Common symbiosis signaling pathway
The common symbiosis signaling pathway (CSSP) is a signaling cascade in plants that seen to be activated in both NOD-factor perception (for nodule forming rhizobia), as well as found in MYC-factor perception that are released from Arbuscular mycorrhizal fungi. The pathway is distinguished from the pathogen recognition pathways, but may have some common receptors involved in both pathogen recognition as well as CSSP. A recent work [1] by Kevin Cope and colleagues shown that possibly other type of mycorrhizae may involve the CSSP components such as Myc-factor recognition.
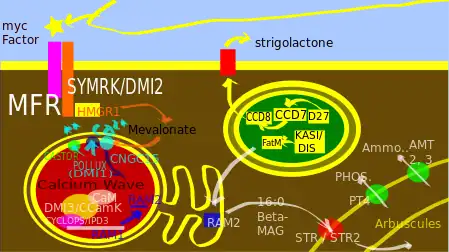

The AMF colonization requires the following chain[2] of events that can be roughly divided into following steps - 1: The Pre-Contact Signaling,
1: The Pre-Contact Signaling,
2: The CSSP
2: A: Perception
2: B: Transmission
2: C: Transcription
3: The Accommodation program
Outline
To accurately recognize the infection thread of a different species of organism, and to establish a mutually beneficial association requires robust signaling.[3] AM fungi are also fatty acid auxotrophs;[4][2] therefore they depend on plant for supply of fatty acid supply.[5]
At the pre-symbiotic signaling; both symbionts release chemical factors in their surroundings so that the partners can find each other.[6]' Plant root exudates play role in complex microbial interaction,[7] by releasing a lot of versatile materials.[7][8][9] among which strigolactone has been identified to facilitate both AMF colonisation and pathogen infection.[8]
It is seen that phosphate starvation in plant induces strigolactone production as well as AMF colonisation.[8] Plants release strigolactone, a class of caroteinoid-based plant hormone which also attracts the fungal symbionts and stimulate the fungal oxidative metabolism and also growth and branching of the fungal partner [2] Strigolactone promotes hyphal branching in germinating AMF spores [9] It plays role in intense ramification of the AMF hyphae at the vicinity of root and then colonization [10]
The common symbiosis signalling pathway is called so because it has common components for fungal symbiosis as well as rhizobial symbiosis. The common signalling pathway probably evolved when the existing pathway for arbuscular mycorrhizae was exploited by rhizobia,.[2][11]
The perception happens when fungal Myc factor is detected by plant. Myc factors are comparable to rhizobial nod factors. The chemical nature of Myc factor has recently been revealed as lipo-chito-oligosaccharide (Myc-LCOs) and chito-oligosaccharides (Myc-COs) that work as symbiotic signal,[10][12][13]
Presence of Strigolactone enhances the production of Myc-CO production by AMF [10]
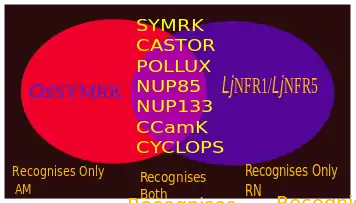
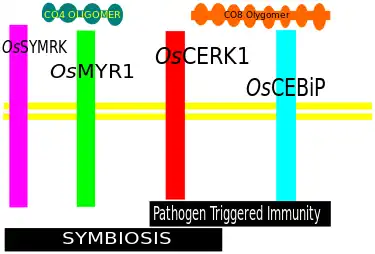
Myc factor receptor (MFR) is still putative, however However, another protein DMI2 (or SYMRK) that have prominent role in perception process and it is thought to be a co-receptor of MFR. Rice plant probably show a different mechanism using OsCERK1 and OsCEBiP which probably detect chitin oligomers[2][14][15]
The transmission happens when the signal transmitted after detection to the gene expression stage. This process is mediated by two nucleoporins NUP85 and NUP133,[11] Alternatively, another hypothesis says HMG-CoA reductase is activated on perception, which then converts HMG-CoA into mevalonate, this mevalonate acts as a second messenger and activates a nuclear K+ cation channel (DMI-1 or Pollux).[2][16] The transmission stage ends by creating a ‘calcium spike’ into the nucleus [17]
The transcription stage starts when a Calcium and Calmodulin dependent kinase (CCaMK) is activated.[2] then it stimulates a target protein CYCLOPS.[2] CCaMK and CYCLOPS probably forms a complex that along with DELLA protein, regulates the expression of RAM1 (Reduced Arbuscular Mycorrhyza1) gene expression.[2]
The accommodation process is the extensive remodelling of host cortical cells. This includes invagination of host plasmalemma, proliferation of endoplasmic reticulum, golgi apparatus, trans-golgi network and secretary vesicles. Plastids multiply and form “stromules”. Vacuoles also goes through extensive reorganization [11]
