Mangrove forest
Mangrove forests, also called mangrove swamps, mangrove thickets or mangals, are productive wetlands that occur in coastal intertidal zones.[1][2] Mangrove forests grow mainly at tropical and subtropical latitudes because mangroves cannot withstand freezing temperatures. There are about 80 different species of mangroves, all of which grow in areas with low-oxygen soil, where slow-moving waters allow fine sediments to accumulate.[3]
| Marine habitats |
|---|
 |
Open ocean |
Many mangrove forests can be recognised by their dense tangle of prop roots that make the trees appear to be standing on stilts above the water. This tangle of roots allows the trees to handle the daily rise and fall of tides, as most mangroves get flooded at least twice per day. The roots slow the movement of tidal waters, causing sediments to settle out of the water and build up the muddy bottom. Mangrove forests stabilise the coastline, reducing erosion from storm surges, currents, waves, and tides. The intricate root system of mangroves also makes these forests attractive to fish and other organisms seeking food and shelter from predators.[3]
Mangrove forests live at the interface between the land, the ocean, and the atmosphere, and are centres for the flow of energy and matter between these systems. They have attracted much research interest because of the various ecological functions of the mangrove ecosystems, including runoff and flood prevention, storage and recycling of nutrients and wastes, cultivation and energy conversion.[4] The forests are major blue carbon systems, storing considerable amounts of carbon in marine sediments, thus becoming important regulators of climate change.[5][6] Marine microorganisms are key parts of these mangrove ecosystems. However, much remains to be discovered about how mangrove microbiomes contribute to high ecosystem productivity and efficient cycling of elements.[7]
Overview
| External video | |
|---|---|
– Mangrove Action Project |
There are about 80 different species of mangrove trees. All of these trees grow in areas with low-oxygen soil, where slow-moving waters allow fine sediments to accumulate. Mangrove forests grow only at tropical and subtropical latitudes near the equator because they cannot withstand freezing temperatures.[8] Many mangrove forests can be recognised by their dense tangle of prop roots that make the trees appear to be standing on stilts above the water. This tangle of roots allows the trees to handle the daily rise and fall of tides, which means that most mangroves get flooded at least twice per day. The roots slow the movement of tidal waters, causing sediments to settle out of the water and build up the muddy bottom. Mangrove forests stabilise the coastline, reducing erosion from storm surges, currents, waves, and tides. The intricate root system of mangroves makes these forests attractive to fishes and other organisms seeking food and shelter from predators.[9]
The main contribution of mangroves to the larger ecosystem comes from litter fall from the trees, which is then decomposed by primary consumers. Bacteria and protozoans colonise the plant litter and break it down chemically into organic compounds, minerals, carbon dioxide, and nitrogenous wastes.[9] The intertidal existence to which these trees are adapted represents the major limitation to the number of species able to thrive in their habitat. High tide brings in salt water, and when the tide recedes, solar evaporation of the seawater in the soil leads to further increases in salinity. The return of tide can flush out these soils, bringing them back to salinity levels comparable to that of seawater.[10][11] At low tide, organisms are exposed to increases in temperature and reduced moisture before being then cooled and flooded by the tide. Thus, for a plant to survive in this environment, it must tolerate broad ranges of salinity, temperature, and moisture, as well as several other key environmental factors—thus only a select few species make up the mangrove tree community.[11][10]
A mangrove swamp typically features only a small number of tree species. It is not uncommon for a mangrove forest in the Caribbean to feature only three or four tree species. For comparison, a tropical rainforest biome may contain thousands of tree species, but this is not to say mangrove forests lack diversity. Though the trees are few in species, the ecosystem that these trees create provides a habitat for a great variety of other species, including as many as 174 species of marine megafauna.[12]

Seagrass and oyster beds can inhabit the shallow subtidal zone
Mangrove plants require a number of physiological adaptations to overcome the problems of low environmental oxygen levels, high salinity, and frequent tidal flooding. Each species has its own solutions to these problems; this may be the primary reason why, on some shorelines, mangrove tree species show distinct zonation. Small environmental variations within a mangal may lead to greatly differing methods for coping with the environment. Therefore, the mix of species is partly determined by the tolerances of individual species to physical conditions, such as tidal flooding and salinity, but may also be influenced by other factors, such as crabs preying on plant seedlings.[14]
Once established, mangrove roots provide an oyster habitat and slow water flow, thereby enhancing sediment deposition in areas where it is already occurring. The fine, anoxic sediments under mangroves act as sinks for a variety of heavy (trace) metals which colloidal particles in the sediments have concentrated from the water. Mangrove removal disturbs these underlying sediments, often creating problems of trace metal contamination of seawater and organisms of the area.[15]
Mangrove swamps protect coastal areas from erosion, storm surge (especially during tropical cyclones), and tsunamis.[16][17][18] They limit high-energy wave erosion mainly during events such as storm surges and tsunamis.[19] The mangroves' massive root systems are efficient at dissipating wave energy.[20] Likewise, they slow down tidal water enough so that its sediment is deposited as the tide comes in, leaving all except fine particles when the tide ebbs.[21] In this way, mangroves build their environments.[16] Because of the uniqueness of mangrove ecosystems and the protection against erosion they provide, they are often the object of conservation programs,[11] including national biodiversity action plans.[17]
Distribution


Distribution of delta, estuary, lagoon and open coast mangrove types
in (i) South Asia, (ii) Southeast Asia and (iii) East Asia [23]
Bar charts show percentage change in area between 1996 and 2016
Worldwide there are about 80 described species of mangroves that live along marine coasts. About 60 of these species are true mangroves which live only in the intertidal zone between high and low tides.[24] "Mangroves once covered three-quarters of the world's tropical coastlines, with Southeast Asia hosting the greatest diversity. Only 12 species live in the Americas. Mangroves range in size from small bushes to the 60-meter giants found in Ecuador. Within a given mangrove forest, different species occupy distinct niches. Those that can handle tidal soakings grow in the open sea, in sheltered bays, and on fringe islands. Trees adapted to drier, less salty soil can be found farther from the shoreline. Some mangroves flourish along riverbanks far inland, as long as the freshwater current is met by ocean tides."[24]

orange: mangroves dominate green: salt marshes dominate
Mangroves can be found in 118 countries and territories in the tropical and subtropical regions of the world.[22] The largest percentage of mangroves is found between the 5° N and 5° S latitudes. Approximately 75% of world's mangroves are found in just 15 countries.[22] Estimates of mangrove area based on remote sensing and global data tend to be lower than estimates based on literature and surveys for comparable periods.[10]
In 2018, the Global Mangrove Watch Initiative released a global baseline based on remote sensing and global data for 2010.[22] They estimated the total mangrove forest area of the world as of 2010 at 137,600 km2 (53,100 sq mi), spanning 118 countries and territories.[10][25] Following the conventions for identifying geographic regions from the Ramsar Convention on Wetlands, researchers reported that Asia has the largest share (38.7%) of the world's mangroves, followed by Latin America and the Caribbean (20.3%), Africa (20.0%), Oceania (11.9%), and Northern America (8.4%).[25]
Sundarbans
The largest mangrove forest in the world is in the Sundarbans. The Sundarban forest lies in the vast delta on the Bay of Bengal formed by the super confluence of the Brahmaputra and Meghna rivers with distributaries of the Ganges. The seasonally flooded Sundarbans freshwater swamp forests lie inland from the mangrove forests on the coastal fringe. The forest covers 10,000 km2 (3,900 sq mi) of which about 6,000 km2 (2,300 sq mi) are in Bangladesh.[26]
The Sundarbans is intersected by a complex network of tidal waterways, mudflats and small islands of salt-tolerant mangrove forests. The interconnected network of waterways makes almost every portion of the forest accessible by boat. The area is known as an important habitat for the endangered Bengal tiger, as well as numerous fauna including species of birds, spotted deer, crocodiles and snakes. The fertile soils of the delta have been subject to intensive human use for centuries, and the ecoregion has been mostly converted to intensive agriculture, with few enclaves of forest remaining.[27] Additionally, the Sundarbans serves a crucial function as a protective barrier for millions of inhabitants against floods that result from cyclones.
