Coccolith
Coccoliths are individual plates or scales of calcium carbonate formed by coccolithophores (single-celled phytoplankton such as Emiliania huxleyi) and cover the cell surface arranged in the form of a spherical shell, called a coccosphere.

Overview
Coccolithophores are spherical cells about 5–100 micrometres across, enclosed by calcareous plates called coccoliths, which are about 2–25 micrometres across.[1] Coccolithophores are an important group of about 200 marine phytoplankton species [2] which cover themselves with a calcium carbonate shell called a "coccosphere". They are ecologically and biogeochemically important but the reason why they calcify remains elusive. One key function may be that the coccosphere offers protection against microzooplankton predation, which is one of the main causes of phytoplankton death in the ocean.[3]
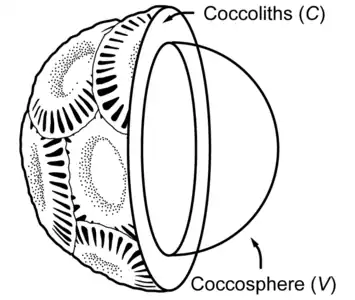 Partial cross section of a coccolithophore with coccolith layer [4]
Partial cross section of a coccolithophore with coccolith layer [4]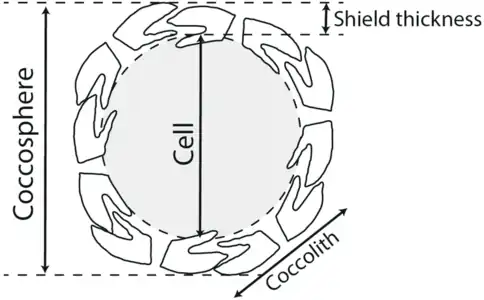 Coccolithophore cell surrounded by its shield of coccoliths. The coccolith-bearing cell is called the coccosphere.[5][6]
Coccolithophore cell surrounded by its shield of coccoliths. The coccolith-bearing cell is called the coccosphere.[5][6]
Coccolithophores have been an integral part of marine plankton communities since the Jurassic.[7][8] Today, coccolithophores contribute ~1–10% to primary production in the surface ocean [9] and ~50% to pelagic CaCO3 sediments.[10] Their calcareous shell increases the sinking velocity of photosynthetically fixed CO2 into the deep ocean by ballasting organic matter.[11][12] At the same time, the biogenic precipitation of calcium carbonate during coccolith formation reduces the total alkalinity of seawater and releases CO2.[13][14] Thus, coccolithophores play an important role in the marine carbon cycle by influencing the efficiency of the biological carbon pump and the oceanic uptake of atmospheric CO2.[3]
As of 2021, it is not known why coccolithophores calcify and how their ability to produce coccoliths is associated with their ecological success.[15][16][17][18][19] The most plausible benefit of having a coccosphere seems to be a protection against predators or viruses.[20][18] Viral infection is an important cause of phytoplankton death in the oceans,[21] and it has recently been shown that calcification can influence the interaction between a coccolithophore and its virus.