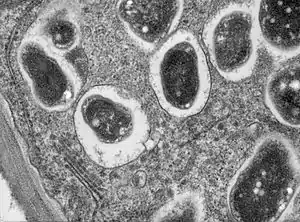 A TEM section of root nodule showing symbiosome made by Rhizobia, a kind of nitrogen fixing bacteria, in this case Bradyrhizobium japonicum in soybean root. |
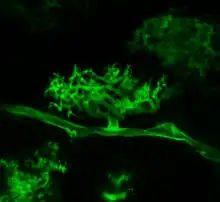 An arbuscle formed by Arbuscular mycorrhizal fungus Rhizophagus irregularis in Maize root, stained with WGA-Alexa fluor, seen using fluorescence microscopy. |
|---|
The Pre Contact Signaling
Chemical signalling starts prior to two symbionts come into contact. From the host plant's side, it synthesizes and releases a range of caroteinoid based phytohormone, called strigolactones.[2] They have a conserved tricyclic lactone structure also known as ABC rings.[18] Strigolactone biosynthesis occurs mainly in plastid,[19] where D27 (Rice DWARF 27; Arabidopsis ortholog ATD27), an Iron binding beta-carotene isomerase works at upstream of strigolactone biosynthesis [19] Then carotenoid cleavage dioxygenase enzyme CCD7 and CCD8 modifies the structure, which has following orthologs:
| Gene name | Localization | function | Rice ortholog | Pea ortholog | Petunia ortholog | Arabidopsis ortholog |
|---|---|---|---|---|---|---|
| CCD7 | Plastid proteins | involved in strigolactone biosynthesis | D17/ HTD1 | RMS5 | DAD3 | MAX3 |
| CCD8 | Plastid proteins | involved in strigolactone biosynthesis | D10 | RMS1 | DAD4 | MAX4 |
| Alpha/Beta fold hydrolase | Nuclear proteins | involved in strigolactone perception | D14 | RMS3 | DAD2 | ? |
The alpha/beta fold hydrolase D3 and also D14L (D14-Like) (Later one has Arabidopsis ortholog KAI2, or KARRIKIN INSENSITIVE-2) is reported to have important roles in mycorrhizal symbiosis [3], notably, D3, D14 and D14L are localised in nucleus.[2]
NOPE1 or 'NO PERCEPTION 1', newly discovered transporter protein in Rice (Oryza sativa) and Maize (Zea mays), also required for the priming stage for colonisation by the fungus. NOPE1 is a member of Major Facilitator Super family of transport proteins, capable of N-acetylglucosamine transport. Since nope1 mutant's root exudates fail to elicited transcriptional responses in fungi, it strongly seems NOPE1 secretes something (not yet characterised) that promotes fungal response [2]
Perception
.svg.png.webp)
There are two main type of root symbiosis; one is root nodule symbiosis by Rhizobia (RN-type) and another is Arbuscular Mycorrhiza (AM-type). There are common genes involved in between these two pathways.[20] these key common components, form the Common Symbiosis pathway (CSP or CSSP).[20] It has been proposed that, RN symbiosis has originated from AM symbiosis.[11] The perception of presence of fungal symbiont, takes place mainly through fungal chemical secretions generally termed as Myc factors. Receptors for Myc factors are yet to be identified. However, DMI2/SYMRK probably acts as a co-receptor of Myc factor receptor (MFR). The AM fungal secreted materials relevant to symbiosis are Myc-LCOs, Myc-COs, N-Acetylglucosamine [2][21]
| Myc factor | Plant protein it mainly act on |
|---|---|
| Myc-LCOs | LYS11 in Lotus japonicus |
| Short chain chitin oligomers (COs) | OsCERK1 and OsCEBiP in rice |
| N-acetylglucosamine | NOPE-1 in maize |
Myc-LCOs. (lipochitooligosaccharides)
Like Rhizobial LCOs (Nod factors); Myc-LCOs play important role in perception stage. They are kind of secreted materials from AM fungi, mainly mixtures of lipo-chito-oligosaccharides (Myc-LCOs) . In Lotus japonicus, LYS11, a receptor for LCOs, was expressed in root cortex cells associated with intra-radical colonizing arbuscular mycorrhizal fungi [21]
Short chain chitin oligomers (Myc-COs)
AM host plants show symbiotic-like calcium wave upon exposure to short chain chitin oligomers. It has been reported that production of these molecules by AM fungus Rhizophagus irregularis, gets strongly stimulated upon exposure to strigolactones [2] This gives hint to a model that plants secrete strigolactones and as a reply to it, this fungus increases short chain chitin oligomer, which in turns elicits the plant response to accommodate the fungus. The lysine motif OsCERK1 and OsCEBiP is thought to be involved with perception of short chain chitin oligomers.[2]
N-Acetylglucosamine.