Four protected areas in the Sundarbans are listed as UNESCO World Heritage Sites.[28] Despite these protections, the Indian Sundarbans were assessed as endangered in 2020 under the IUCN Red List of Ecosystems framework.[29] There is a consistent pattern of depleted biodiversity or loss of species and the ecological quality of the forest is declining.[30]
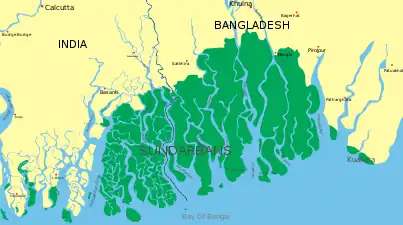 Map of the Sundarbans
Map of the Sundarbans Bengal tiger in the Sunderbans
Bengal tiger in the Sunderbans Channel in low tide
Channel in low tide
Ecosystem
The unique ecosystem found in the intricate mesh of mangrove roots offers a quiet marine habitat for young organisms.[31] In areas where roots are permanently submerged, the organisms they host include algae, barnacles, oysters, sponges, and bryozoa, which all require a hard surface for anchoring while they filter-feed. Shrimp and mud lobsters use the muddy bottoms as their home.[32] Mangrove crabs eat the mangrove leaves, adding nutrients to the mangal mud for other bottom feeders.[33] In at least some cases, the export of carbon fixed in mangroves is important in coastal food webs.[34] Mangrove plantations host several commercially important species of fish and crustaceans.[35]
In Puerto Rico, the red, white, and black mangroves occupy different ecological niches and have slightly different chemical compositions, so the carbon content varies between the species, as well between the different tissues of the plant (e.g., leaf matter versus roots).[36] There is a clear succession of these three trees from the lower elevations, which are dominated by red mangroves, to farther inland with a higher concentration of white mangroves.[36]
Mangrove forests are an important part of the cycling and storage of carbon in tropical coastal ecosystems.[36] Knowing this, scientists seek to reconstruct the environment and investigate changes to the coastal ecosystem over thousands of years using sediment cores.[37] However, an additional complication is the imported marine organic matter that also gets deposited in the sediment through the tidal flushing of mangrove forests.[36]
Mangrove forests can decay into peat deposits because of fungal and bacterial processes as well as by the action of termites.[36] It becomes peat in good geochemical, sedimentary, and tectonic conditions.[36] The nature of these deposits depends on the environment and the types of mangroves involved. Termites process fallen leaf litter, root systems and wood from mangroves into peat to build their nests.[36] Termites stabilise the chemistry of this peat and represent approximately 2% of above ground carbon storage in mangroves.[36] As the nests are buried over time this carbon is stored in the sediment, and the carbon cycle continues.[36]
Mangroves are an important source of blue carbon. Globally, mangroves stored 4.19 Gt (9.2×1012 lb) of carbon in 2012.[38] Two percent of global mangrove carbon was lost between 2000 and 2012, equivalent to a maximum potential of 0.316996250 Gt (6.9885710×1011 lb) of CO2 emissions.[38] Globally, mangroves have been shown to provide measurable economic protections to coastal communities affected by tropical storms.[39]
Biodiversity
Birds
Heterogeneity in landscape ecology is a measure of how different parts of a landscape are from one another. It can manifest in an ecosystem from the abiotic or biotic characteristics of the environment. For example, coastal mangrove forests are located at the land-sea interface, so their functioning is influenced by abiotic factors such as tides, as well as biotic factors such as the extent and configuration of adjacent vegetation.[40] For forest birds, tidal inundation means that the availability of many mangrove resources fluctuates daily, suggesting foraging flexibility is likely to be important. Mangroves also offer estuarine prey items, such as mudskippers and crabs, that are not found in terrestrial forest types. Further, mangroves are often situated in a complex mosaic of adjacent vegetation types such as grasslands, saltmarshes, and woodlands, and this can mean that flexibility in foraging strategy and choice of foraging habitat may be advantageous for highly mobile forest birds.[40] Relative to other forest types, mangroves support few bird species that are obligate habitat (mangrove) specialists and instead host many species with generalised foraging niches.[41][40]
 Mangrove forests host many bird species with generalised foraging niches
Mangrove forests host many bird species with generalised foraging niches

_-_Flickr_-_Lip_Kee.jpg.webp) Mangrove kingfishers are found particularly in mangrove zones
Mangrove kingfishers are found particularly in mangrove zones.jpg.webp)
 Brown pelicans fish and nest in mangrove forests
Brown pelicans fish and nest in mangrove forests Little blue heron. The water is reflecting green mangrove trees.
Little blue heron. The water is reflecting green mangrove trees. Three great egrets fishing along a mangrove shore
Three great egrets fishing along a mangrove shore Pelicans and cormorants high in the mangrove trees
Pelicans and cormorants high in the mangrove trees
- Bird sanctuaries
Mangrove forests are home and sanctuaries for many of aquatic bird species, including:
Fish
The intricate root system of mangrove forests makes them attractive to adult fish seeking food and juvenile fish seeking shelter.[3]

 Mangrove snapper swimming among mangrove roots
Mangrove snapper swimming among mangrove roots_(6055617687).jpg.webp) Old World silversides schooling among mangrove roots
Old World silversides schooling among mangrove roots Barracuda lurks among mangrove root
Barracuda lurks among mangrove root Yellow seahorses breed and give birth in Asia's flooded mangrove forests. Tangled roots and submerged branches offer shadowy shelter to pregnant dads and their offspring
Yellow seahorses breed and give birth in Asia's flooded mangrove forests. Tangled roots and submerged branches offer shadowy shelter to pregnant dads and their offspring.jpg.webp) Mudskippers can be found in mangrove swamps
Mudskippers can be found in mangrove swamps Mangrove whipray
Mangrove whipray
Mangrove crab holobiont
Mangrove forests are among the more productive and diverse ecosystems on the planet, despite limited nitrogen availability. Under such conditions, animal-microbe associations (holobionts) are often key to ecosystem functioning. An example is the role of fiddler crabs and their carapace-associated microbial biofilm as hotspots of microbial nitrogen transformations and sources of nitrogen within the mangrove ecosystem.[42]
Among coastal ecosystems, mangrove forests are of great importance as they account for three quarters of the tropical coastline and provide different ecosystem services.[43][44] Mangrove ecosystems generally act as a net sink of carbon, although they release organic matter to the sea in the form of dissolved refractory macromolecules, leaves, branches and other debris.[45][46] In pristine environments, mangroves are among the most productive ecosystems on the planet, despite growing in tropical waters that are often nutrient depleted.[47] The refractory nature of the organic matter produced and retained in mangroves can slow the recycling of nutrients, particularly of nitrogen.[45][48] Nitrogen limitation in such systems may be overcome by microbial nitrogen fixation when combined with high rates of bioturbation by macrofauna, such as crabs and lobsters.[49][50][42]
Bioturbation by macrofauna affect nitrogen availability and multiple nitrogen related microbial processes through sediment reworking, burrow construction and bioirrigation, feeding and excretion.[51] Macrofauna mix old and fresh organic matter, extend oxic–anoxic sediment interfaces, increase the availability of energy-yielding electron acceptors and increase nitrogen turnover via direct excretion.[52][53] Thus, macrofauna may alleviate nitrogen limitation by priming the remineralisation of refractory nitrogen (that is, the nitrogen that can't be biologically decomposed), reducing plant-microbe competition.[54][55] Such activity ultimately promotes nitrogen recycling, plant assimilation and high nitrogen retention, as well as favours its loss by stimulating coupled nitrification and denitrification.[56][42]

Dry weight of crab biofilm and mean dry weight of incubated crab expressed as µmol nitrogen per crab per day

Mangrove sediments are highly bioturbated by decapods such as crabs.[59] Crab populations continuously rework sediment by constructing burrows, creating new niches, transporting or selectively grazing on sediment microbial communities.[59][60][61][62] In addition, crabs can affect organic matter turnover by assimilating leaves and producing finely fragmented faeces, or by carrying them into their burrows.[63][64] Therefore, crabs are considered important ecosystem engineers shaping biogeochemical processes in intertidal muddy banks of mangroves.[65][66] In contrast to burrowing polychaetes or amphipods, the abundant Ocipodid crabs, mainly represented by fiddler crabs, do not permanently ventilate their burrows. These crabs may temporarily leave their burrows for surface activities,[62] or otherwise plug their burrow entrance during tidal inundation in order to trap air.[67] A recent study showed that these crabs can be associated with a diverse microbial community, either on their carapace or in their gut.[61][42]
The exoskeleton of living animals, such as shells or carapaces, offers a habitat for microbial biofilms which are actively involved in different N-cycling pathways such as nitrification, denitrification and dissimilatory nitrate reduction to ammonium (DNRA).[68][69][70][71][72][73] Colonizing the carapace of crabs may be advantageous for specific bacteria, because of host activities such as respiration, excretion, feeding and horizontal and vertical migrations.[74] However, the ecological interactions between fiddler crabs and bacteria, their regulation and significance as well as their implications at scales spanning from the single individual to the ecosystem are not well understood.[61][75][42]

 Mangrove crab, possibly Neosarmatium asiaticum
Mangrove crab, possibly Neosarmatium asiaticum Mangrove tree crab, Aratus pisonii
Mangrove tree crab, Aratus pisonii
Biogeochemistry
Carbon cycle

B.) Isotopic profile of sediments across the transition from mangrove to intertidal mudflats and seagrass beds, illustrating the retention of mangrove productivity within the forest.
