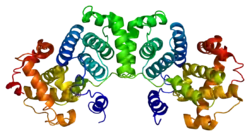
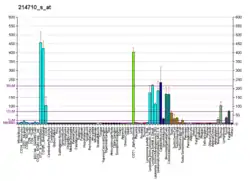
Cyclin B1
G2/mitotic-specific cyclin-B1 is a protein that in humans is encoded by the CCNB1 gene.[5]
Function
Cyclin B1 is a regulatory protein involved in mitosis. The gene product complexes with p34 (Cdk1) to form the maturation-promoting factor (MPF). Two alternative transcripts have been found, a constitutively expressed transcript and a cell cycle-regulated transcript that is expressed predominantly during G2/M phase of the cell cycle. The different transcripts result from the use of alternate transcription initiation sites.[6]
Cyclin B1 contributes to the switch-like all or none behavior of the cell in deciding to commit to mitosis. Its activation is well-regulated, and positive feedback loops ensure that once the cyclin B1-Cdk1 complex is activated, it is not deactivated. Cyclin B1-Cdk1 is involved in the early events of mitosis, such as chromosome condensation, nuclear envelope breakdown, and spindle pole assembly.
Once activated, cyclin B1-Cdk1 promotes several of the events of early mitosis. The active complex phosphorylates and activates 13S condensin,[7] which helps to condense chromosomes.
Another important function of the cyclin B1-Cdk1 complex is to break down the nuclear envelope. The nuclear envelope is a membranous structure containing large protein complexes supported by a network of nuclear lamins. Phosphorylation of the lamins by cyclin B1-Cdk1 causes them to dissociate,[8] compromising the structural integrity of the nuclear envelope so that it breaks down. The destruction of the nuclear envelope is important because it allows the mitotic spindle to access the chromosomes.
Regulation
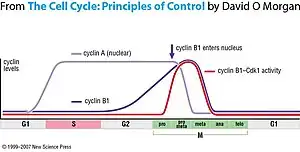
Like all cyclins, levels of cyclin B1 oscillate over the course of the cell cycle. Just prior to mitosis, a large amount of cyclin B1 is present in the cell, but it is inactive due to phosphorylation of Cdk1 by the Wee1 kinase. The complex is activated by dephosphorylation by the phosphatase Cdc25.[9] Cdc25 is always present in the cell but must be activated by phosphorylation. A possible trigger for activation is phosphorylation by cyclin A-Cdk, which functions before cyclin B1-Cdk in the cell cycle. Active Cdk1 is also capable of phosphorylating and activating Cdc25 and thus promote its own activation, resulting in a positive feedback loop. Once cyclin B1-Cdk1 is activated, it remains stably active for the rest of mitosis.
Another mechanism by which cyclin B1-Cdk1 activity is regulated is through subcellular localization. Before mitosis almost all cyclin B1 in the cell is located in the cytoplasm, but in late prophase it relocates to the nucleus. This is regulated by the phosphorylation of cyclin B1, in contrast to phosphorylation of Cdk1 regulating the activity of the complex. Phosphorylation of cyclin B1 causes it to be imported to the nucleus,[10] and phosphorylation also prevents export from the nucleus by blocking the nuclear export signal.[11] Cyclin B1 is phosphorylated by Polo kinase and Cdk1, again setting up a positive feedback loop that commits cyclin B1-Cdk1 to its fate.
At the end of mitosis, cyclin B1 is targeted for degradation by the APC through its APC localization sequence, permitting the cell to exit mitosis.