NOPE-1 transporter has been described already. NOPE-1 also shows a strong N-acetylglucosamine uptake activity, and is thought to be associated with recognition of presence of fungal symbiont.[2]
Some plant proteins suspected to recognise Myc-factors, Rice OsCERK1 Lysin motif (LysM) receptor-like kinase, is one of them.[15]
Cell Surface Receptors
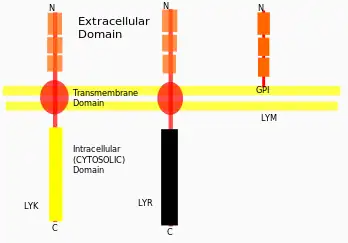
There are multiple families of pattern recognition receptors and co-receptors involved in recognition of microbial pathogens and symbionts. Some of the relevant families involved in CSSP, are Membrane bound LysMs (LYM), Soluble LysM Receptor like Protein, LYK (LysM receptors with active Kinase domain), LYR (LysM proteins with inactive kinase domain), etc. (ref)
Seemingly, different combinations of a LYK and LYR generates differential signals, such as some combinations generate a pathogen recognition signal whereas some combinations rise to the symbiotic signal [22][23][24][25]
Receptor like Kinase (RLKs)
DMI2/ SYMRK is a receptor like kinase, an important protein in endosymbiosis signal perception, reported in several plants (Mt-DMI2 or Mt-NORK in Medicago trancatula; Lj-SYMRK in Lotus japonicas; Ps-SYM19 in Pisum sativum; OsSYMRK in Rice). OsSYMRK lacks an N terminal domain and exclusively regulate AM symbiosis, does not work for RN symbiosis.[26] Notably, it has been found that a Nodulation-factor inducible gene, MtENOD11 get activated in presence of AMF exudates; Little is known about this phenomenon.[27][28]
LysM receptor-like kinase
Lysin Motif (LysM) receptor-like kinase are a subfamily related to membrane bound Receptor like kinase (RLKs) with an extracellular region consisting of 3 Lysine motifs. They have some important orthologs in different plants, that vary in their function. (In some plant they are involved in AM symbiosis, in some plants they are not). Tomato (Solanum lycopersicum), a non-legume dicot, also have a similar LysM receptor, SlLYK10 that Promotes AM symbiosis. There are some co-receptors of Myc-factor receptor viz., OsCEBiP in Rice, a LysM membrane protein can function as a co-receptor of OsCERK1 but it works in a different pathway.[29][30][31]
Most of these kinases are Serine/Threonine kinase, some are Tyrosine kinase.[32] Also they are type-1 transmembrane proteins, that indicates their N-terminal domain towards the outside of the cell, and the C-terminal domain is towards inside of the cell.[24]
| Medicago truncatula | Lotus japonicus | Pisum sativum
(pea) |
Prunus persica | Arabidopsis thalliana | Brassica rapa | Solanum lycopersicum
(Tomato) |
Brachypodium distachyon | Oryza sativa
(Rice) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lysine Motif
Receptor-Like Kinase and Lysine Motif Receptor like Protein |
LYM | LYMI | LYM1 | PpLYM1 | AtLYM1
AtLYM3 |
SlLYM1 | BdLYM1
BdLYM3 |
OsLYP6
OsLYP5, OsLYP4 | |||||
| LYMII | LYM2 | PpLYM3
PpLYM2 |
AtLYM2 | SlLYM3
SlLYM2 |
BdLYM2
BdLYM4 |
OsCEBiP
OsLYP3 | |||||||
| LYR | LYR 1 | LYRIA | MtNFP
MtLYR1 |
LjNFR5
LjLYS11 |
PpLYR1 | SlLyk10 | Bd LYR1 | OsNFR5 | |||||
| LYRIB | MtLYR8 | PpLYR2 | SlLYK9 | Bd LYR2 | |||||||||
| LYR 2 | LIRIIA | MtLYR10 | LjLYS16 | PpLYR6 | AtLYK2 | SlLYK2 | |||||||
| LYRIIB | MtLYR9 | LjLYS15 | PpLYR7 | SlLYK15 | |||||||||
| LYR 3 | LYRIIIA | MtLYR3 | LjLYS12 | PpLYR3 | AtLYK4 | SlLYK4 | Bd LYR4 | OsLYK6 | |||||
| LYRIIIB | MtLYR2 | PpLYR4 | SlLYK7
SlLYK6 |
||||||||||
| LYRIIIC | MtLYR4
MtLYR7 |
LjLYS13
LjLYS14 |
AtLYK5 | Bd LYR3 | OsLYK3
OsLYK2, OsLYK4 | ||||||||
| LYR 4 | LYRIV | MtLYR5
MtLYR6 |
LjLYS20 | PpLYR5 | |||||||||
| LYK | LYKI | LYK1, LYK4, LYK5, LYK6, LYK7, LYK2, LYK3, LYK9, LYK8 | LjLYS2
LjLYS1, LjNFR1, LjLYS6, LjLYS7 |
PpLYK2
PpLyk1 |
AtLYK1/
AtCERK1 |
SlLYK13
SlLYK1/ SlBti9, SlLYK12, SlLYK11 |
BdLYK1 | OsCERK1 | |||||
| LYKII | LYK10 | LjLYS3/
EPR3 |
PpLYK3
PpLYK4 |
||||||||||
| LYKII | PpLYK5 | AtLYK3 | SlLYK3 | BdLYK3 | |||||||||
| Receptor like Kinase | RLK | Mt-DMI2/
Mt-NORK |
Lj-SYMRK | Ps-SYM19 | OsSYMRK | ||||||||
Transmission
The transmission of signal cascades into nucleus is not well understood. However, this transmission includes carrying the message up to the nuclear membrane and generation of a calcium wave.[33] Some elements involved in this process are as follows:
Nucleoporins
Lotus japonicas Nucleoporins LjNUP85 and LjNUP133 has potential role in transmission of the signal.[34] Lj-NENA is another important nucleoporin that plays role in AM symbiosis.[35]
HMGR and Mevalonate.