Mangrove forests are amongst the world's most productive marine ecosystems,[79] with net primary productivity (NPP) in the order of 208 Tg C yr−1.[78] Mangrove forests achieve a steady state once the forest reaches maximum biomass at around 20–30 years through a constant process of mortality and renewal [80] so, assuming the living biomass is not becoming more carbon dense, then carbon has to be lost at a rate equal to the amount of carbon fixed as NPP. Hence this productivity is either retained within the mangrove forest, as a standing stock of live material such as wood, buried in sediments, or exported to neighbouring habitats as litter, particulate and dissolved organic carbon (POC and DOC) and dissolved inorganic carbon (DIC), or lost to the atmosphere.[78][81][79][77]
The out-welling hypothesis argues that export of locally derived POC and DOC is an important ecosystem function of mangroves, which drives detrital based food webs in adjacent coastal habitats.[82][83] Export of mangrove carbon has been estimated to make a significant trophic contribution to adjacent ecosystems.[84][85][86][87] The theory of outwelling is supported by mass balance evaluations that show the amount of carbon fixed by mangroves normally greatly exceeds the amount stored within the forest,[79][88] although the scale of outwelling varies considerably between forests,[89] due to differences in coastal geomorphology, tidal regimes, freshwater flow and productivity.[90][91][77]

In the 1990s, global estimates could account for 48% of the total global mangrove primary production of 218 ± 72 million tons C yr−1 (see diagram on the right). By incorporating information on carbon burial, CO2 efflux and carbon outwelled as leaf litter, POC and DOC, the remaining 52% was thought outwelled as DIC, though there was insufficient data to confirm this.[78] More recent assessments of DIC export at two sites in Australia [81][94] supported the estimates of Bouillon et al. in 2008,[78] although in 2014 Alongi suggested that only 40% of NPP was exported as DIC.[79][77]
Nitrogen assimilation
Mangrove forests and coastal marshes are typically considered N-limited ecosystems because of their high primary production.[95][96] Therefore, mangrove plants are highly efficient at utilising soil nitrogen, making them an important sink for excess nitrogen from upstream.[97][47] However, different mangrove species may still utilise nitrogen at different efficiencies,[98] even though they share similar nitrogen pathways (see diagram on right). Reported nitrogen assimilation rates in mangrove plants ranged from 2 to 8 μmol g−1 h−1 under ambient nitrogen conditions,[99] and 19 to 251 μmol g−1 h−1 when the nitrogen supply was unlimited.[100][93]
In addition to species variation, different environmental conditions can also affect the nitrogen assimilation rates in mangrove plants. Because Cl− ions can reduce protein synthesis and nitrogen assimilation,[101] soil pore water salinity appears to be a negative factor that significantly alters the nitrogen uptake rates of mangrove plants.[100][102][93]
Exploitation and conservation


Adequate data is only available for about half of the global area of mangroves. However, of those areas for which data has been collected, it appears that 35% of the mangroves have been destroyed.[103] Since the 1980s, around 2% of mangrove area is estimated to be lost each year.[104] Assessments of global variation in mangrove loss indicates that national regulatory quality mediates how different drivers and pressures influence loss rates.[105]
Shrimp farming causes approximately a quarter of the destruction of mangrove forests.[106][107] Likewise, the 2010 update of the World Mangrove Atlas indicated that approximately one fifth of the world's mangrove ecosystems have been lost since 1980,[108] although this rapid loss rate appears to have decreased since 2000 with global losses estimated at between 0.16% and 0.39% annually between 2000 and 2012.[109] Despite global loss rates decreasing since 2000, Southeast Asia remains an area of concern with loss rates between 3.6% and 8.1% between 2000 and 2012.[109] By far the most damaging form of shrimp farming is when a closed ponds system (non-integrated multi-trophic aquaculture) is used, as these require destruction of a large part of the mangrove, and use antibiotics and disinfectants to suppress diseases that occur in this system, and which may also leak into the surrounding environment. Far less damage occurs when integrated mangrove-shrimp aquaculture is used, as this is connected to the sea and subjected to the tides, and less diseases occur, and as far less mangrove is destroyed for it.[110]
Grassroots efforts to protect mangroves from development and from citizens cutting down the mangroves for charcoal production,[111][112] cooking, heating and as a building material are becoming more popular. Solar cookers are distributed by many non-government organizations as a low-cost alternative to wood and charcoal stoves. These may help in reducing the demand for charcoal.
- In Thailand, community management has been effective in restoring damaged mangroves.[113] Also, production of mangrove honey is practiced, as a way to generate sustainable income for nearby people, keeping them from destroying the mangrove and generate a short-term revenue.[114][115]
- In Madagascar, honey is also produced in mangroves as a source of (non-destructive) income generation. In addition, silk pods from endemic silkworm species are also collected in the Madagascar mangroves for wild silk production.[116][112]
- In the Bahamas, for example, active efforts to save mangroves are occurring on the islands of Bimini and Great Guana Cay.
- In Trinidad and Tobago as well, efforts are underway to protect a mangrove threatened by the construction of a steel mill and a port.
- Within northern Ecuador, mangrove regrowth is reported in almost all estuaries and stems primarily from local actors responding to earlier periods of deforestation in the Esmeraldas region.[117]
Mangroves have been reported to be able to help buffer against tsunami, cyclones, and other storms, and as such may be considered a flagship system for ecosystem-based adaptation to the impacts of climate change. One village in Tamil Nadu was protected from tsunami destruction—the villagers in Naluvedapathy planted 80,244 saplings to get into the Guinness Book of World Records. This created a kilometre-wide belt of trees of various varieties. When the 2004 tsunami struck, much of the land around the village was flooded, but the village suffered minimal damage.[118]

Ocean deoxygenation
Compared to seagrass meadows and coral reefs, hypoxia is more common on a regular basis in mangrove ecosystems, through ocean deoxygenation is compounding the negative effects by anthropogenic nutrient inputs and land use modification.[119]
Like seagrass, mangrove trees transport oxygen to roots of rhizomes, reduce sulfide concentrations, and alter microbial communities. Dissolved oxygen is more readily consumed in the interior of the mangrove forest. Anthropogenic inputs may push the limits of survival in many mangrove microhabitats. For example, shrimp ponds constructed in mangrove forests are considered the greatest anthropogenic threat to mangrove ecosystems. These shrimp ponds reduce estuary circulation and water quality which leads to the promotion of diel-cycling hypoxia. When the quality of the water degrades, the shrimp ponds are quickly abandoned leaving massive amounts of wastewater. This is a major source of water pollution that promotes ocean deoxygenation in the adjacent habitats.[119][120]
Due to these frequent hypoxic conditions, the water does not provide habitats to fish. When exposed to extreme hypoxia, ecosystem function can completely collapse. Extreme deoxygenation will affect the local fish populations, which are an essential food source. The environmental costs of shrimp farms in the mangrove forests grossly outweigh the economic benefits of them. Cessation of shrimp production and restoration of these areas reduce eutrophication and anthropogenic hypoxia.[119]
Reforestation


In some areas, mangrove reforestation and mangrove restoration is also underway. Red mangroves are the most common choice for cultivation, used particularly in marine aquariums in a sump to reduce nitrates and other nutrients in the water. Mangroves also appear in home aquariums, and as ornamental plants, such as in Japan.
