Leptomycin
 | |
| Names | |
|---|---|
| Preferred IUPAC name
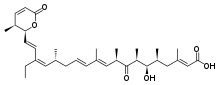
(2E,5S,6R,7S,9R,10E,12E,15R,16Z,18E)-17-Ethyl-6-hydroxy-3,5,7,9,11,15-hexamethyl-19-[(2S,3S)-3-methyl-6-oxo-3,6-dihydro-2H-pyran-2-yl]-8-oxononadeca-2,10,12,16,18-pentaenoic acid | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.125.530 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C33H48O6 |
| Molar mass | 540.741 g·mol−1 |
| Density | 1.072 g/mL |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Leptomycins are secondary metabolites produced by Streptomyces spp.
Leptomycin B (LMB) was originally discovered as a potent antifungal compound. [1] Leptomycin B was found to cause cell elongation of the fission yeast Schizosaccharomyces pombe. Since then this elongation effect has been used for the bioassay of leptomycin. However, recent data shows that leptomycin causes G1 cell cycle arrest in mammalian cells and is a potent anti-tumor agent against murine experimental tumors in combination therapy.[2]
Leptomycin B has been shown to be a potent and specific nuclear export inhibitor in human[3] and the fission yeast S. pombe.[4] Leptomycin B alkylates and inhibits CRM1 (chromosomal region maintenance)/exportin 1 (XPO1), a protein required for nuclear export of proteins containing a nuclear export sequence (NES), by glycosylating a cysteine residue (cysteine 529 in S. pombe).[5] In addition to antifungal and antibacterial activities, leptomycin B blocks the cell cycle and is a potent anti-tumor agent. At low nM concentrations, leptomycin B blocks the nuclear export of many proteins including HIV-1 Rev, MAPK/ERK, and NF-κB/IκB, and it inhibits the inactivation of p53.[6] Leptomycin B also inhibits the export and translation of many RNAs, including COX-2 and c-Fos mRNAs, by inhibiting export of ribonucleoproteins.
Leptomycin A (LPA) was discovered together with LMB. LMB is twice as potent as LPA.
See also
References
- ↑ Hamamoto T, Seto H, Beppu T (1983). "Leptomycins A and B, new antifungal antibiotics. II. Structure elucidation". J. Antibiot. 36 (6): 646–50. doi:10.7164/antibiotics.36.646. PMID 6874586.
- ↑ Lu, Chuanwen; Changxia Shao; Everardo Cobos; Kamaleshwar P. Singh; Weimin Gao (March 2012). "Chemotherapeutic Sensitization of Leptomycin B Resistant Lung Cancer Cells by Pretreatment with Doxorubicin". PLOS ONE. United States. 7 (3): e32895. Bibcode:2012PLoSO...732895L. doi:10.1371/journal.pone.0032895. ISSN 1932-6203. PMC 3296751. PMID 22412944.
- ↑ Kudo N, Wolff B, Sekimoto T, et al. (August 1998). "Leptomycin B inhibition of signal-mediated nuclear export by direct binding to CRM1". Exp. Cell Res. 242 (2): 540–7. doi:10.1006/excr.1998.4136. PMID 9683540.
- ↑ Nishi K, Yoshida M, Fujiwara D, Nishikawa M, Horinouchi S, Beppu T (March 1994). "Leptomycin B targets a regulatory cascade of crm1, a fission yeast nuclear protein, involved in control of higher order chromosome structure and gene expression". J. Biol. Chem. 269 (9): 6320–4. doi:10.1016/S0021-9258(17)37374-X. PMID 8119981.
- ↑ Kudo N, Matsumori N, Taoka H, et al. (August 1999). "Leptomycin B inactivates CRM1/exportin 1 by covalent modification at a cysteine residue in the central conserved region". Proc. Natl. Acad. Sci. U.S.A. 96 (16): 9112–7. Bibcode:1999PNAS...96.9112K. doi:10.1073/pnas.96.16.9112. PMC 17741. PMID 10430904.
- ↑ Hietanen S, Lain S, Krausz E, Blattner C, Lane DP (2000). "Activation of p53 in cervical carcinoma cells by small molecules". Proc Natl Acad Sci U S A. 97 (15): 8501–6. Bibcode:2000PNAS...97.8501H. doi:10.1073/pnas.97.15.8501. PMC 26977. PMID 10900010.
External links
- leptomycin+B at the US National Library of Medicine Medical Subject Headings (MeSH)
Original data copied with permission from Leptomycin B manufacturer product page (Fermentek)