It has been proposed that the enzyme 3-hydroxy-3-methylglutaryl-CoA reductase (HMG CoA reductase or HMGR) has potential role in the transmission stage. The enzyme is activated by SYMRK/DMI2, and forms mevalonate.[36][37] This mevalonate acts as a second messenger, and activates a nuclear potassium channel, DMI1 or pollux.[37]
| Nuclear envelope Protein | Function | Rice | Lotus japonicus | Medicago truncatula | Pisum |
| CNGC15 | Cyclic-nucleotide gated Calcium-channel | Mt-CNGC15 | |||
| Castor | Potassium cation channel | Os-Castor | Lj-Castor | ||
| POLLUX or DMI1 | Potassium cation channel | OsPOLLUX | LjPOLLUX | Mt-DMI1 | Ps-SYM8 |
Nuclear membrane cation channels.
The nuclear calcium channel CNGC15, which is cyclic nucleotide gated ion channel; mediates the symbiotic nuclear Ca2+ influx, and it is countered by K+ efflux by DMI1[36]
Transcription
| Protein | Function | Name of the Plant | |||
| Rice | Lotus japonicus | Medicago Truncatula | Pisum sativum | ||
| CCamK | Calcium calmodulin-dependent kinase with role in AMF symbiosis | Os-DMI3 or
Os-CCaMK |
Lj-CCaMK | Mt-DMI3 | Ps-SYM9 |
| CYCLOPS | Coiled coil domain containing proteins that respond to CCamK and promote AMF symbiosis | Os-CYCLOPS | Lj-CYCLOPS | Mt-IPD3 | Ps-SYM33 |
| DELLA | Promote AMF symbiosis | Os-SLR1 | Mt-DELLA1
Mt-DELLA2 |
Ps-LA
Ps-CRY | |
Calmodulin is a widespread regulatory protein that functions along with Ca2+ in various biological processes. In AM symbiosis signalling it modulates the CCaMK.[36] CCaMK or DMI3 is a calcium-and-calmodulin-dependent kinase (CCaMK) is thought to be a key decoder of Ca2+ oscillation and an important regulatory kinase protein. Nuclear Ca2+ spiking promotes binding of Ca2+ calmodulin with CCaMK.[36] Binding of Ca2+ calmodulin with CCaMK causes conformational change of CCaMK that stimulates a target protein CYCLOPS which has different orthologs.[36] CYCLOPS is a coiled coil domain containing protein [36] possibly form a complex with CCaMK[36] that works along with DELLA proteins. DELLA proteins are kind of GRAS-domain protein and were originally identified as repressors of Gibberellin signalling pathway, however now it is seen that DELLA provides a mechanism for crosstalk between many signalling pathways.[39] There are two DELLA proteins in Medicago trancatula and Pisum sativum play role in symbiosis whereas in rice plant only one DELLA protein fulfils this task.[36] Reduced Arbuscular Mycorrhiza or RAM1[36] is a GRAS[40] protein whose gene is directly regulated by DELLA and CCaMK/ CYCLOPS.[36] Using Chromatin immune precipitation assay it has been shown that RAM1 binds to RAM2 gene promoter.[36] RAM1 also play role in many of the fungal accommodation genes directly or indirectly.
A bunch of GRAS proteins play role in AM symbiosis whose roles are not yet fully understood. These includes RAD1 (REQUIRED FOR ARBUSCLE DEVELOPMENT 1), MIG1 (MYCORRHIZA INDUCED GRAS1), NSP1, NSP2 etc.[36] WRKY transcription factor genes are thought to play very important roles in establishment of mycorrhizal symbiosis and they perhaps work through regulating plant defense genes.[41]
The Accommodation program
Huge reorganization of cortex cells takes place in order to make accommodation for the fungal endosymbiont. The pre-penetration apparatus (PPA) like structure and the peri-arbuscular membrane have to form and the cytoplasm have to retract,[34] so, the vacuole retract in size, the nucleus and nucleolus enlarge in size and chromatin decondense indicating heightened transcriptional activity,[34] Plastids multiply and stay connected with “stromulus”.[34] Furthermore, it was suggested that the apoplastic longitudinal hyphal growth is probably regulated by plant genes such as taci1 and CDPK1.[42]
Genes and proteins playing role in accommodation programme
Although various proteins have been identified which may play role on how this accommodation process happens, the detailed signalling cascade is not well understood. Some of the proteins and machineries involved in the deposition on peri-arbuscular membrane are EXOCYST complex, EXO70 subunit, a symbiosis-specific splice variant of SYP132, VAPYRIN, two variants of VAMP721 etc.[36] Plant enzymes FatM and RAM2[43] and ABC transporter STR/STR2 is involved in synthesis and supplying of a lipid 16:0 β-monoacylglycerol to the AM fungi.[43][44]
The protein composition of peri-arbuscular membrane is very different from plasma membrane. It includes some special transporters such as phosphate transporter (Mt-PT4, Os-PT11, Os-PT13), Ammonium transporter (Mt-AMT2 and 3) etc. and ABC transporters such as lipid transporter STR/STR2[36][45]
Evolutionary significance
AM fungi and plants co-evolved and developed a very complex interaction that allow the plant accommodate the AM-fungal host [46][47][48] It has been proposed that, RN symbiosis has originated from AM symbiosis[34][35]
See also
References
- Cope, Kevin R.; Bascaules, Adeline; Irving, Thomas B.; Venkateshwaran, Muthusubramanian; Maeda, Junko; Garcia, Kevin; Rush, Tomás A.; Ma, Cathleen; Labbé, Jessy; Jawdy, Sara; Steigerwald, Edward; Setzke, Jonathan; Fung, Emmeline; Schnell, Kimberly G.; Wang, Yunqian; Schleif, Nathaniel; Bücking, Heike; Strauss, Steven H.; Maillet, Fabienne; Jargeat, Patricia; Bécard, Guillaume; Puech-Pagès, Virginie; Ané, Jean-Michel (October 2019). "The Ectomycorrhizal Fungus Laccaria bicolor Produces Lipochitooligosaccharides and Uses the Common Symbiosis Pathway to Colonize Populus Roots". The Plant Cell. 31 (10): 2386–2410. doi:10.1105/tpc.18.00676. PMC 6790088. PMID 31416823.