| External video | |
|---|---|
– Mangrove Action Project |
The Manzanar Mangrove Initiative is an ongoing experiment in Arkiko, Eritrea, part of the Manzanar Project founded by Gordon H. Sato, establishing new mangrove plantations on the coastal mudflats. Initial plantings failed, but observation of the areas where mangroves did survive by themselves led to the conclusion that nutrients in water flow from inland were important to the health of the mangroves. Trials with the Eritrean Ministry of Fisheries followed, and a planting system was designed to provide the nitrogen, phosphorus, and iron missing from seawater.[121][122]
The propagules are planted inside a reused galvanized steel can with the bottom knocked out; a small piece of iron and a pierced plastic bag with fertilizer containing nitrogen and phosphorus are buried with the propagule. As of 2007, after six years of planting, 700,000 mangroves are growing; providing stock feed for sheep and habitat for oysters, crabs, other bivalves, and fish.[121][122]
Another method of restoring mangroves is by using quadcopters (which are able to carry and deposit seed pods). According to Irina Fedorenko, an amount of work equivalent to weeks of planting using traditional methods can be done by a drone in days, and at a fraction of the cost.[123]
Seventy percent of mangrove forests have been lost in Java, Indonesia. Mangroves formerly protected the island's coastal land from flooding and erosion.[124] Wetlands International, an NGC based in the Netherlands, in collaboration with nine villages in Demak where lands and homes had been flooded, began reviving mangrove forests in Java. Wetlands International introduced the idea of developing tropical versions of techniques traditionally used by the Dutch to catch sediment in North Sea coastal salt marshes.[124] Originally, the villagers constructed a sea barrier by hammering two rows of vertical bamboo poles into the seabed and filling the gaps with brushwood held in place with netting. Later the bamboo was replaced by PVC pipes filled with concrete. As sediment gets deposited around the brushwood, it serves to catch floating mangrove seeds and provide them with a stable base to germinate, take root and regrow. This creates a green belt of protection around the islands. As the mangroves mature, more sediment is held in the catchment area; the process is repeated until a mangrove forest has been restored. Eventually the protective structures will not be needed.[124] By late 2018, 16 km (9.9 mi) of brushwood barriers along the coastline had been completed.[124]
A concern over reforestation is that although it supports increases in mangrove area it may actually result in a decrease in global mangrove functionality and poor restoration processes may result in longer term depletion of the mangrove resource.[125]
National and international studies
In terms of local and national studies of mangrove loss, the case of Belize's mangroves is illustrative in its contrast to the global picture. A recent, satellite-based study[126]—funded by the World Wildlife Fund and conducted by the Water Center for the Humid Tropics of Latin America and the Caribbean (CATHALAC)—indicates Belize's mangrove cover declined by a mere 2% over a 30-year period. The study was born out of the need to verify the popular conception that mangrove clearing in Belize was rampant.[127]
Instead, the assessment showed, between 1980 and 2010, under 16 km2 (6.2 sq mi) of mangroves had been cleared, although clearing of mangroves near Belize's main coastal settlements (e.g. Belize City and San Pedro) was relatively high. The rate of loss of Belize's mangroves—at 0.07% per year between 1980 and 2010—was much lower than Belize's overall rate of forest clearing (0.6% per year in the same period).[128] These findings can also be interpreted to indicate Belize's mangrove regulations (under the nation's)[129] have largely been effective. Nevertheless, the need to protect Belize's mangroves is imperative, as a 2009 study by the World Resources Institute (WRI) indicates the ecosystems contribute US$174 to US$249 million per year to Belize's national economy.[130]
| External video | |
|---|---|
In May 2019, ORNL DAAC News announced that NASA's Carbon Monitoring System (CMS), using new satellite-based maps of global mangrove forests across 116 countries, had created a new dataset to characterize the "distribution, biomass, and canopy height of mangrove-forested wetlands".[131][132] Mangrove forests move carbon dioxide "from the atmosphere into long-term storage" in greater quantities than other forests, making them "among the planet's best carbon scrubbers" according to a NASA-led study.[132][133]
See also
References
- Luo, Ling; Gu, Ji-Dong (2018). "Nutrient limitation status in a subtropical mangrove ecosystem revealed by analysis of enzymatic stoichiometry and microbial abundance for sediment carbon cycling". International Biodeterioration & Biodegradation. 128: 3–10. doi:10.1016/j.ibiod.2016.04.023.
- Tue, Nguyen Tai; Ngoc, Nguyen Thi; Quy, Tran Dang; Hamaoka, Hideki; Nhuan, Mai Trong; Omori, Koji (2012). "A cross-system analysis of sedimentary organic carbon in the mangrove ecosystems of Xuan Thuy National Park, Vietnam". Journal of Sea Research. 67 (1): 69–76. Bibcode:2012JSR....67...69T. doi:10.1016/j.seares.2011.10.006.
- What is a mangrove forest? National Ocean Service, NOAA. Updated: 25 March 2021. Retrieved: 4 October 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - De Groot, Rudolf S. (1992). Functions of Nature: Evaluation of Nature in Environmental Planning, Management and Decision Making. Wolters-Noordhoff. ISBN 9789001355944.
- Alongi, D. M.; Murdiyarso, D.; Fourqurean, J. W.; Kauffman, J. B.; Hutahaean, A.; Crooks, S.; Lovelock, C. E.; Howard, J.; Herr, D.; Fortes, M.; Pidgeon, E.; Wagey, T. (2016). "Indonesia's blue carbon: A globally significant and vulnerable sink for seagrass and mangrove carbon". Wetlands Ecology and Management. 24: 3–13. doi:10.1007/s11273-015-9446-y. S2CID 4983675.
- Qu, Wu; Gao, Boliang; Wu, Jie; Jin, Min; Wang, Jianxin; Zeng, Runying (2020). "High-throughput amplicon sequencing reveals the spatiotemporal effects, abiotic and biotic shaping factors for the microbial communities in tropical mangrove sediments in Sanya, China". doi:10.21203/rs.3.rs-34433/v1. S2CID 234724143.
{{cite journal}}: Cite journal requires|journal=(help) Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License. - Lin, Xiaolan; Hetharua, Buce; Lin, Lian; Xu, Hong; Zheng, Tianling; He, Zhili; Tian, Yun (4 October 2018). "Mangrove Sediment Microbiome: Adaptive Microbial Assemblages and Their Routed Biogeochemical Processes in Yunxiao Mangrove National Nature Reserve, China". Microbial Ecology. Springer Science and Business Media LLC. 78 (1): 57–69. doi:10.1007/s00248-018-1261-6. ISSN 0095-3628. PMID 30284602. S2CID 52917236.
- Komiyama, Akira; Ong, Jin Eong; Poungparn, Sasitorn (2008-08-01). "Allometry, biomass, and productivity of mangrove forests: A review". Aquatic Botany. Mangrove Ecology – Applications in Forestry and Costal Zone Management. 89 (2): 128–137. doi:10.1016/j.aquabot.2007.12.006. ISSN 0304-3770.
- Mangroves are trees and shrubs that have adapted to life in a saltwater environment National Marine Sanctuaries, NOAA. Retrieved 18 November 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - Friess, D. A.; Rogers, K.; Lovelock, C. E.; Krauss, K. W.; Hamilton, S. E.; Lee, S. Y.; Lucas, R.; Primavera, J.; Rajkaran, A.; Shi, S. (2019). "The State of the World's Mangrove Forests: Past, Present, and Future". Annual Review of Environment and Resources. 44 (1): 89–115. doi:10.1146/annurev-environ-101718-033302.
- Zimmer, Katarina (22 July 2021). "Many mangrove restorations fail. Is there a better way?". Knowable Magazine. doi:10.1146/knowable-072221-1. Retrieved 11 August 2021.
- Sievers, Michael; Brown, Christopher J.; Tulloch, Vivitskaia J.D.; Pearson, Ryan M.; Haig, Jodie A.; Turschwell, Mischa P.; Connolly, Rod M. (2019). "The Role of Vegetated Coastal Wetlands for Marine Megafauna Conservation". Trends in Ecology & Evolution. 34 (9): 807–817. doi:10.1016/j.tree.2019.04.004. hdl:10072/391960. ISSN 0169-5347. PMID 31126633. S2CID 164219103.
