Sevelamer
 | |
| Names | |
|---|---|
| Pronunciation | (/sɛˈvɛləmər/ or /sɛˈvɛləmɪər/) |
| Trade names | Renagel, Renvela |
IUPAC name
| |
| Clinical data | |
| Main uses | Hyperphosphatemia in chronic kidney disease[1] |
| Side effects | Abdominal pain, diarrhea, nausea, constipation[1] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | By mouth |
| Typical dose | 800 to 2,400 mg TID[1] |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601248 |
| Legal | |
| License data | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | 0% |
| Excretion | Feces 100% |
| Chemical and physical data | |
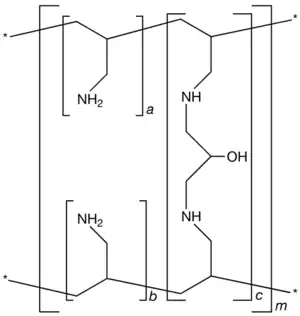
| Formula | [(C3H7N)a+b.(C9H17N2O)c]m where a+b:c = 9:1 |
| Molar mass | variable |
Sevelamer, sold under the brand name Renagel among others, is a medication used to treat hyperphosphatemia in chronic kidney disease.[1] In the United Kingdom it is a second line treatment.[2] It is taken by mouth with meals.[1]
Common side effects include abdominal pain, diarrhea, nausea, and constipation.[1] A skin rash may also occur.[2] It works by binding phosphate in the intestines.[1]
Sevelamer was approved for medical use in the United States in 1998.[1] In the United States it costs about 48 USD for 270 tablets of 800 mg as of 2021.[3] This amount in the United Kingdom costs the NHS about £75.[2]
Medical uses
Sevelamer is used in the management of hyperphosphatemia in adult patients with stage 4 and 5 chronic kidney disease (CKD) on hemodialysis. Its efficacy at lowering phosphate levels is similar to that of calcium acetate, but without the accompanying risk of hypercalcemia and arterial calcification.[4][5] In patients with CKD, it has also been shown to reduce triglycerides and LDL, and increase HDL.[6]
Dosage
Sevelamer is used at a dose of 800 mg to 2,400 mg three times per day.[1]
Contraindications
Sevelamer therapy is contraindicated in hypophosphatemia or bowel obstruction. In hypophosphatemia, sevelamer could exacerbate the condition by further lowering phosphate levels in the blood, which could be fatal.[7]
Side effects
.png.webp)
Common side effects associated with the use of sevelamer include: hypotension, hypertension, nausea and vomiting, dyspepsia, diarrhea, flatulence, and/or constipation.
Pharmacology
Sevelamer consists of polyallylamine that is crosslinked with epichlorohydrin.[8] The marketed form sevelamer hydrochloride is a partial hydrochloride salt being present as approximately 40% amine hydrochloride and 60% sevelamer base. The amine groups of sevelamer become partially protonated in the intestine and interact with phosphate ions through ionic and hydrogen bonding.
History
It was invented and developed by GelTex Pharmaceuticals. Sevelamer is marketed by Sanofi under the brand names Renagel (sevelamer hydrochloride) and Renvela (sevelamer carbonate).
Society and culture
Cost
This medication in the U.S. has a cost of $48 (USD) for 270 tablets (800 mg) as of 2021.[3]
.svg.png.webp) Sevelamer costs (US)
Sevelamer costs (US).svg.png.webp) Sevelamer prescriptions (US)
Sevelamer prescriptions (US)
Research
Sevelamer can significantly reduce serum uric acid.[9] This reduction has no known detrimental effect and several beneficial effects, including reducing hyperuricemia, uric acid nephrolithiasis, and gout.
Sevelamer is able to sequester advanced glycation end products (AGEs) in the gut, preventing their absorption into the blood. AGEs contribute to oxidative stress, which can damage cells (like beta cells, which produce insulin in the pancreas). As Vlassara and Uribarri explain in a 2014 review on AGEs, this may explain why sevelamer, but not calcium carbonate (a phosphate binder that does not sequester AGEs), has been shown to lower AGEs in the blood, as well as oxidative stress and inflammatory markers.[10]
References
- 1 2 3 4 5 6 7 8 9 "Sevelamer Monograph for Professionals". Drugs.com. Archived from the original on 30 November 2020. Retrieved 12 October 2021.
- 1 2 3 BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 1116. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - 1 2 "Sevelamer Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 19 January 2021. Retrieved 7 April 2021.
- ↑ Burke SK (September 2000). "Renagel: reducing serum phosphorus in haemodialysis patients". review. Hospital Medicine. 61 (9): 622–7. doi:10.12968/hosp.2000.61.9.1419. PMID 11048603.
- ↑ Habbous S, Przech S, Acedillo R, Sarma S, Garg AX, Martin J (January 2017). "The efficacy and safety of sevelamer and lanthanum versus calcium-containing and iron-based binders in treating hyperphosphatemia in patients with chronic kidney disease: a systematic review and meta-analysis". review. Nephrology, Dialysis, Transplantation. 32 (1): 111–125. doi:10.1093/ndt/gfw312. PMID 27651467.
- ↑ Patel L, Bernard LM, Elder GJ (February 2016). "Sevelamer Versus Calcium-Based Binders for Treatment of Hyperphosphatemia in CKD: A Meta-Analysis of Randomized Controlled Trials". review. Clinical Journal of the American Society of Nephrology. 11 (2): 232–44. doi:10.2215/CJN.06800615. PMC 4741042. PMID 26668024.
- ↑ Emmett M (September 2004). "A comparison of clinically useful phosphorus binders for patients with chronic kidney failure". review. Kidney International Supplements. 66 (90): S25–32. doi:10.1111/j.1523-1755.2004.09005.x. PMID 15296504.
- ↑ Ramsdell R (June 1999). "Renagel: a new and different phosphate binder". review. ANNA Journal. 26 (3): 346–7. PMID 10633608.
- ↑ Locatelli F, Del Vecchio L (May 2015). "Cardiovascular mortality in chronic kidney disease patients: potential mechanisms and possibilities of inhibition by resin-based phosphate binders". review. Expert Review of Cardiovascular Therapy. 13 (5): 489–99. doi:10.1586/14779072.2015.1029456. PMID 25804298. S2CID 32586527.
- ↑ Vlassara H, Uribarri J (January 2014). "Advanced glycation end products (AGE) and diabetes: cause, effect, or both?". review. Current Diabetes Reports. 14 (1): 453. doi:10.1007/s11892-013-0453-1. PMC 3903318. PMID 24292971.
External links
| Identifiers: |
|---|
- "Sevelamer". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2020-11-20. Retrieved 2020-12-16.
- "Sevelamer hydrochloride". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2021-08-29. Retrieved 2020-12-16.
- "Sevelamer carbonate". Drug Information Portal. U.S. National Library of Medicine. Archived from the original on 2020-10-22. Retrieved 2020-12-16.