- MacLean, Allyson M.; Bravo, Armando; Harrison, Maria J. (October 2017). "Plant Signaling and Metabolic Pathways Enabling Arbuscular Mycorrhizal Symbiosis". The Plant Cell. 29 (10): 2319–2335. doi:10.1105/tpc.17.00555. PMC 5940448. PMID 28855333.
- Barman, Jagnaseni; Samanta, Aveek; Saha, Babita; Datta, Siraj (December 2016). "Mycorrhiza: The oldest association between plant and fungi". Resonance. 21 (12): 1093–1104. doi:10.1007/s12045-016-0421-6. ISSN 0971-8044. S2CID 109932925.
- Luginbuehl, Leonie H.; Menard, Guillaume N.; Kurup, Smita; Van Erp, Harrie; Radhakrishnan, Guru V.; Breakspear, Andrew; Oldroyd, Giles E. D.; Eastmond, Peter J. (16 June 2017). "Fatty acids in arbuscular mycorrhizal fungi are synthesized by the host plant". Science. 356 (6343): 1175–1178. doi:10.1126/science.aan0081. PMID 28596311. S2CID 206658467.
- Bravo, Armando; Brands, Mathias; Wewer, Vera; Dörmann, Peter; Harrison, Maria J. (June 2017). "Arbuscular mycorrhiza‐specific enzymes FatM and RAM 2 fine‐tune lipid biosynthesis to promote development of arbuscular mycorrhiza". New Phytologist. 214 (4): 1631–1645. doi:10.1111/nph.14533. PMID 28380681. S2CID 3481181.
- Paszkowski, Uta (October 2006). "A journey through signaling in arbuscular mycorrhizal symbioses 2006". New Phytologist. 172 (1): 35–46. doi:10.1111/j.1469-8137.2006.01840.x. PMID 16945087.
- Bais, Harsh P.; Weir, Tiffany L.; Perry, Laura G.; Gilroy, Simon; Vivanco, Jorge M. (1 June 2006). "The Role of Root Exudates in Rhizosphere Interactions with Plants and Other Organisms". Annual Review of Plant Biology. 57 (1): 233–266. doi:10.1146/annurev.arplant.57.032905.105159. PMID 16669762.
- Badri, Dayakar V.; Vivanco, Jorge M. (June 2009). "Regulation and function of root exudates". Plant, Cell & Environment. 32 (6): 666–681. doi:10.1111/j.1365-3040.2009.01926.x. PMID 19143988.
- De-la-Peña, Clelia; Badri, Dayakar V.; Loyola-Vargas, Víctor M. (2012). "Plant Root Secretions and Their Interactions with Neighbors". Secretions and Exudates in Biological Systems. Signaling and Communication in Plants. Vol. 12. pp. 1–26. doi:10.1007/978-3-642-23047-9_1. ISBN 978-3-642-23046-2.
- Genre, Andrea; Chabaud, Mireille; Balzergue, Coline; Puech‐Pagès, Virginie; Novero, Mara; Rey, Thomas; Fournier, Joëlle; Rochange, Soizic; Bécard, Guillaume; Bonfante, Paola; Barker, David G. (April 2013). "Short‐chain chitin oligomers from arbuscular mycorrhizal fungi trigger nuclear C a 2+ spiking in M edicago truncatula roots and their production is enhanced by strigolactone". New Phytologist. 198 (1): 190–202. doi:10.1111/nph.12146. PMID 23384011. S2CID 34009711.
- Bonfante, Paola; Genre, Andrea (27 July 2010). "Mechanisms underlying beneficial plant–fungus interactions in mycorrhizal symbiosis". Nature Communications. 1 (1): 48. Bibcode:2010NatCo...1...48B. doi:10.1038/ncomms1046. PMID 20975705.
- Maillet, Fabienne; Poinsot, Véréna; André, Olivier; Puech-Pagès, Virginie; Haouy, Alexandra; Gueunier, Monique; Cromer, Laurence; Giraudet, Delphine; Formey, Damien; Niebel, Andreas; Martinez, Eduardo Andres; Driguez, Hugues; Bécard, Guillaume; Dénarié, Jean (2011). "Fungal lipochitooligosaccharide symbiotic signals in arbuscular mycorrhiza". Nature. 469 (7328): 58–63. Bibcode:2011Natur.469...58M. doi:10.1038/nature09622. PMID 21209659. S2CID 4373531.