- Giraldes, Bruno Welter; Goodwin, Claire; Al-Fardi, Noora A. A.; Engmann, Amanda; Leitão, Alexandra; Ahmed, Asma A.; Ahmed, Kamelia O.; Abdulkader, Hadil A.; Al-Korbi, Halah A.; Al Easa, Hala Sultan Saif; Ahmed Eltai, Nahla O.; Hanifi-Moghaddam, Pejman (13 May 2020). Bianchi, Carlo Nike (ed.). "Two new sponge species (Demospongiae: Chalinidae and Suberitidae) isolated from hyperarid mangroves of Qatar with notes on their potential antibacterial bioactivity". PLOS ONE. Public Library of Science (PLoS). 15 (5): e0232205. Bibcode:2020PLoSO..1532205G. doi:10.1371/journal.pone.0232205. ISSN 1932-6203. PMC 7219822. PMID 32401792.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License. - Cannicci, Stefano; Fusi, Marco; Cimó, Filippo; Dahdouh-Guebas, Farid; Fratini, Sara (December 2018). "Interference competition as a key determinant for spatial distribution of mangrove crabs". BMC Ecology. 18 (1): 8. doi:10.1186/s12898-018-0164-1. PMC 5815208. PMID 29448932.
- Saenger, P.; McConchie, D. (2004). "Heavy metals in mangroves: methodology, monitoring and management". Envis Forest Bulletin. 4: 52–62. CiteSeerX 10.1.1.961.9649.
- Mazda, Y.; Kobashi, D.; Okada, S. (2005). "Tidal-Scale Hydrodynamics within Mangrove Swamps". Wetlands Ecology and Management. 13 (6): 647–655. CiteSeerX 10.1.1.522.5345. doi:10.1007/s11273-005-0613-4. S2CID 35322400.
- Danielsen, F.; Sørensen, M. K.; Olwig, M. F; Selvam, V; Parish, F; Burgess, N. D; Hiraishi, T.; Karunagaran, V. M.; Rasmussen, M. S.; Hansen, L. B.; Quarto, A.; Suryadiputra, N. (2005). "The Asian Tsunami: A Protective Role for Coastal Vegetation". Science. 310 (5748): 643. doi:10.1126/science.1118387. PMID 16254180. S2CID 31945341.
- Takagi, H.; Mikami, T.; Fujii, D.; Esteban, M.; Kurobe, S. (2016). "Mangrove forest against dyke-break-induced tsunami on rapidly subsiding coasts". Natural Hazards and Earth System Sciences. 16 (7): 1629–1638. Bibcode:2016NHESS..16.1629T. doi:10.5194/nhess-16-1629-2016.
- Dahdouh-Guebas, F.; Jayatissa, L. P.; Di Nitto, D.; Bosire, J. O.; Lo Seen, D.; Koedam, N. (2005). "How effective were mangroves as a defence against the recent tsunami?". Current Biology. 15 (12): R443–447. doi:10.1016/j.cub.2005.06.008. PMID 15964259. S2CID 8772526.
- Massel, S. R.; Furukawa, K.; Brinkman, R. M. (1999). "Surface wave propagation in mangrove forests". Fluid Dynamics Research. 24 (4): 219. Bibcode:1999FlDyR..24..219M. doi:10.1016/s0169-5983(98)00024-0. S2CID 122572658.
- Mazda, Y.; Wolanski, E.; King, B.; Sase, A.; Ohtsuka, D.; Magi, M. (1997). "Drag force due to vegetation in mangrove swamps". Mangroves and Salt Marshes. 1 (3): 193. doi:10.1023/A:1009949411068. S2CID 126945589.
- Giri, C.; Ochieng, E.; Tieszen, L. L.; Zhu, Z.; Singh, A.; Loveland, T.; Masek, J.; Duke, N. (January 2011). "Status and distribution of mangrove forests of the world using earth observation satellite data: Status and distributions of global mangroves". Global Ecology and Biogeography. 20 (1): 154–159. doi:10.1111/j.1466-8238.2010.00584.x. Retrieved 12 August 2021.
- Worthington, Thomas A.; zu Ermgassen, Philine S. E.; Friess, Daniel A.; Krauss, Ken W.; Lovelock, Catherine E.; Thorley, Julia; Tingey, Rick; Woodroffe, Colin D.; Bunting, Pete; Cormier, Nicole; Lagomasino, David; Lucas, Richard; Murray, Nicholas J.; Sutherland, William J.; Spalding, Mark (4 September 2020). "A global biophysical typology of mangroves and its relevance for ecosystem structure and deforestation". Scientific Reports. Springer Science and Business Media LLC. 10 (1): 14652. Bibcode:2020NatSR..1014652W. doi:10.1038/s41598-020-71194-5. ISSN 2045-2322. PMC 7473852. PMID 32887898.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License. - What's a Mangrove? And How Does It Work? American Museum of Natural History. Accessed 8 November 2021.
- Bunting, Pete; Rosenqvist, Ake; Lucas, Richard; Rebelo, Lisa-Maria; Hilarides, Lammert; Thomas, Nathan; Hardy, Andy; Itoh, Takuya; Shimada, Masanobu; Finlayson, C. (22 October 2018). "The Global Mangrove Watch—A New 2010 Global Baseline of Mangrove Extent". Remote Sensing. 10 (10): 1669. Bibcode:2018RemS...10.1669B. doi:10.3390/rs10101669.
- Pani, D. R.; Sarangi, S. K.; Subudhi, H. N.; Misra, R. C.; Bhandari, D. C. (2013). "Exploration, evaluation and conservation of salt tolerant rice genetic resources from Sundarbans region of West Bengal" (PDF). Journal of the Indian Society of Coastal Agricultural Research. 30 (1): 45–53.
- Iftekhar, M. S.; Islam, M. R. (2004). "Managing mangroves in Bangladesh: A strategy analysis" (PDF). Journal of Coastal Conservation. 10 (1): 139–146. doi:10.1652/1400-0350(2004)010[0139:MMIBAS]2.0.CO;2. S2CID 130056584.
- Giri, C.; Pengra, B.; Zhu, Z.; Singh, A.; Tieszen, L. L. (2007). "Monitoring mangrove forest dynamics of the Sundarbans in Bangladesh and India using multi-temporal satellite data from 1973 to 2000". Estuarine, Coastal and Shelf Science. 73 (1–2): 91–100. Bibcode:2007ECSS...73...91G. doi:10.1016/j.ecss.2006.12.019.
- Sievers, M.; Chowdhury, M. R.; Adame, M. F.; Bhadury, P.; Bhargava, R.; Buelow, C.; Friess, D. A.; Ghosh, A.; Hayes, M. A.; McClure, E. C.; Pearson, R. M. (2020). "Indian Sundarbans mangrove forest considered endangered under Red List of Ecosystems, but there is cause for optimism" (PDF). Biological Conservation. 251: 108751. doi:10.1016/j.biocon.2020.108751. S2CID 222206165. Archived from the original (PDF) on 2021-10-15. Retrieved 2021-11-07.
- Manna, S.; Chaudhuri, K.; Bhattacharyya, S.; Bhattacharyya, M. (2010). "Dynamics of Sundarban estuarine ecosystem: Eutrophication induced threat to mangroves". Saline Systems. 6: 8. doi:10.1186/1746-1448-6-8. PMC 2928246. PMID 20699005.
- Bos, AR; Gumanao, GS; Van Katwijk, MM; Mueller, B; Saceda, MM; Tejada, RL (2010). "Ontogenetic habitat shift, population growth, and burrowing behavior of the Indo-Pacific beach star, Archaster typicus (Echinodermata; Asteroidea)". Marine Biology. 158 (3): 639–648. doi:10.1007/s00227-010-1588-0. PMC 3873073. PMID 24391259.
- Encarta Encyclopedia 2005. "Seashore", by Heidi Nepf.
- Skov, M. W.; Hartnoll, R. G. (2002). "Paradoxical selective feeding on a low-nutrient diet: Why do mangrove crabs eat leaves?". Oecologia. 131 (1): 1–7. Bibcode:2002Oecol.131....1S. doi:10.1007/s00442-001-0847-7. PMID 28547499. S2CID 23407273.
- Abrantes KG, Johnston R, Connolly RM, Sheaves M (2015-01-01). "Importance of Mangrove Carbon for Aquatic Food Webs in Wet–Dry Tropical Estuaries". Estuaries and Coasts. 38 (1): 383–99. doi:10.1007/s12237-014-9817-2. hdl:10072/141734. ISSN 1559-2731. S2CID 3957868.
- Gupta, S. K.; Goyal, Megh R. (2017-03-16). Soil Salinity Management in Agriculture: Technological Advances and Applications. CRC Press. ISBN 978-1-315-34177-4.