[22][23] The major predators of marine phytoplankton are microzooplankton like ciliates and dinoflagellates. These are estimated to consume about two-thirds of the primary production in the ocean [24] and microzooplankton can exert a strong grazing pressure on coccolithophore populations.[25] Although calcification does not prevent predation, it has been argued that the coccosphere reduces the grazing efficiency by making it more difficult for the predator to utilise the organic content of coccolithophores.[26] Heterotrophic protists are able to selectively choose prey on the basis of its size or shape and through chemical signals [27][28] and may thus favor other prey that is available and not protected by coccoliths.[3]
Formation and composition
| Part of a series related to |
| Biomineralization |
|---|
 |
|
Coccoliths are formed within the cell in vesicles derived from the golgi body. When the coccolith is complete these vesicles fuse with the cell wall and the coccolith is exocytosed and incorporated in the coccosphere. The coccoliths are either dispersed following death and breakup of the coccosphere, or are shed continually by some species. They sink through the water column to form an important part of the deep-sea sediments (depending on the water depth). Thomas Huxley was the first person to observe these forms in modern marine sediments and he gave them the name 'coccoliths' in a report published in 1858.[29][30] Coccoliths are composed of calcium carbonate as the mineral calcite and are the main constituent of chalk deposits such as the white cliffs of Dover (deposited in Cretaceous times), in which they were first described by Henry Clifton Sorby in 1861.[31]
 Collapsed coccosphere of Pleurochrysis carterae
Collapsed coccosphere of Pleurochrysis carterae
Types
There are two main types of coccoliths, heterococcoliths and holococcoliths. Heterococcoliths are formed of a radial array of elaborately shaped crystal units. Holococcoliths are formed of minute (~0.1 micrometre) calcite rhombohedra, arranged in continuous arrays. The two coccolith types were originally thought to be produced by different families of coccolithophores. Now, however, it is known through a mix of observations on field samples and laboratory cultures, that the two coccolith types are produced by the same species but at different life cycle phases. Heterococcoliths are produced in the diploid life-cycle phase and holococcoliths in the haploid phase. Both in field samples and laboratory cultures, there is the possibility of observing a cell covered by a combination of heterococcoliths and holococcoliths. This indicates the transition from the diploid to the haploid phase of the species. Such combination of coccoliths has been observed in field samples, with many of them coming from the Mediterranean.[32][33]