- Sun, Jongho; Miller, J. Benjamin; Granqvist, Emma; Wiley-Kalil, Audrey; Gobbato, Enrico; Maillet, Fabienne; Cottaz, Sylvain; Samain, Eric; Venkateshwaran, Muthusubramanian; Fort, Sébastien; Morris, Richard J.; Ané, Jean-Michel; Dénarié, Jean; Oldroyd, Giles E.D. (9 April 2015). "Activation of Symbiosis Signaling by Arbuscular Mycorrhizal Fungi in Legumes and Rice". The Plant Cell. 27 (3): 823–838. doi:10.1105/tpc.114.131326. PMC 4558648. PMID 25724637.
- Genre, A.; Bonfante, P. (February 2020). "A Rice Receptor for Mycorrhizal Fungal Signals Opens New Opportunities for the Development of Sustainable Agricultural Practices". Molecular Plant. 13 (2): 181–183. doi:10.1016/j.molp.2020.01.009. PMID 31981734. S2CID 210912425.
- Miyata, Kana; Kozaki, Toshinori; Kouzai, Yusuke; Ozawa, Kenjirou; Ishii, Kazuo; Asamizu, Erika; Okabe, Yoshihiro; Umehara, Yosuke; Miyamoto, Ayano; Kobae, Yoshihiro; Akiyama, Kohki; Kaku, Hanae; Nishizawa, Yoko; Shibuya, Naoto; Nakagawa, Tomomi (November 2014). "The Bifunctional Plant Receptor, OsCERK1, Regulates Both Chitin-Triggered Immunity and Arbuscular Mycorrhizal Symbiosis in Rice". Plant and Cell Physiology. 55 (11): 1864–1872. doi:10.1093/pcp/pcu129. PMID 25231970.
- Venkateshwaran, Muthusubramanian; Jayaraman, Dhileepkumar; Chabaud, Mireille; Genre, Andrea; Balloon, Allison J.; Maeda, Junko; Forshey, Kari; den Os, Désirée; Kwiecien, Nicholas W.; Coon, Joshua J.; Barker, David G.; Ané, Jean-Michel (4 August 2015). "A role for the mevalonate pathway in early plant symbiotic signaling". Proceedings of the National Academy of Sciences. 112 (31): 9781–9786. Bibcode:2015PNAS..112.9781V. doi:10.1073/pnas.1413762112. PMC 4534228. PMID 26199419.
- Charpentier, Myriam; Oldroyd, Giles E.D. (8 October 2013). "Nuclear Calcium Signaling in Plants". Plant Physiology. 163 (2): 496–503. doi:10.1104/pp.113.220863. PMC 3793031. PMID 23749852.
- Saeed, Wajeeha; Naseem, Saadia; Ali, Zahid (28 August 2017). "Strigolactones Biosynthesis and Their Role in Abiotic Stress Resilience in Plants: A Critical Review". Frontiers in Plant Science. 8: 1487. doi:10.3389/fpls.2017.01487. PMC 5581504. PMID 28894457.
- Al-Babili, Salim; Bouwmeester, Harro J. (29 April 2015). "Strigolactones, a Novel Carotenoid-Derived Plant Hormone". Annual Review of Plant Biology. 66 (1): 161–186. doi:10.1146/annurev-arplant-043014-114759. PMID 25621512.
- Nakagawa, Tomomi; Imaizumi-Anraku, Haruko (December 2015). "Rice arbuscular mycorrhiza as a tool to study the molecular mechanisms of fungal symbiosis and a potential target to increase productivity". Rice. 8 (1): 32. doi:10.1186/s12284-015-0067-0. PMC 4626465. PMID 26516078.
- Rasmussen, S. R.; Füchtbauer, W.; Novero, M.; Volpe, V.; Malkov, N.; Genre, A.; Bonfante, P.; Stougaard, J.; Radutoiu, S. (20 July 2016). "Intraradical colonization by arbuscular mycorrhizal fungi triggers induction of a lipochitooligosaccharide receptor". Scientific Reports. 6 (1): 29733. Bibcode:2016NatSR...629733R. doi:10.1038/srep29733. PMC 4951684. PMID 27435342.
- Genre, A.; Bonfante, P. (February 2020). "A Rice Receptor for Mycorrhizal Fungal Signals Opens New Opportunities for the Development of Sustainable Agricultural Practices". Molecular Plant. 13 (2): 181–183. doi:10.1016/j.molp.2020.01.009. PMID 31981734. S2CID 210912425.
- Miyata, Kana; Kozaki, Toshinori; Kouzai, Yusuke; Ozawa, Kenjirou; Ishii, Kazuo; Asamizu, Erika; Okabe, Yoshihiro; Umehara, Yosuke; Miyamoto, Ayano; Kobae, Yoshihiro; Akiyama, Kohki; Kaku, Hanae; Nishizawa, Yoko; Shibuya, Naoto; Nakagawa, Tomomi (November 2014). "The Bifunctional Plant Receptor, OsCERK1, Regulates Both Chitin-Triggered Immunity and Arbuscular Mycorrhizal Symbiosis in Rice". Plant and Cell Physiology. 55 (11): 1864–1872. doi:10.1093/pcp/pcu129. ISSN 1471-9053. PMID 25231970.