- Vane, CH; Kim, AW; Moss-Hayes, V; Snape, CE; Diaz, MC; Khan, NS; Engelhart, SE; Horton, BP (2013). "Degradation of mangrove tissues by arboreal termites (Nasutitermes acajutlae) and their role in the mangrove C cycle (Puerto Rico): Chemical characterization and organic matter provenance using bulk δ13C, C/N, alkaline CuO oxidation-GC/MS, and solid-state" (PDF). Geochemistry, Geophysics, Geosystems. 14 (8): 3176. Bibcode:2013GGG....14.3176V. doi:10.1002/ggge.20194.
- Versteegh, GJ; et al. (2004). "Taraxerol and Rhizophora pollen as proxies for tracking past mangrove ecosystems". Geochimica et Cosmochimica Acta. 68 (3): 411–22. Bibcode:2004GeCoA..68..411V. doi:10.1016/S0016-7037(03)00456-3.
- Hamilton, SE; Friess, DA (2018). "Global carbon stocks and potential emissions due to mangrove deforestation from 2000 to 2012". Nature Climate Change. 8 (3): 240–244. arXiv:1611.00307. Bibcode:2018NatCC...8..240H. doi:10.1038/s41558-018-0090-4. S2CID 89785740.
- Hochard, Jacob P.; Hamilton, S; Barbier, EB (2019-06-03). "Mangroves shelter coastal economic activity from cyclones". Proceedings of the National Academy of Sciences. 116 (25): 12232–37. Bibcode:2019PNAS..11612232H. doi:10.1073/pnas.1820067116. ISSN 0027-8424. PMC 6589649. PMID 31160457.
- Buelow, Christina A.; Reside, April E.; Baker, Ronald; Sheaves, Marcus (2018). "Stable isotopes reveal opportunistic foraging in a spatiotemporally heterogeneous environment: Bird assemblages in mangrove forests". PLOS ONE. 13 (11): e0206145. Bibcode:2018PLoSO..1306145B. doi:10.1371/journal.pone.0206145. PMC 6237324. PMID 30439959.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License. - Buelow, Christina A.; Reside, April E.; Baker, Ronald; Sheaves, Marcus (2018). "Stable isotopes reveal opportunistic foraging in a spatiotemporally heterogeneous environment: Bird assemblages in mangrove forests". PLOS ONE. 13 (11): e0206145. Bibcode:2018PLoSO..1306145B. doi:10.1371/journal.pone.0206145. PMC 6237324. PMID 30439959.
- Zilius, Mindaugas; Bonaglia, Stefano; Broman, Elias; Chiozzini, Vitor Gonsalez; Samuiloviene, Aurelija; Nascimento, Francisco J. A.; Cardini, Ulisse; Bartoli, Marco (2020). "N2 fixation dominates nitrogen cycling in a mangrove fiddler crab holobiont". Scientific Reports. 10 (1): 13966. doi:10.1038/s41598-020-70834-0. PMC 7435186. PMID 32811860.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License. - Lee, Shing Yip; Primavera, Jurgene H.; Dahdouh-Guebas, Farid; McKee, Karen; Bosire, Jared O.; Cannicci, Stefano; Diele, Karen; Fromard, Francois; Koedam, Nico; Marchand, Cyril; Mendelssohn, Irving; Mukherjee, Nibedita; Record, Sydne (2014). "Ecological role and services of tropical mangrove ecosystems: A reassessment". Global Ecology and Biogeography. 23 (7): 726–743. doi:10.1111/geb.12155. hdl:10862/2247. S2CID 52904699.
- Kathiresan, K.; Bingham, B.L. (2001). Biology of mangroves and mangrove Ecosystems. Advances in Marine Biology. Vol. 40. pp. 81–251. doi:10.1016/S0065-2881(01)40003-4. ISBN 9780120261406.
- Dittmar, Thorsten; Hertkorn, Norbert; Kattner, Gerhard; Lara, Rubén J. (2006). "Mangroves, a major source of dissolved organic carbon to the oceans". Global Biogeochemical Cycles. 20 (1): n/a. Bibcode:2006GBioC..20.1012D. doi:10.1029/2005GB002570. S2CID 128922131.
- Kristensen, Erik; Bouillon, Steven; Dittmar, Thorsten; Marchand, Cyril (2008). "Organic carbon dynamics in mangrove ecosystems: A review". Aquatic Botany. 89 (2): 201–219. doi:10.1016/j.aquabot.2007.12.005.
- Reef, R.; Feller, I. C.; Lovelock, C. E. (2010). "Nutrition of mangroves". Tree Physiology. 30 (9): 1148–1160. doi:10.1093/treephys/tpq048. PMID 20566581.
- Woolfe, Ken J.; Dale, Paul J.; Brunskill, Gregg J. (1995). "Sedimentary C/S relationships in a large tropical estuary: Evidence for refractory carbon inputs from mangroves". Geo-Marine Letters. 15 (3–4): 140–144. Bibcode:1995GML....15..140W. doi:10.1007/BF01204455. S2CID 128709551.
- Woitchik, A.F.; Ohowa, B.; Kazungu, J.M.; Rao, R.G.; Goeyens, L.; Dehairs, F. (1997). "Nitrogen enrichment during decomposition of mangrove leaf litter in an east African coastal lagoon (Kenya): Relative importance of biological nitrogen fixation". Biogeochemistry. 39: 15–35. doi:10.1023/A:1005850032254. S2CID 91314553.
- Zuberer, D. A.; Silver, W. S. (1978). "Biological Dinitrogen Fixation (Acetylene Reduction) Associated with Florida Mangroves". Applied and Environmental Microbiology. 35 (3): 567–575. Bibcode:1978ApEnM..35..567Z. doi:10.1128/aem.35.3.567-575.1978. PMC 242881. PMID 637550.
- Kristensen, E.; Penha-Lopes, G.; Delefosse, M.; Valdemarsen, T.; Quintana, CO; Banta, GT (2012). "What is bioturbation? The need for a precise definition for fauna in aquatic sciences". Marine Ecology Progress Series. 446: 285–302. Bibcode:2012MEPS..446..285K. doi:10.3354/meps09506.
- Welsh, David T. (2003). "It's a dirty job but someone has to do it: The role of marine benthic macrofauna in organic matter turnover and nutrient recycling to the water column". Chemistry and Ecology. 19 (5): 321–342. doi:10.1080/0275754031000155474. S2CID 94773926.
- Stief, P. (2013). "Stimulation of microbial nitrogen cycling in aquatic ecosystems by benthic macrofauna: Mechanisms and environmental implications". Biogeosciences. 10 (12): 7829–7846. Bibcode:2013BGeo...10.7829S. doi:10.5194/bg-10-7829-2013.
- Gilbertson, William W.; Solan, Martin; Prosser, James I. (2012). "Differential effects of microorganism-invertebrate interactions on benthic nitrogen cycling". FEMS Microbiology Ecology. 82 (1): 11–22. doi:10.1111/j.1574-6941.2012.01400.x. PMID 22533682.
- Laverock, Bonnie; Gilbert, Jack A.; Tait, Karen; Osborn, A. Mark; Widdicombe, Steve (2011). "Bioturbation: Impact on the marine nitrogen cycle". Biochemical Society Transactions. 39 (1): 315–320. doi:10.1042/BST0390315. PMID 21265795.
- Magri, M.; Benelli, S.; Bondavalli, C.; Bartoli, M.; Christian, R. R.; Bodini, A. (2018). "Benthic N pathways in illuminated and bioturbated sediments studied with network analysis". Limnology and Oceanography. 63 (S1): S68–S84. Bibcode:2018LimOc..63S..68M. doi:10.1002/lno.10724. S2CID 89783098.
- Kelvin K. P. Lim, Dennis H. Murphy, T. Morgany, N. Sivasothi, Peter K. L. Ng, B. C. Soong, Hugh T. W. Tan, K. S. Tan & T. K. Tan (1999). "Mud lobster, Thalassina anomala". In Peter K. L. Ng; N. Sivasothi (eds.). A Guide to the Mangroves of Singapore. Singapore Science Centre. ISBN 981-04-1308-4.
{{cite book}}: CS1 maint: multiple names: authors list (link) - Ria Tan (2001). "Mud Lobster Thalassina anomala". Archived from the original on 2007-08-27.
- Kristensen, Erik (2008). "Mangrove crabs as ecosystem engineers; with emphasis on sediment processes". Journal of Sea Research. 59 (1–2): 30–43. Bibcode:2008JSR....59...30K. doi:10.1016/j.seares.2007.05.004.
