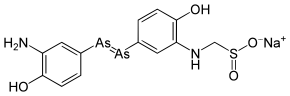
Neosalvarsan
 | |
 | |
| Names | |
|---|---|
| Other names
Sodium 3,3'-diamino-4,4'-dihydroxyarsenobenzene-N-formaldehydesulfoxylate; Neoarsphenamine;914 | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.006.613 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C13H13As2N2NaO4S |
| Molar mass | 466.15 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Neosalvarsan is a synthetic chemotherapeutic that is an organoarsenic compound. It became available in 1912 and superseded the more toxic and less water-soluble salvarsan as an effective treatment for syphilis. Because both of these arsenicals carried considerable risk of side effects, they were replaced for this indication by penicillin in the 1940s.
Both salvarsan and neosalvarsan were developed in the laboratory of Paul Ehrlich in Frankfurt, Germany. Their discoveries were the result of the first organized team effort to optimize the biological activity of a lead compound through systematic chemical modifications.[1] This scheme is the basis for most modern pharmaceutical research. Both salvarsan and neosalvarsan are prodrugs — that is, they are metabolised into the active drug in the body.
Although, like salvarsan, it was originally believed to contain an arsenic-arsenic double bond, this is now known to be incorrect, and exists as a mixture of differently sized rings with arsenic-arsenic single bonds.[2]
References
- ↑ Strebhardt K, Ullrich A (May 2008). "Paul Ehrlich's magic bullet concept: 100 years of progress". Nat. Rev. Cancer. 8 (6): 473–80. doi:10.1038/nrc2394. PMID 18469827. S2CID 30063909.
- ↑ Lloyd, Nicholas C.; Morgan, Hugh W.; Nicholson, Brian K.; Ronimus, Ron S. (2005). "The Composition of Ehrlich's Salvarsan: Resolution of a Century-Old Debate" (PDF). Angewandte Chemie International Edition. 44 (6): 941–944. doi:10.1002/anie.200461471. hdl:10289/207. ISSN 1521-3773. PMID 15624113.