- Buendia, Luis; Girardin, Ariane; Wang, Tongming; Cottret, Ludovic; Lefebvre, Benoit (2018-10-24). "LysM Receptor-Like Kinase and LysM Receptor-Like Protein Families: An Update on Phylogeny and Functional Characterization". Frontiers in Plant Science. 9: 1531. doi:10.3389/fpls.2018.01531. ISSN 1664-462X. PMC 6207691. PMID 30405668.
- He, Jiangman; Zhang, Chi; Dai, Huiling; Liu, Huan; Zhang, Xiaowei; Yang, Jun; Chen, Xi; Zhu, Yayun; Wang, Dapeng; Qi, Xiaofeng; Li, Weichao; Wang, Zhihui; An, Guoyong; Yu, Nan; He, Zuhua (December 2019). "A LysM Receptor Heteromer Mediates Perception of Arbuscular Mycorrhizal Symbiotic Signal in Rice". Molecular Plant. 12 (12): 1561–1576. doi:10.1016/j.molp.2019.10.015. PMID 31706032. S2CID 207966375.
- Nakagawa, Tomomi; Imaizumi-Anraku, Haruko (December 2015). "Rice arbuscular mycorrhiza as a tool to study the molecular mechanisms of fungal symbiosis and a potential target to increase productivity". Rice. 8 (1): 32. doi:10.1186/s12284-015-0067-0. ISSN 1939-8425. PMC 4626465. PMID 26516078.
- Paszkowski, Uta (October 2006). "A journey through signaling in arbuscular mycorrhizal symbioses 2006". New Phytologist. 172 (1): 35–46. doi:10.1111/j.1469-8137.2006.01840.x. ISSN 0028-646X. PMID 16945087.
- Kosuta, Sonja; Chabaud, Mireille; Lougnon, Géraldine; Gough, Clare; Dénarié, Jean; Barker, David G.; Bécard, Guillaume (2003-03-01). "A Diffusible Factor from Arbuscular Mycorrhizal Fungi Induces Symbiosis-Specific MtENOD11 Expression in Roots of Medicago truncatula". Plant Physiology. 131 (3): 952–962. doi:10.1104/pp.011882. ISSN 1532-2548. PMC 166861. PMID 12644648.
- MacLean, Allyson M.; Bravo, Armando; Harrison, Maria J. (October 2017). "Plant Signaling and Metabolic Pathways Enabling Arbuscular Mycorrhizal Symbiosis". The Plant Cell. 29 (10): 2319–2335. doi:10.1105/tpc.17.00555. ISSN 1040-4651. PMC 5940448. PMID 28855333.
- Genre, A.; Bonfante, P. (February 2020). "A Rice Receptor for Mycorrhizal Fungal Signals Opens New Opportunities for the Development of Sustainable Agricultural Practices". Molecular Plant. 13 (2): 181–183. doi:10.1016/j.molp.2020.01.009. PMID 31981734. S2CID 210912425.
- Miyata, Kana; Kozaki, Toshinori; Kouzai, Yusuke; Ozawa, Kenjirou; Ishii, Kazuo; Asamizu, Erika; Okabe, Yoshihiro; Umehara, Yosuke; Miyamoto, Ayano; Kobae, Yoshihiro; Akiyama, Kohki; Kaku, Hanae; Nishizawa, Yoko; Shibuya, Naoto; Nakagawa, Tomomi (November 2014). "The Bifunctional Plant Receptor, OsCERK1, Regulates Both Chitin-Triggered Immunity and Arbuscular Mycorrhizal Symbiosis in Rice". Plant and Cell Physiology. 55 (11): 1864–1872. doi:10.1093/pcp/pcu129. ISSN 1471-9053. PMID 25231970.
- Buendia, Luis; Girardin, Ariane; Wang, Tongming; Cottret, Ludovic; Lefebvre, Benoit (2018-10-24). "LysM Receptor-Like Kinase and LysM Receptor-Like Protein Families: An Update on Phylogeny and Functional Characterization". Frontiers in Plant Science. 9: 1531. doi:10.3389/fpls.2018.01531. ISSN 1664-462X. PMC 6207691. PMID 30405668.
- 3. MacLean AM, Bravo A, Harrison MJ. Plant signaling and metabolic pathways enabling arbuscularmycorrhizal symbiosis. The Plant Cell. 2017; 29(10):2319-2335. https://doi.org/10.1105/tpc.17.00555
- Bonfante, Paola; Genre, Andrea (2010-07-27). "Mechanisms underlying beneficial plant–fungus interactions in mycorrhizal symbiosis". Nature Communications. 1 (1): 48. Bibcode:2010NatCo...1...48B. doi:10.1038/ncomms1046. ISSN 2041-1723. PMID 20975705.
- Nakagawa, Tomomi; Imaizumi-Anraku, Haruko (December 2015). "Rice arbuscular mycorrhiza as a tool to study the molecular mechanisms of fungal symbiosis and a potential target to increase productivity". Rice. 8 (1): 32. doi:10.1186/s12284-015-0067-0. ISSN 1939-8425. PMC 4626465. PMID 26516078.