- Booth, Jenny Marie; Fusi, Marco; Marasco, Ramona; Mbobo, Tumeka; Daffonchio, Daniele (2019). "Fiddler crab bioturbation determines consistent changes in bacterial communities across contrasting environmental conditions". Scientific Reports. 9 (1): 3749. Bibcode:2019NatSR...9.3749B. doi:10.1038/s41598-019-40315-0. PMC 6403291. PMID 30842580.
- Cuellar-Gempeler, Catalina; Leibold, Mathew A. (2018). "Multiple colonist pools shape fiddler crab-associated bacterial communities". The ISME Journal. 12 (3): 825–837. doi:10.1038/s41396-017-0014-8. PMC 5864236. PMID 29362507.
- Reinsel, K.A. (2004). "Impact of fiddler crab foraging and tidal inundation on an intertidal sandflat: Season-dependent effects in one tidal cycle". Journal of Experimental Marine Biology and Ecology. 313: 1–17. doi:10.1016/j.jembe.2004.06.003.
- Nordhaus, Inga; Diele, Karen; Wolff, Matthias (2009). "Activity patterns, feeding and burrowing behaviour of the crab Ucides cordatus (Ucididae) in a high intertidal mangrove forest in North Brazil". Journal of Experimental Marine Biology and Ecology. 374 (2): 104–112. doi:10.1016/j.jembe.2009.04.002.
- Nordhaus, Inga; Wolff, Matthias (2007). "Feeding ecology of the mangrove crab Ucides cordatus (Ocypodidae): Food choice, food quality and assimilation efficiency". Marine Biology. 151 (5): 1665–1681. doi:10.1007/s00227-006-0597-5. S2CID 88582703.
- Fanjul, Eugenia; Bazterrica, María C.; Escapa, Mauricio; Grela, María A.; Iribarne, Oscar (2011). "Impact of crab bioturbation on benthic flux and nitrogen dynamics of Southwest Atlantic intertidal marshes and mudflats". Estuarine, Coastal and Shelf Science. 92 (4): 629–638. Bibcode:2011ECSS...92..629F. doi:10.1016/j.ecss.2011.03.002.
- Quintana, Cintia O.; Shimabukuro, Maurício; Pereira, Camila O.; Alves, Betina G. R.; Moraes, Paula C.; Valdemarsen, Thomas; Kristensen, Erik; Sumida, Paulo Y. G. (2015). "Carbon mineralization pathways and bioturbation in coastal Brazilian sediments". Scientific Reports. 5: 16122. Bibcode:2015NatSR...516122Q. doi:10.1038/srep16122. PMC 4630785. PMID 26525137.
- de la Iglesia, Horacio O.; Rodríguez, Enrique M.; Dezi, Rubén E. (1994). "Burrow plugging in the crab Uca uruguayensis and its synchronization with photoperiod and tides". Physiology & Behavior. 55 (5): 913–919. doi:10.1016/0031-9384(94)90079-5. PMID 8022913. S2CID 7366251.
- Arfken, Ann; Song, Bongkeun; Bowman, Jeff S.; Piehler, Michael (2017). "Denitrification potential of the eastern oyster microbiome using a 16S rRNA gene based metabolic inference approach". PLOS ONE. 12 (9): e0185071. Bibcode:2017PLoSO..1285071A. doi:10.1371/journal.pone.0185071. PMC 5608302. PMID 28934286.
- Caffrey, Jane M.; Hollibaugh, James T.; Mortazavi, Behzad (2016). "Living oysters and their shells as sites of nitrification and denitrification". Marine Pollution Bulletin. 112 (1–2): 86–90. Bibcode:2016MarPB.112...86C. doi:10.1016/j.marpolbul.2016.08.038. PMID 27567196.
- Glud, Ronnie N.; Grossart, Hans‐Peter; Larsen, Morten; Tang, Kam W.; Arendt, Kristine E.; Rysgaard, Søren; Thamdrup, Bo; Gissel Nielsen, Torkel (2015). "Copepod carcasses as microbial hot spots for pelagic denitrification". Limnology and Oceanography. 60 (6): 2026–2036. Bibcode:2015LimOc..60.2026G. doi:10.1002/lno.10149. S2CID 45429253.
- Heisterkamp, Ines M.; Schramm, Andreas; Larsen, Lone H.; Svenningsen, Nanna B.; Lavik, Gaute; De Beer, Dirk; Stief, Peter (2013). "Shell biofilm-associated nitrous oxide production in marine molluscs: Processes, precursors and relative importance". Environmental Microbiology. 15 (7): 1943–1955. doi:10.1111/j.1462-2920.2012.02823.x. PMID 22830624.
- Ray, NE; Henning, MC; Fulweiler, RW (2019). "Nitrogen and phosphorus cycling in the digestive system and shell biofilm of the eastern oyster Crassostrea virginica". Marine Ecology Progress Series. 621: 95–105. Bibcode:2019MEPS..621...95R. doi:10.3354/meps13007. S2CID 198261071.
- Stief, Peter; Lundgaard, Ann Sofie Birch; Treusch, Alexander H.; Thamdrup, Bo; Grossart, Hans-Peter; Glud, Ronnie N. (2018). "Freshwater copepod carcasses as pelagic microsites of dissimilatory nitrate reduction to ammonium". FEMS Microbiology Ecology. 94 (10). doi:10.1093/femsec/fiy144. PMC 6084575. PMID 30060193.
- Wahl, Martin; Goecke, Franz; Labes, Antje; Dobretsov, Sergey; Weinberger, Florian (2012). "The Second Skin: Ecological Role of Epibiotic Biofilms on Marine Organisms". Frontiers in Microbiology. 3: 292. doi:10.3389/fmicb.2012.00292. PMC 3425911. PMID 22936927.
- Yazdani Foshtomi, Maryam; Braeckman, Ulrike; Derycke, Sofie; Sapp, Melanie; Van Gansbeke, Dirk; Sabbe, Koen; Willems, Anne; Vincx, Magda; Vanaverbeke, Jan (2015). "The Link between Microbial Diversity and Nitrogen Cycling in Marine Sediments is Modulated by Macrofaunal Bioturbation". PLOS ONE. 10 (6): e0130116. Bibcode:2015PLoSO..1030116Y. doi:10.1371/journal.pone.0130116. PMC 4477903. PMID 26102286.
- Soldier Crab Australian Museum. Updated: 15 December 2020.
- Walton, M.E.M.; Al-Maslamani, I.; Chatting, M.; Smyth, D.; Castillo, A.; Skov, M.W.; Le Vay, L. (2021). "Faunal mediated carbon export from mangroves in an arid area". Science of the Total Environment. 755 (Pt 1): 142677. Bibcode:2021ScTEn.755n2677W. doi:10.1016/j.scitotenv.2020.142677. PMID 33077211. S2CID 224810608.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License. - Bouillon, Steven; Borges, Alberto V.; Castañeda-Moya, Edward; Diele, Karen; Dittmar, Thorsten; Duke, Norman C.; Kristensen, Erik; Lee, Shing Y.; Marchand, Cyril; Middelburg, Jack J.; Rivera-Monroy, Victor H.; Smith, Thomas J.; Twilley, Robert R. (2008). "Mangrove production and carbon sinks: A revision of global budget estimates". Global Biogeochemical Cycles. 22 (2): n/a. Bibcode:2008GBioC..22.2013B. doi:10.1029/2007gb003052. S2CID 42641476.
- Alongi, Daniel M. (2014). "Carbon Cycling and Storage in Mangrove Forests". Annual Review of Marine Science. 6: 195–219. Bibcode:2014ARMS....6..195A. doi:10.1146/annurev-marine-010213-135020. PMID 24405426.
- Lugo, Ariel E. (1980). "Mangrove Ecosystems: Successional or Steady State?". Biotropica. 12 (2): 65–72. doi:10.2307/2388158. JSTOR 2388158.
- Maher, D. T.; Santos, I. R.; Golsby-Smith, L.; Gleeson, J.; Eyre, B. D. (2013). "Groundwater-derived dissolved inorganic and organic carbon exports from a mangrove tidal creek: The missing mangrove carbon sink?". Limnology and Oceanography. 58 (2): 475–488. Bibcode:2013LimOc..58..475M. doi:10.4319/lo.2013.58.2.0475. S2CID 86841519.
- Odum, E.P. (1968) "A research challenge: evaluating the productivity of coastal and estuarine water". In: Proceedings of the second sea grant conference", pages 63-64. University of Rhode Island.
