Chorismate lyase
The enzyme chorismate lyase (EC 4.1.3.40) catalyzes the first step in ubiquinone biosynthesis, the removal of pyruvate from chorismate, to yield 4-hydroxybenzoate in Escherichia coli and other Gram-negative bacteria.[1] It belongs to the family of lyases, specifically the oxo-acid-lyases, which cleave carbon-carbon bonds. The systematic name of this enzyme class is chorismate pyruvate-lyase (4-hydroxybenzoate-forming). Other names in common use include CL, CPL, and UbiC.
| Chorismate lyase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC no. | 4.1.3.40 | ||||||||
| CAS no. | 157482-18-3 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
This enzyme catalyses the chemical reaction:[2]
- chorismate 4-hydroxybenzoate + pyruvate
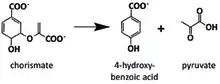 The chorismate pyruvate lyase (CPL) catalyzed reaction.
The chorismate pyruvate lyase (CPL) catalyzed reaction.
Activity
| Chorismate lyase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 chorismate lyase with product, 1.0 a resolution | |||||||||
| Identifiers | |||||||||
| Symbol | Chor_lyase | ||||||||
| Pfam | PF04345 | ||||||||
| Pfam clan | CL0122 | ||||||||
| InterPro | IPR007440 | ||||||||
| SCOP2 | 1jd3 / SCOPe / SUPFAM | ||||||||
| |||||||||
Catalytic activity
- This enzyme has an optimum pH at 7.5
Enzymatic activity
Inhibited by:
- Vanillate
- 4-hydroxybenzaldehyde
- 3-carboxylmethylaminmethyl-4-hydroxybenzoic acid
- 4HB - ubiC is inhibited by the product of the reaction, which scientists believe serves as a control mechanism for the pathway
Pathway
The pathway used is called the ubiquinone biosynthesis pathway, it catalyzes the first step in the biosynthesis of ubiquinone in E. coli. Ubiquinone is a lipid-soluble electron-transporting coenzyme. They are essential electron carriers in prokaryotes and are essential in aerobic organisms to achieve ATP synthesis.[4]
Nomenclature
There are several different names for chorismate lyase. It is also called chorismate pyruvate lyase (4-hydroxybenzoate-forming) and it is also abbreviated several different ways: CPL, CL, and ubiC. It is sometimes referred to as ubiC, because that is the gene name. This enzyme belongs to the class lyases; more specifically the ox-acid-lyase or the carbon-carbon-lyases.[5]
Taxonomic lineage:
- bacteria → proteobacteria → gammaproteobacteria → enterobacteriales → enterobacteriaceae → escherichia → Escherichia coli
Structure
This enzyme is a monomer. Its secondary structure contains helixes, turns, and beta-strands. It has a mass of 18,777 daltons and its sequence is 165 amino acids long.[5]
Binding sites
- position: 35(M)
- position: 77(R)
- position: 115(L)
Mutagenesis
- position: 91G → A; increases product inhibition by 40%. No effect on substrate affinity.
- position: 156E → K; loss of activity
References
- Nichols BP, Green JM (August 1992). "Cloning and sequencing of Escherichia coli ubiC and purification of chorismate lyase". J. Bacteriol. 174 (16): 5309–16. doi:10.1128/jb.174.16.5309-5316.1992. PMC 206367. PMID 1644758.
- "EC 4.1.3.40".
- Siebert M, Severin K, Heide L (April 1994). "Formation of 4-hydroxybenzoate in Escherichia coli: characterization of the ubiC gene and its encoded enzyme chorismate pyruvate-lyase". Microbiology. 140 (4): 897–904. doi:10.1099/00221287-140-4-897. PMID 8012607.
- "KEGG PATHWAY: Ubiquinone and other terpenoid-quinone biosynthesis - Reference pathway". www.genome.jp. Retrieved 2021-04-09.
- "UniprotID: P26602".
Further reading
- Nichols BP, Green JM (1992). "Cloning and sequencing of Escherichia coli ubiC and purification of chorismate lyase". J. Bacteriol. 174 (16): 5309–16. doi:10.1128/jb.174.16.5309-5316.1992. PMC 206367. PMID 1644758.
- Siebert M, Severin K, Heide L (1994). "Formation of 4-hydroxybenzoate in Escherichia coli: characterization of the ubiC gene and its encoded enzyme chorismate pyruvate-lyase". Microbiology. 140 (4): 897–904. doi:10.1099/00221287-140-4-897. PMID 8012607.
- Meganathan R (2001). "Ubiquinone biosynthesis in microorganisms". FEMS Microbiol. Lett. 203 (2): 131–9. doi:10.1111/j.1574-6968.2001.tb10831.x. PMID 11583838.