Cancer
One of the hallmarks of cancer is the lack of regulation in the cell cycle. The role of cyclin B1 is to transition the cell from G2 to M phase but becomes unregulated in cancer cells where overexpression of cyclin B1 can lead to uncontrolled cell growth by binding to its partner Cdks. Binding of Cdks can lead to phosphorylation of other substrates at inappropriate time and unregulated proliferation.[19] This is a consequence of p53, tumor suppressor protein, being inactivated. Wild-type p53 have been shown to suppress cyclin B1 expression.[20][21]
Previous work has shown that high cyclin B1 expression levels are found in variety of cancers such as breast, cervical, gastric, colorectal, head and neck squamous cell, non-small-cell lung cancer, colon, prostate, oral and esophageal.[19][22][23][24][25] High expression levels are usually seen before the tumor cells become immortalized and aneuploid which can contribute to the chromosomal instability and the aggressive nature of certain cancers.[26] These high levels of cyclin B1 can also be associated to the extent of tumor invasion and aggressiveness therefore concentration of cyclin B1 can be used to determine the prognosis of cancer patients.[22][27] For example, an increase in expression of cyclin B1/cdc2 is significantly higher in breast tumor tissue and shown to increase lymph node metastasis in breast cancer.[22][28]
Cyclin B1 can reside in the nucleus or the cytoplasm which can have an effect on the malignant potential of cyclin B1 when overexpressed in each location. Nuclear-dominant expression of cyclin B1 leads to poorer prognosis due to its weak activity compared to cytoplasmic cyclin B1.[26] This trend has been observed in esophageal cancer, head and neck squamous cell cancer and breast cancer.[19][29]
Down regulation and tumor suppression
While the exact mechanism that explains how cyclin B1 becomes overexpressed is not very well understood, previous work has shown that down regulation of cyclin B1 can lead to tumor regression. A possible treatment option for tumor suppression is to deliver gene or protein to target the degradation of cyclin B1. Previous work done has shown that cyclin B1 is essential for tumor cell survival and proliferation and that a decrease in expression levels only leads to tumor-specific and not normal cell death.[30] Reduction of cyclin B1 can stop cells in the G2 phase of the cell cycle and triggers cell death by preventing the chromosomes from condensing and aligning. The specific downregulation of cyclin B1, however, did not influence other molecules that facilitated the transition from the G2 to M phase such as Cdk1, Cdc25c, Plk1 and cyclin A. Therefore the delivery of a therapeutic gene to correct these mutations is a viable treatment option for tumor suppression.[19]
Tumor antigen
In early stages of cancer when the cyclin B1 concentration is high, it is recognized by the immune system, leading to the production of antibodies and T cells. It would then be possible to take advantage of this to monitor the immune response for early cancer detection.[31] An ELISA (Enzyme-linked immunosorbent assay) can be performed to the measure the antibodies recognizing cyclin B1.
Breast cancer
Cyclin B1 expression levels can be used as a tool to determine prognosis of patients with breast cancers. The intracellular concentration can have important implications for cancer prognosis. High levels of nuclear cyclin B1 is associated with high tumor grade, larger tumor size and higher metastasis probability, therefore a high level of cyclin B1 is a predictor of poor prognosis.[26]
Lung cancer
Studies in non-small cell lung cancer demonstrated that high levels of cyclin B1 are associated with poorer prognosis. The study also found that this correlation between expression levels was only found in patients with squamous cell carcinoma. This finding indicates the possibility of using cyclin B1 expression as a prognostic marker for patients with early stage non-small cell lung cancer.[32]
See also
References
- GRCh38: Ensembl release 89: ENSG00000134057 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000048574 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Sartor H, Ehlert F, Grzeschik KH, Müller R, Adolph S (Aug 1992). "Assignment of two human cell cycle genes, CDC25C and CCNB1, to 5q31 and 5q12, respectively". Genomics. 13 (3): 911–2. doi:10.1016/0888-7543(92)90190-4. PMID 1386342.
- "Entrez Gene: CCNB1 cyclin B1".
- Kimura K, Hirano M, Kobayashi R, Hirano T (October 1998). "Phosphorylation and activation of 13S condensin by Cdc2 in vitro". Science. 282 (5388): 487–90. Bibcode:1998Sci...282..487K. doi:10.1126/science.282.5388.487. PMID 9774278.
- Heald R, McKeon F (May 1990). "Mutations of phosphorylation sites in lamin A that prevent nuclear lamina disassembly in mitosis". Cell. 61 (4): 579–89. doi:10.1016/0092-8674(90)90470-Y. PMID 2344612. S2CID 45118388.
- Berry LD, Gould KL (1996). "Regulation of Cdc2 activity by phosphorylation at T14/Y15". Prog Cell Cycle Res. 2: 99–105. doi:10.1007/978-1-4615-5873-6_10. ISBN 978-1-4613-7693-4. PMID 9552387.
- Hagting, Anja; Jackman, Mark; Simpson, Karen; Pines, Jonathon (1999). "Translocation of cyclin B1 to the nucleus at prophase requires a phosphorylation-dependent nuclear import signal". Current Biology. 9 (13): 680–689. doi:10.1016/S0960-9822(99)80308-X. PMID 10395539. S2CID 10697376.
- Yang J, Song H, Walsh S, Bardes ES, Kornbluth S (February 2001). "Combinatorial control of cyclin B1 nuclear trafficking through phosphorylation at multiple sites". J. Biol. Chem. 276 (5): 3604–9. doi:10.1074/jbc.M008151200. PMID 11060306.
