadid G (June 2005). "Increased arachidonic acid concentration in the brain of Flinders Sensitive Line rats, an animal model of depression". Journal of Lipid Research. 46 (6): 1093–1096. doi:10.1194/jlr.C500003-JLR200. PMID 15805551.
- ↑ KP Su; SY Huang; CC Chiu; WW Shen (2003). "Omega-3 fatty acids in major depressive disorder. A preliminary double-blind, placebo-controlled ?" (PDF). Archived from the original (PDF) on February 8, 2005. Retrieved February 22, 2006.
- ↑ Phinney SD, Odin RS, Johnson SB, Holman RT (March 1990). "Reduced arachidonate in serum phospholipids and cholesteryl esters associated with vegetarian diets in humans". The American Journal of Clinical Nutrition. 51 (3): 385–392. doi:10.1093/ajcn/51.3.385. PMID 2106775. Archived from the original on February 12, 2007. Retrieved February 11, 2006.
- "[D]ietary arachidonic acid enriches its circulating pool in humans; however, 20:5n-3 is not similarly responsive to dietary restriction."
- ↑ Guivernau M, Meza N, Barja P, Roman O (November 1994). "Clinical and experimental study on the long-term effect of dietary gamma-linolenic acid on plasma lipids, platelet aggregation, thromboxane formation, and prostacyclin production". Prostaglandins, Leukotrienes, and Essential Fatty Acids. 51 (5): 311–316. doi:10.1016/0952-3278(94)90002-7. PMID 7846101.
- GLA decreases triglycerides, LDL, increases HDL, decreases TXB2 and other inflammatory markers. Review article; human and rat studies.
- 1 2 Karlstad MD, DeMichele SJ, Leathem WD, Peterson MB (November 1993). "Effect of intravenous lipid emulsions enriched with gamma-linolenic acid on plasma n-6 fatty acids and prostaglandin biosynthesis after burn and endotoxin injury in rats". Critical Care Medicine. 21 (11): 1740–1749. doi:10.1097/00003246-199311000-00025. PMID 8222692. S2CID 36538810.
- IV Supplementation with gamma-linolenic acid increased serum GLA but did not increase the plasma percentage of arachidonic acid (rat study), decreased TXB2.
- ↑ Belch JJ, Hill A (January 2000). "Evening primrose oil and borage oil in rheumatologic conditions". The American Journal of Clinical Nutrition. 71 (1 Suppl): 352S–356S. doi:10.1093/ajcn/71.1.352s. PMID 10617996.
- "DGLA itself cannot be converted to LTs but can form a 15-hydroxyl derivative that blocks the transformation of arachidonic acid to LTs. Increasing DGLA intake may allow DGLA to act as a competitive inhibitor of 2-series PGs and 4-series LTs and thus suppress inflammation."
- 1 2 Fan YY, Chapkin RS (September 1998). "Importance of dietary gamma-linolenic acid in human health and nutrition". The Journal of Nutrition. 128 (9): 1411–1414. doi:10.1093/jn/128.9.1411. PMID 9732298.
- "[D]ietary GLA increases the content of its elongase product, dihomo-gamma linolenic acid (DGLA), within cell membranes without concomitant changes in arachidonic acid (AA). Subsequently, upon stimulation, DGLA can be converted by inflammatory cells to 15-(S)-hydroxy-8,11,13-eicosatrienoic acid and prostaglandin E1. This is noteworthy because these compounds possess both anti-inflammatory and antiproliferative properties."
- ↑ Fischer S, Weber PC (September 1985). "Thromboxane (TX)A3 and prostaglandin (PG)I3 are formed in man after dietary eicosapentaenoic acid: identification and quantification by capillary gas chromatography-electron impact mass spectrometry". Biomedical Mass Spectrometry. 12 (9): 470–476. doi:10.1002/bms.1200120905. PMID 2996649.
- ↑ Prescott SM (June 1984). "The effect of eicosapentaenoic acid on leukotriene B production by human neutrophils". The Journal of Biological Chemistry. 259 (12): 7615–7621. doi:10.1016/S0021-9258(17)42835-3. PMID 6330066.
- ↑ Johnson MM, Swan DD, Surette ME, Stegner J, Chilton T, Fonteh AN, Chilton FH (August 1997). "Dietary supplementation with gamma-linolenic acid alters fatty acid content and eicosanoid production in healthy humans". The Journal of Nutrition. 127 (8): 1435–1444. doi:10.1093/jn/127.8.1435. PMID 9237935.
- ↑ Stone KJ, Willis AL, Hart WM, Kirtland SJ, Kernoff PB, McNicol GP (February 1979). "The metabolism of dihomo-gamma-linolenic acid in man". Lipids. 14 (2): 174–180. doi:10.1007/BF02533869. PMID 423720. S2CID 41372225.