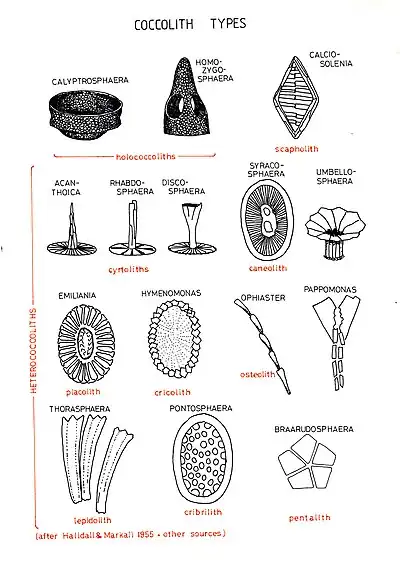
Shape
Coccoliths are also classified depending on shape. Common shapes include:[34][35]
- Calyptrolith – basket-shaped with openings near the base
- Caneolith – disc- or bowl-shaped
- Ceratolith – horseshoe or wishbone shaped
- Cribrilith – disc-shaped, with numerous perforations in the central area
- Cyrtolith – convex disc shaped, may with a projecting central process
- Discolith – ellipsoidal with a raised rim, in some cases the high rim forms a vase or cup-like structure
- Helicolith – a placolith with a spiral margin
- Lopadolith – basket or cup-shaped with a high rim, opening distally
- Pentalith – pentagonal shape composed of five four-sided crystals
- Placolith – rim composed of two plates stacked on top of one another
- Prismatolith – polygonal, may have perforations
- Rhabdolith – a single plate with a club-shaped central process
- Scapholith – rhombohedral, with parallel lines in center

.png.webp)

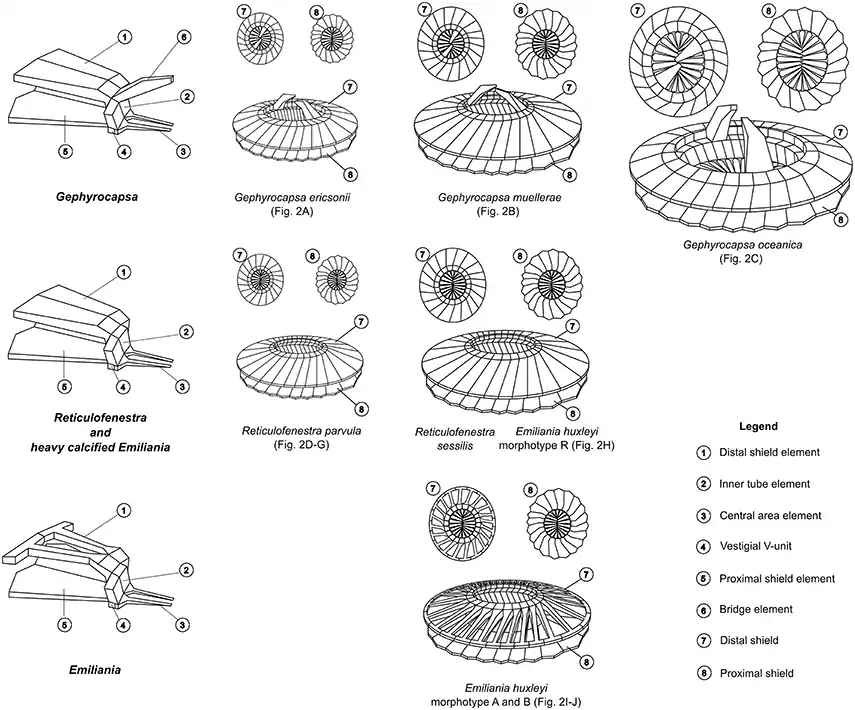 Coccolith structures of representative Noelaerhabdaceae.[36]
Coccolith structures of representative Noelaerhabdaceae.[36]
Each morphospecies is associated with a SEM image in the next diagram
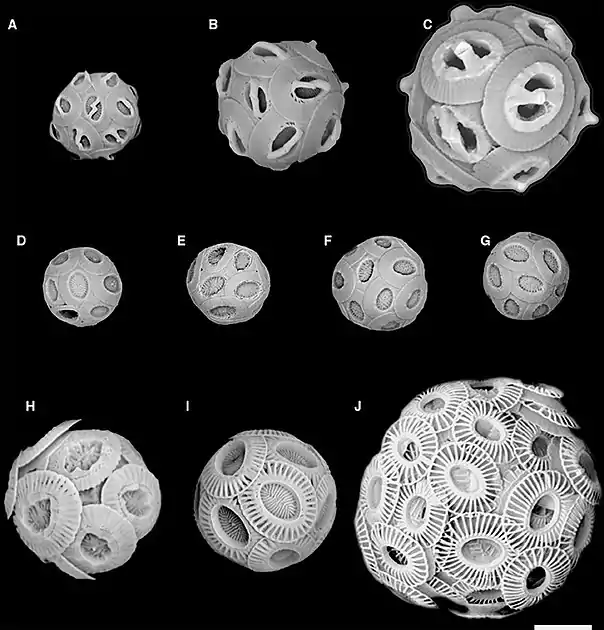 SEM images correspond to coccolith drawings in the previous diagram
SEM images correspond to coccolith drawings in the previous diagram
(A) Gephyrocapsa ericsonii RCC4032 (B) Gephyrocapsa muellerae (C) Gephyrocapsa oceanica (D) Reticulofenestra parvular RCC4033; (E) Reticulofenestra parvular RCC4034; (F) Reticulofenestra parvular RCC4035; (G) Reticulofenestra parvular RCC4036; (H) Emiliania huxleyi morphotype R; (I) Emiliania huxleyi morphotype A; (J) Emiliania huxleyi morphotype B.
Function
Although coccoliths are remarkably elaborate structures whose formation is a complex product of cellular processes, their function is unclear. Hypotheses include defence against grazing by zooplankton or infection by bacteria or viruses; maintenance of buoyancy; release of carbon dioxide for photosynthesis; to filter out harmful UV light; or in deep-dwelling species, to concentrate light for photosynthesis.
Fossil record
Because coccoliths are formed of low-Mg calcite, the most stable form of calcium carbonate, they are readily fossilised. They are found in sediments together with similar microfossils of uncertain affinities (nanoliths) from the Upper Triassic to recent. They are widely used as biostratigraphic markers and as paleoclimatic proxies. Coccoliths and related fossils are referred to as calcareous nanofossils or calcareous nannoplankton (nanoplankton).