- Shanahan F, Seghezzi W, Parry D, Mahony D, Lees E (February 1999). "Cyclin E associates with BAF155 and BRG1, components of the mammalian SWI-SNF complex, and alters the ability of BRG1 to induce growth arrest". Mol. Cell. Biol. 19 (2): 1460–9. doi:10.1128/mcb.19.2.1460. PMC 116074. PMID 9891079.
- Yang Q, Manicone A, Coursen JD, Linke SP, Nagashima M, Forgues M, Wang XW (November 2000). "Identification of a functional domain in a GADD45-mediated G2/M checkpoint". J. Biol. Chem. 275 (47): 36892–8. doi:10.1074/jbc.M005319200. PMID 10973963.
- Pines, Jonathon; Hunter, Tony (1989). "Isolation of a human cyclin cDNA: Evidence for cyclin mRNA and protein regulation in the cell cycle and for interaction with p34cdc2". Cell. 58 (5): 833–846. doi:10.1016/0092-8674(89)90936-7. PMID 2570636. S2CID 20336733.
- Kong M, Barnes EA, Ollendorff V, Donoghue DJ (March 2000). "Cyclin F regulates the nuclear localization of cyclin B1 through a cyclin-cyclin interaction". EMBO J. 19 (6): 1378–88. doi:10.1093/emboj/19.6.1378. PMC 305678. PMID 10716937.
- Zhan Q, Antinore MJ, Wang XW, Carrier F, Smith ML, Harris CC, Fornace AJ (May 1999). "Association with Cdc2 and inhibition of Cdc2/Cyclin B1 kinase activity by the p53-regulated protein Gadd45". Oncogene. 18 (18): 2892–900. doi:10.1038/sj.onc.1202667. PMID 10362260.
- Vairapandi M, Balliet AG, Hoffman B, Liebermann DA (September 2002). "GADD45b and GADD45g are cdc2/cyclinB1 kinase inhibitors with a role in S and G2/M cell cycle checkpoints induced by genotoxic stress". J. Cell. Physiol. 192 (3): 327–38. doi:10.1002/jcp.10140. PMID 12124778. S2CID 19138273.
- Rossé C, L'Hoste S, Offner N, Picard A, Camonis J (August 2003). "RLIP, an effector of the Ral GTPases, is a platform for Cdk1 to phosphorylate epsin during the switch off of endocytosis in mitosis". J. Biol. Chem. 278 (33): 30597–604. doi:10.1074/jbc.M302191200. PMID 12775724.
- Yuan J, Krämer A, Matthess Y, Yan R, Spänkuch B, Gätje R, Knecht R, Kaufmann M, Strebhardt K (March 2006). "Stable gene silencing of cyclin B1 in tumor cells increases susceptibility to taxol and leads to growth arrest in vivo". Oncogene. 25 (12): 1753–62. doi:10.1038/sj.onc.1209202. PMID 16278675.
- Yu M, Zhan Q, Finn OJ (May 2002). "Immune recognition of cyclin B1 as a tumor antigen is a result of its overexpression in human tumors that is caused by non-functional p53". Mol. Immunol. 38 (12–13): 981–7. doi:10.1016/S0161-5890(02)00026-3. PMID 12009577.
- Innocente SA, Abrahamson JL, Cogswell JP, Lee JM (March 1999). "p53 regulates a G2 checkpoint through cyclin B1". Proc. Natl. Acad. Sci. U.S.A. 96 (5): 2147–52. Bibcode:1999PNAS...96.2147I. doi:10.1073/pnas.96.5.2147. PMC 26751. PMID 10051609.
- Kawamoto H, Koizumi H, Uchikoshi T (January 1997). "Expression of the G2-M checkpoint regulators cyclin B1 and cdc2 in nonmalignant and malignant human breast lesions: immunocytochemical and quantitative image analyses". Am. J. Pathol. 150 (1): 15–23. PMC 1858517. PMID 9006317.
- Wang A, Yoshimi N, Ino N, Tanaka T, Mori H (1997). "Overexpression of cyclin B1 in human colorectal cancers". J. Cancer Res. Clin. Oncol. 123 (2): 124–7. doi:10.1007/BF01269891. PMID 9030252. S2CID 10436036.
- Mashal RD, Lester S, Corless C, Richie JP, Chandra R, Propert KJ, Dutta A (September 1996). "Expression of cell cycle-regulated proteins in prostate cancer". Cancer Res. 56 (18): 4159–63. PMID 8797586.
- Kushner J, Bradley G, Young B, Jordan RC (February 1999). "Aberrant expression of cyclin A and cyclin B1 proteins in oral carcinoma". Journal of Oral Pathology & Medicine. 28 (2): 77–81. doi:10.1111/j.1600-0714.1999.tb02000.x. PMID 9950254.