- MacLean, Allyson M.; Bravo, Armando; Harrison, Maria J. (October 2017). "Plant Signaling and Metabolic Pathways Enabling Arbuscular Mycorrhizal Symbiosis". The Plant Cell. 29 (10): 2319–2335. doi:10.1105/tpc.17.00555. ISSN 1040-4651. PMC 5940448. PMID 28855333.
- Venkateshwaran, Muthusubramanian; Jayaraman, Dhileepkumar; Chabaud, Mireille; Genre, Andrea; Balloon, Allison J.; Maeda, Junko; Forshey, Kari; den Os, Désirée; Kwiecien, Nicholas W.; Coon, Joshua J.; Barker, David G.; Ané, Jean-Michel (2015-08-04). "A role for the mevalonate pathway in early plant symbiotic signaling". Proceedings of the National Academy of Sciences. 112 (31): 9781–9786. Bibcode:2015PNAS..112.9781V. doi:10.1073/pnas.1413762112. ISSN 0027-8424. PMC 4534228. PMID 26199419.
- Kosuta, Sonja; Chabaud, Mireille; Lougnon, Géraldine; Gough, Clare; Dénarié, Jean; Barker, David G.; Bécard, Guillaume (2003-03-01). "A Diffusible Factor from Arbuscular Mycorrhizal Fungi Induces Symbiosis-Specific MtENOD11 Expression in Roots of Medicago truncatula". Plant Physiology. 131 (3): 952–962. doi:10.1104/pp.011882. ISSN 1532-2548. PMC 166861. PMID 12644648.
- Pimprikar, Priya; Gutjahr, Caroline (2018-04-01). "Transcriptional Regulation of Arbuscular Mycorrhiza Development". Plant and Cell Physiology. 59 (4): 678–695. doi:10.1093/pcp/pcy024. ISSN 0032-0781. PMID 29425360.
- Hirsch, Sibylle; Oldroyd, Giles E.D. (August 2009). "GRAS-domain transcription factors that regulate plant development". Plant Signaling & Behavior. 4 (8): 698–700. doi:10.4161/psb.4.8.9176. ISSN 1559-2324. PMC 2801379. PMID 19820314.
- Mohanta, Tapan Kumar; Bae, Hanhong (January 2015). "Functional genomics and signaling events in mycorrhizal symbiosis". Journal of Plant Interactions. 10 (1): 21–40. doi:10.1080/17429145.2015.1005180. ISSN 1742-9145. S2CID 85269506.
- Paszkowski, Uta (October 2006). "A journey through signaling in arbuscular mycorrhizal symbioses 2006". New Phytologist. 172 (1): 35–46. doi:10.1111/j.1469-8137.2006.01840.x. ISSN 0028-646X. PMID 16945087.
- Luginbuehl, Leonie H.; Menard, Guillaume N.; Kurup, Smita; Van Erp, Harrie; Radhakrishnan, Guru V.; Breakspear, Andrew; Oldroyd, Giles E. D.; Eastmond, Peter J. (2017-06-16). "Fatty acids in arbuscular mycorrhizal fungi are synthesized by the host plant". Science. 356 (6343): 1175–1178. doi:10.1126/science.aan0081. ISSN 0036-8075. PMID 28596311. S2CID 206658467.
- Bravo, Armando; Brands, Mathias; Wewer, Vera; Dörmann, Peter; Harrison, Maria J. (June 2017). "Arbuscular mycorrhiza‐specific enzymes FatM and RAM 2 fine‐tune lipid biosynthesis to promote development of arbuscular mycorrhiza". New Phytologist. 214 (4): 1631–1645. doi:10.1111/nph.14533. ISSN 0028-646X. PMID 28380681. S2CID 3481181.
- Wang, Wanxiao; Shi, Jincai; Xie, Qiujin; Jiang, Yina; Yu, Nan; Wang, Ertao (September 2017). "Nutrient Exchange and Regulation in Arbuscular Mycorrhizal Symbiosis". Molecular Plant. 10 (9): 1147–1158. doi:10.1016/j.molp.2017.07.012. PMID 28782719.
- Barman, Jagnaseni; Samanta, Aveek; Saha, Babita; Datta, Siraj (December 2016). "Mycorrhiza: The oldest association between plant and fungi". Resonance. 21 (12): 1093–1104. doi:10.1007/s12045-016-0421-6. ISSN 0971-8044. S2CID 109932925.
- Ercolin, Flavia; Reinhardt, Didier (July 2011). "Successful joint ventures of plants: arbuscular mycorrhiza and beyond". Trends in Plant Science. 16 (7): 356–362. doi:10.1016/j.tplants.2011.03.006. PMID 21459657.
- De-la-Peña, Clelia; Badri, Dayakar V.; Loyola-Vargas, Víctor M. (2012), Vivanco, Jorge M.; Baluška, František (eds.), "Plant Root Secretions and Their Interactions with Neighbors", Secretions and Exudates in Biological Systems, Signaling and Communication in Plants, Berlin, Heidelberg: Springer Berlin Heidelberg, vol. 12, pp. 1–26, doi:10.1007/978-3-642-23047-9_1, ISBN 978-3-642-23046-2, retrieved 2023-10-24