References
- Moheimani, N.R.; Webb, J.P.; Borowitzka, M.A. (2012), "Bioremediation and other potential applications of coccolithophorid algae: A review. . Bioremediation and other potential applications of coccolithophorid algae: A review", Algal Research, 1 (2): 120–133, doi:10.1016/j.algal.2012.06.002
- Young, J. R.; Geisen, M.; Probert, I. (2005). "A review of selected aspects of coccolithophore biology with implications for paleobiodiversity estimation" (PDF). Micropaleontology. 51 (4): 267–288. doi:10.2113/gsmicropal.51.4.267.
- Haunost, Mathias; Riebesell, Ulf; D'Amore, Francesco; Kelting, Ole; Bach, Lennart T. (30 June 2021). "Influence of the Calcium Carbonate Shell of Coccolithophores on Ingestion and Growth of a Dinoflagellate Predator". Frontiers in Marine Science. Frontiers Media SA. 8. doi:10.3389/fmars.2021.664269. ISSN 2296-7745.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License. - Irie, Takahiro; Bessho, Kazuhiro; Findlay, Helen S.; Calosi, Piero (2010-10-15). "Increasing Costs Due to Ocean Acidification Drives Phytoplankton to Be More Heavily Calcified: Optimal Growth Strategy of Coccolithophores". PLoS ONE. Public Library of Science (PLoS). 5 (10): e13436. doi:10.1371/journal.pone.0013436. ISSN 1932-6203.
 Modified material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Modified material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License. - Aloisi, G. (6 August 2015). "Covariation of metabolic rates and cell size in coccolithophores". Biogeosciences. Copernicus GmbH. 12 (15): 4665–4692. Bibcode:2015BGeo...12.4665A. doi:10.5194/bg-12-4665-2015. ISSN 1726-4189. S2CID 6227548.
- Henderiks, Jorijntje (2008). "Coccolithophore size rules — Reconstructing ancient cell geometry and cellular calcite quota from fossil coccoliths". Marine Micropaleontology. Elsevier BV. 67 (1–2): 143–154. Bibcode:2008MarMP..67..143H. doi:10.1016/j.marmicro.2008.01.005. ISSN 0377-8398.
- Bown, Paul R.; Lees, Jackie A.; Young, Jeremy R. (2004). "Calcareous nannoplankton evolution and diversity through time". Coccolithophores. pp. 481–508. doi:10.1007/978-3-662-06278-4_18. ISBN 978-3-642-06016-8.
- Hay, William W. (2004). "Carbonate fluxes and calcareous nannoplankton". Coccolithophores. pp. 509–528. doi:10.1007/978-3-662-06278-4_19. ISBN 978-3-642-06016-8.
- Poulton, Alex J.; Adey, Tim R.; Balch, William M.; Holligan, Patrick M. (2007). "Relating coccolithophore calcification rates to phytoplankton community dynamics: Regional differences and implications for carbon export". Deep Sea Research Part II: Topical Studies in Oceanography. 54 (5–7): 538–557. Bibcode:2007DSRII..54..538P. doi:10.1016/j.dsr2.2006.12.003.
- Broecker, Wallace; Clark, Elizabeth (2009). "Ratio of coccolith CaCO3to foraminifera CaCO3in late Holocene deep sea sediments". Paleoceanography. 24 (3). Bibcode:2009PalOc..24.3205B. doi:10.1029/2009PA001731.
- Klaas, Christine; Archer, David E. (2002). "Association of sinking organic matter with various types of mineral ballast in the deep sea: Implications for the rain ratio". Global Biogeochemical Cycles. 16 (4): 1116. Bibcode:2002GBioC..16.1116K. doi:10.1029/2001GB001765. S2CID 34159028.