- Suzuki T, Urano T, Miki Y, Moriya T, Akahira J, Ishida T, Horie K, Inoue S, Sasano H (May 2007). "Nuclear cyclin B1 in human breast carcinoma as a potent prognostic factor". Cancer Sci. 98 (5): 644–51. doi:10.1111/j.1349-7006.2007.00444.x. PMID 17359284. S2CID 24203952.
- Dutta A, Chandra R, Leiter LM, Lester S (June 1995). "Cyclins as markers of tumor proliferation: immunocytochemical studies in breast cancer". Proc. Natl. Acad. Sci. U.S.A. 92 (12): 5386–90. Bibcode:1995PNAS...92.5386D. doi:10.1073/pnas.92.12.5386. PMC 41699. PMID 7539916.
- Winters ZE, Hunt NC, Bradburn MJ, Royds JA, Turley H, Harris AL, Norbury CJ (December 2001). "Subcellular localisation of cyclin B, Cdc2 and p21(WAF1/CIP1) in breast cancer. association with prognosis". Eur. J. Cancer. 37 (18): 2405–12. doi:10.1016/S0959-8049(01)00327-6. PMID 11720835.
- Nozoe T, Korenaga D, Kabashima A, Ohga T, Saeki H, Sugimachi K (March 2002). "Significance of cyclin B1 expression as an independent prognostic indicator of patients with squamous cell carcinoma of the esophagus". Clin. Cancer Res. 8 (3): 817–22. PMID 11895914.
- Yuan J, Yan R, Krämer A, Eckerdt F, Roller M, Kaufmann M, Strebhardt K (July 2004). "Cyclin B1 depletion inhibits proliferation and induces apoptosis in human tumor cells". Oncogene. 23 (34): 5843–52. doi:10.1038/sj.onc.1207757. PMID 15208674.
- Egloff AM, Weissfeld J, Land SR, Finn OJ (December 2005). "Evaluation of anticyclin B1 serum antibody as a diagnostic and prognostic biomarker for lung cancer". Ann. N. Y. Acad. Sci. 1062 (1): 29–40. Bibcode:2005NYASA1062...29E. doi:10.1196/annals.1358.005. PMID 16461786. S2CID 28812067.
- Soria JC, Jang SJ, Khuri FR, Hassan K, Liu D, Hong WK, Mao L (August 2000). "Overexpression of cyclin B1 in early-stage non-small cell lung cancer and its clinical implication". Cancer Res. 60 (15): 4000–4. PMID 10945597.
Further reading
- Kino T, Chrousos GP (2004). "Human immunodeficiency virus type-1 accessory protein Vpr: a causative agent of the AIDS-related insulin resistance/lipodystrophy syndrome?". Ann. N. Y. Acad. Sci. 1024 (1): 153–67. Bibcode:2004NYASA1024..153K. doi:10.1196/annals.1321.013. PMID 15265780. S2CID 23655886.
- Bailly E, Pines J, Hunter T, Bornens M (1992). "Cytoplasmic accumulation of cyclin B1 in human cells: association with a detergent-resistant compartment and with the centrosome". J. Cell Sci. 101 (3): 529–45. doi:10.1242/jcs.101.3.529. PMID 1387877.
- Galaktionov K, Beach D (1991). "Specific activation of cdc25 tyrosine phosphatases by B-type cyclins: evidence for multiple roles of mitotic cyclins". Cell. 67 (6): 1181–94. doi:10.1016/0092-8674(91)90294-9. PMID 1836978. S2CID 9659637.
- Pines J, Hunter T (1989). "Isolation of a human cyclin cDNA: evidence for cyclin mRNA and protein regulation in the cell cycle and for interaction with p34cdc2". Cell. 58 (5): 833–46. doi:10.1016/0092-8674(89)90936-7. PMID 2570636. S2CID 20336733.
- He J, Choe S, Walker R, Di Marzio P, Morgan DO, Landau NR (1995). "Human immunodeficiency virus type 1 viral protein R (Vpr) arrests cells in the G2 phase of the cell cycle by inhibiting p34cdc2 activity". J. Virol. 69 (11): 6705–11. doi:10.1128/JVI.69.11.6705-6711.1995. PMC 189580. PMID 7474080.
- Re F, Braaten D, Franke EK, Luban J (1995). "Human immunodeficiency virus type 1 Vpr arrests the cell cycle in G2 by inhibiting the activation of p34cdc2-cyclin B". J. Virol. 69 (11): 6859–64. doi:10.1128/JVI.69.11.6859-6864.1995. PMC 189600. PMID 7474100.