- Honjo, Susumu; Manganini, Steven J.; Krishfield, Richard A.; Francois, Roger (2008). "Particulate organic carbon fluxes to the ocean interior and factors controlling the biological pump: A synthesis of global sediment trap programs since 1983". Progress in Oceanography. 76 (3): 217–285. Bibcode:2008PrOce..76..217H. doi:10.1016/j.pocean.2007.11.003.
- Frankignoulle, Michel; Canon, Christine; Gattuso, Jean-Pierre (1994). "Marine calcification as a source of carbon dioxide: Positive feedback of increasing atmospheric CO2". Limnology and Oceanography. 39 (2): 458–462. Bibcode:1994LimOc..39..458F. doi:10.4319/lo.1994.39.2.0458. hdl:2268/246251.
- Rost, Björn; Riebesell, Ulf (2004). "Coccolithophores and the biological pump: Responses to environmental changes". Coccolithophores. pp. 99–125. doi:10.1007/978-3-662-06278-4_5. ISBN 978-3-642-06016-8.
- Young, J. R. (1987). Possible Functional Interpretations of Coccolith Morphology. New York: Springer-Verlag, 305–313.
- Young, J. R. (1994). "Functions of coccoliths," in Coccolithophores, eds A. Winter and W. G. Siesser (Cambridge: Cambridge University Press), 63–82.
- Raven, JA; Crawfurd, K. (2012). "Environmental controls on coccolithophore calcification". Marine Ecology Progress Series. 470: 137–166. Bibcode:2012MEPS..470..137R. doi:10.3354/meps09993.
- Monteiro, Fanny M.; Bach, Lennart T.; Brownlee, Colin; Bown, Paul; Rickaby, Rosalind E. M.; Poulton, Alex J.; Tyrrell, Toby; Beaufort, Luc; Dutkiewicz, Stephanie; Gibbs, Samantha; Gutowska, Magdalena A.; Lee, Renee; Riebesell, Ulf; Young, Jeremy; Ridgwell, Andy (2016). "Why marine phytoplankton calcify". Science Advances. 2 (7): e1501822. Bibcode:2016SciA....2E1822M. doi:10.1126/sciadv.1501822. PMC 4956192. PMID 27453937.
- Müller, Marius N. (2019). "On the Genesis and Function of Coccolithophore Calcification". Frontiers in Marine Science. 6. doi:10.3389/fmars.2019.00049.
- Hamm, Christian; Smetacek, Victor (2007). "Armor: Why, when, and How". Evolution of Primary Producers in the Sea. pp. 311–332. doi:10.1016/B978-012370518-1/50015-1. ISBN 9780123705181.
- Brussaard, Corina P. D. (2004). "Viral Control of Phytoplankton Populations-a Review1". The Journal of Eukaryotic Microbiology. 51 (2): 125–138. doi:10.1111/j.1550-7408.2004.tb00537.x. PMID 15134247. S2CID 21017882.
- Johns, Christopher T.; Grubb, Austin R.; Nissimov, Jozef I.; Natale, Frank; Knapp, Viki; Mui, Alwin; Fredricks, Helen F.; Van Mooy, Benjamin A. S.; Bidle, Kay D. (2019). "The mutual interplay between calcification and coccolithovirus infection". Environmental Microbiology. 21 (6): 1896–1915. doi:10.1111/1462-2920.14362. PMC 7379532. PMID 30043404.
- Haunost, Mathias; Riebesell, Ulf; Bach, Lennart T. (2020). "The Calcium Carbonate Shell of Emiliania huxleyi Provides Limited Protection Against Viral Infection". Frontiers in Marine Science. 7. doi:10.3389/fmars.2020.530757.
- Calbet, Albert; Landry, Michael R. (2004). "Phytoplankton growth, microzooplankton grazing, and carbon cycling in marine systems". Limnology and Oceanography. 49 (1): 51–57. Bibcode:2004LimOc..49...51C. doi:10.4319/lo.2004.49.1.0051. hdl:10261/134985. S2CID 22995996.
- Mayers, K.M.J.; Poulton, A.J.; Daniels, C.J.; Wells, S.R.; Woodward, E.M.S.; Tarran, G.A.; Widdicombe, C.E.; Mayor, D.J.; Atkinson, A.; Giering, S.L.C. (2019). "Growth and mortality of coccolithophores during spring in a temperate Shelf Sea (Celtic Sea, April 2015)". Progress in Oceanography. 177: 101928. Bibcode:2019PrOce.17701928M. doi:10.1016/j.pocean.2018.02.024. S2CID 135347218.
- Young, J. R. (1994) "Functions of coccoliths". In: Coccolithophores, Eds A. Winter and W. G. Siesser (Cambridge: Cambridge University Press), 63–82.
- Tillmann, Urban (2004). "Interactions between Planktonic Microalgae and Protozoan Grazers1". The Journal of Eukaryotic Microbiology. 51 (2): 156–168. doi:10.1111/j.1550-7408.2004.tb00540.x. PMID 15134250. S2CID 36526359.
- Breckels, M. N.; Roberts, E. C.; Archer, S. D.; Malin, G.; Steinke, M. (2011). "The role of dissolved infochemicals in mediating predator-prey interactions in the heterotrophic dinoflagellate Oxyrrhis marina". Journal of Plankton Research. 33 (4): 629–639. doi:10.1093/plankt/fbq114.
- Huxley, Thomas Henry (1858). "Appendix A". Deep Sea Soundings in the North Atlantic Ocean between Ireland and Newfoundland, made in H.M.S. Cyclops, Lieut.-Commander Joseph Dayman, in June and July 1857. London: British Admiralty. pp. 63–68 [64].
- Huxley, Thomas Henry (1868). "On some organisms living at great depth in the North Atlantic Ocean". Quarterly Journal of Microscopical Science. New series. 8: 203–212.
- Sorby, Henry Clifton (1861). "On the organic origin of the so-called 'Crystalloids' of the chalk". Annals and Magazine of Natural History. Ser. 3. 8 (45): 193–200. doi:10.1080/00222936108697404.
- Fortuño, José Manuel; Cros, Lluïsa (2002-03-30). "Atlas of Northwestern Mediterranean Coccolithophores". Scientia Marina. 66 (S1): 1–182. doi:10.3989/scimar.2002.66s11. hdl:10261/5441. ISSN 1886-8134.
- Malinverno, E; Dimiza, MD; Triantaphyllou, MV; Dermitzakis, MD; Corselli, C (2008). Coccolithophores of the Eastern Mediterranean sea: A look into the marine microworld. Athens: "ION" Publishing Group. ISBN 978-960-411-660-7.
- Amos Winter; William G. Siesser (2006). Coccolithophores. Cambridge University Press. pp. 54–58. ISBN 978-0-521-03169-1.
- Carmelo R Tomas (2012). Marine Phytoplankton: A Guide to Naked Flagellates and Coccolithophorids. Academic Press. pp. 161–165. ISBN 978-0-323-13827-7.
- Bendif, El Mahdi; Probert, Ian; Díaz-Rosas, Francisco; Thomas, Daniela; van den Engh, Ger; Young, Jeremy R.; von Dassow, Peter (2016-05-24). "Recent Reticulate Evolution in the Ecologically Dominant Lineage of Coccolithophores". Frontiers in Microbiology. Frontiers Media SA. 7. doi:10.3389/fmicb.2016.00784. ISSN 1664-302X.
 Modified material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Modified material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
External links
- The EHUX website - site dedicated to Emiliania huxleyi, containing essays on blooms, coccolith function, etc.
- International Nannoplankton Association site - includes an illustrated guide to coccolith terminology and several image galleries.
- Nannotax - illustrated guide to the taxonomy of coccolithophores and other nannofossils.
- Cocco Express - Coccolithophorids Expressed Sequence Tags (EST) & Microarray Database
- Possible functions of Coccoliths