- Di Marzio P, Choe S, Ebright M, Knoblauch R, Landau NR (1995). "Mutational analysis of cell cycle arrest, nuclear localization and virion packaging of human immunodeficiency virus type 1 Vpr". J. Virol. 69 (12): 7909–16. doi:10.1128/JVI.69.12.7909-7916.1995. PMC 189735. PMID 7494303.
- Jowett JB, Planelles V, Poon B, Shah NP, Chen ML, Chen IS (1995). "The human immunodeficiency virus type 1 vpr gene arrests infected T cells in the G2 + M phase of the cell cycle". J. Virol. 69 (10): 6304–13. doi:10.1128/JVI.69.10.6304-6313.1995. PMC 189529. PMID 7666531.
- Jackman M, Firth M, Pines J (1995). "Human cyclins B1 and B2 are localized to strikingly different structures: B1 to microtubules, B2 primarily to the Golgi apparatus". EMBO J. 14 (8): 1646–54. doi:10.1002/j.1460-2075.1995.tb07153.x. PMC 398257. PMID 7737117.
- Piaggio G, Farina A, Perrotti D, Manni I, Fuschi P, Sacchi A, Gaetano C (1995). "Structure and growth-dependent regulation of the human cyclin B1 promoter". Exp. Cell Res. 216 (2): 396–402. doi:10.1006/excr.1995.1050. PMID 7843284.
- Ookata K, Hisanaga S, Bulinski JC, Murofushi H, Aizawa H, Itoh TJ, Hotani H, Okumura E, Tachibana K, Kishimoto T (1995). "Cyclin B interaction with microtubule-associated protein 4 (MAP4) targets p34cdc2 kinase to microtubules and is a potential regulator of M-phase microtubule dynamics". J. Cell Biol. 128 (5): 849–62. doi:10.1083/jcb.128.5.849. PMC 2120387. PMID 7876309.
- Pines J, Hunter T (1994). "The differential localization of human cyclins A and B is due to a cytoplasmic retention signal in cyclin B". EMBO J. 13 (16): 3772–81. doi:10.1002/j.1460-2075.1994.tb06688.x. PMC 395290. PMID 8070405.
- Xiong Y, Zhang H, Beach D (1993). "Subunit rearrangement of the cyclin-dependent kinases is associated with cellular transformation" (PDF). Genes Dev. 7 (8): 1572–83. doi:10.1101/gad.7.8.1572. PMID 8101826.
- Zheng XF, Ruderman JV (1993). "Functional analysis of the P box, a domain in cyclin B required for the activation of Cdc25". Cell. 75 (1): 155–64. doi:10.1016/S0092-8674(05)80092-3. PMID 8402895. S2CID 45351149.
- O'Connor PM, Ferris DK, Pagano M, Draetta G, Pines J, Hunter T, Longo DL, Kohn KW (1993). "G2 delay induced by nitrogen mustard in human cells affects cyclin A/cdk2 and cyclin B1/cdc2-kinase complexes differently". J. Biol. Chem. 268 (11): 8298–308. doi:10.1016/S0021-9258(18)53096-9. PMID 8463339.
- Sebastian B, Kakizuka A, Hunter T (1993). "Cdc25M2 activation of cyclin-dependent kinases by dephosphorylation of threonine-14 and tyrosine-15". Proc. Natl. Acad. Sci. U.S.A. 90 (8): 3521–4. Bibcode:1993PNAS...90.3521S. doi:10.1073/pnas.90.8.3521. PMC 46332. PMID 8475101.
- Kolesnitchenko V, Wahl LM, Tian H, Sunila I, Tani Y, Hartmann DP, Cossman J, Raffeld M, Orenstein J, Samelson LE, Cohen DI (1995). "Human immunodeficiency virus 1 envelope-initiated G2-phase programmed cell death". Proc. Natl. Acad. Sci. U.S.A. 92 (25): 11889–93. Bibcode:1995PNAS...9211889K. doi:10.1073/pnas.92.25.11889. PMC 40508. PMID 8524869.
- Poon RY, Jiang W, Toyoshima H, Hunter T (1996). "Cyclin-dependent kinases are inactivated by a combination of p21 and Thr-14/Tyr-15 phosphorylation after UV-induced DNA damage". J. Biol. Chem. 271 (22): 13283–91. doi:10.1074/jbc.271.22.13283. PMID 8662825.
- Chen J, Saha P, Kornbluth S, Dynlacht BD, Dutta A (1996). "Cyclin-binding motifs are essential for the function of p21CIP1". Mol. Cell. Biol. 16 (9): 4673–82. doi:10.1128/MCB.16.9.4673. PMC 231467. PMID 8756624.