- Odum, W.E. and Heald, E.J. (1972) ""Trophic analyses of an estuarine mangrove community". Bulletin of Marine Science, 22(3): 671-738.
- Lee, S. Y. (1995). "Mangrove outwelling: A review". Hydrobiologia. 295 (1–3): 203–212. doi:10.1007/bf00029127. S2CID 28803158.
- Jennerjahn, Tim C.; Ittekkot, Venugopalan (2002). "Relevance of mangroves for the production and deposition of organic matter along tropical continental margins". Naturwissenschaften. 89 (1): 23–30. Bibcode:2002NW.....89...23J. doi:10.1007/s00114-001-0283-x. PMID 12008969. S2CID 33556308.
- Dittmar, Thorsten; Hertkorn, Norbert; Kattner, Gerhard; Lara, Rubén J. (2006). "Mangroves, a major source of dissolved organic carbon to the oceans". Global Biogeochemical Cycles. 20 (1): n/a. Bibcode:2006GBioC..20.1012D. doi:10.1029/2005gb002570. S2CID 128922131.
- Abrantes, Kátya G.; Johnston, Ross; Connolly, Rod M.; Sheaves, Marcus (2015). "Importance of Mangrove Carbon for Aquatic Food Webs in Wet–Dry Tropical Estuaries". Estuaries and Coasts. 38: 383–399. doi:10.1007/s12237-014-9817-2. hdl:10072/141734. S2CID 3957868.
- Twilley, Robert R.; Castañeda-Moya, Edward; Rivera-Monroy, Victor H.; Rovai, Andre (2017). "Productivity and Carbon Dynamics in Mangrove Wetlands". Mangrove Ecosystems: A Global Biogeographic Perspective. pp. 113–162. doi:10.1007/978-3-319-62206-4_5. ISBN 978-3-319-62204-0.
- Guest, Michaela A.; Connolly, Rod M. (2004). "Fine-scale movement and assimilation of carbon in saltmarsh and mangrove habitat by resident animals". Aquatic Ecology. 38 (4): 599–609. doi:10.1007/s10452-004-0442-1. S2CID 3901928.
- Granek, Elise F.; Compton, Jana E.; Phillips, Donald L. (2009). "Mangrove-Exported Nutrient Incorporation by Sessile Coral Reef Invertebrates". Ecosystems. 12 (3): 462–472. doi:10.1007/s10021-009-9235-7. S2CID 587963.
- Vaslet, A.; Phillips, D.L.; France, C.; Feller, I.C.; Baldwin, C.C. (2012). "The relative importance of mangroves and seagrass beds as feeding areas for resident and transient fishes among different mangrove habitats in Florida and Belize: Evidence from dietary and stable-isotope analyses". Journal of Experimental Marine Biology and Ecology. 434–435: 81–93. doi:10.1016/j.jembe.2012.07.024.
- Vepraskas, Michael (2016). Wetland soils : genesis, hydrology, landscapes, and classification. Boca Raton, Florida: CRC Press. ISBN 978-1-4398-9800-0. OCLC 928883552.
- Shiau, Yo-Jin; Chiu, Chih-Yu (2020). "Biogeochemical Processes of C and N in the Soil of Mangrove Forest Ecosystems". Forests. 11 (5): 492. doi:10.3390/f11050492.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - Santos, Isaac R.; Maher, Damien T.; Larkin, Reece; Webb, Jackie R.; Sanders, Christian J. (2019). "Carbon outwelling and outgassing vs. Burial in an estuarine tidal creek surrounded by mangrove and saltmarsh wetlands". Limnology and Oceanography. 64 (3): 996–1013. Bibcode:2019LimOc..64..996S. doi:10.1002/lno.11090. hdl:10536/DRO/DU:30144450. S2CID 91613293.
- Feller, Ilka C.; McKee, Karen L.; Whigham, Dennis F.; O'Neill, John P. (2003). "Nitrogen vs. phosphorus limitation across an ecotonal gradient in a mangrove forest". Biogeochemistry. 62 (2): 145–175. doi:10.1023/A:1021166010892. S2CID 42854661.
- Vitousek, Peter M.; Aber, John D.; Howarth, Robert W.; Likens, Gene E.; Matson, Pamela A.; Schindler, David W.; Schlesinger, William H.; Tilman, David G. (1997). "Human Alteration of the Global Nitrogen Cycle: Sources and Consequences". Ecological Applications. 7 (3): 737. doi:10.1890/1051-0761(1997)007[0737:HAOTGN]2.0.CO;2. hdl:1813/60830. ISSN 1051-0761.
- De-León-Herrera, R.; Flores-Verdugo, F.; Flores-De-Santiago, F.; González-Farías, F. (2015). "Nutrient removal in a closed silvofishery system using three mangrove species (Avicennia germinans, Laguncularia racemosa, and Rhizophora mangle)". Marine Pollution Bulletin. 91 (1): 243–248. Bibcode:2015MarPB..91..243D. doi:10.1016/j.marpolbul.2014.11.040. PMID 25499182.
- Alongi, D.M.; Pfitzner, J.; Trott, L.A.; Tirendi, F.; Dixon, P.; Klumpp, D.W. (2005). "Rapid sediment accumulation and microbial mineralization in forests of the mangrove Kandelia candel in the Jiulongjiang Estuary, China". Estuarine, Coastal and Shelf Science. 63 (4): 605–618. Bibcode:2005ECSS...63..605A. doi:10.1016/j.ecss.2005.01.004.
- Datta, R.; Datta, B.K. (1999 ) "Desiccation induced nitrate and ammonium uptake in the red alga Catenella repens (Rhodophyta, Gigartinales)". Indian J. Geo Mar. Sci., 28: 458–460.
- Shiau, Yo-Jin; Lin, Ming-Fen; Tan, Chen-Chung; Tian, Guanglong; Chiu, Chih-Yu (2017). "Assessing N2 fixation in estuarine mangrove soils". Estuarine, Coastal and Shelf Science. 189: 84–89. Bibcode:2017ECSS..189...84S. doi:10.1016/j.ecss.2017.03.005.
- Parida, Asish Kumar; Das, A. B.; Mittra, B. (2004). "Effects of salt on growth, ion accumulation, photosynthesis and leaf anatomy of the mangrove, Bruguiera parviflora". Trees - Structure and Function. 18 (2): 167–174. doi:10.1007/s00468-003-0293-8. S2CID 20302042.
- Khan, M. Ajmal; Aziz, Irfan (2001). "Salinity tolerance in some mangrove species from Pakistan". Wetlands Ecology and Management. Springer Science and Business Media LLC. 9 (3): 229–233. doi:10.1023/a:1011112908069. ISSN 0923-4861. S2CID 40743147.
- Millennium Ecosystem Assessment (2005) Ecosystems and Human Well-being: Synthesis (p.2) Island Press, Washington, DC. World Resources Institute ISBN 1-59726-040-1
- Valiela, I.; Bowen, J.L.; York, J.K. (2001). "Mangrove Forests: One of the World's Threatened Major Tropical Environments: At least 35% of the area of mangrove forests has been lost in the past two decades, losses that exceed those for tropical rain forests and coral reefs, two other well-known threatened environments". BioScience. 51 (10): 807–815. doi:10.1641/0006-3568(2001)051[0807:MFOOTW]2.0.CO;2.
- Turschwell, Mischa P.; Tulloch, Vivitskaia J.D.; Sievers, M.; Pearson, R.M.; Andradi-Brown, D.A.; Ahmadia, G.N.; Connolly, R.M.; Bryan-Brown, D.; Lopez-Marcano, S.; Adame, M.F.; Brown, C.J. (2020). "Multi-scale estimation of the effects of pressures and drivers on mangrove forest loss globally". Biological Conservation. 247: 108637. doi:10.1016/j.biocon.2020.108637. S2CID 219750253.
- Botkin, D. and E. Keller (2003) Environmental Science: Earth as a living planet (p.2) John Wiley & Sons. ISBN 0-471-38914-5
- Hamilton, Stuart (2013). "Assessing the Role of Commercial Aquaculture in Displacing Mangrove Forest". Bulletin of Marine Science. 89 (2): 585–601. doi:10.5343/bms.2012.1069.
- "2010a. ""World Atlas of Mangroves" Highlights the Importance of and Threats to Mangroves: Mangroves among World's Most Valuable Ecosystems." Press release. Arlington, Virginia". The Nature Conservancy. Archived from the original on 2010-07-17. Retrieved 2014-01-25.
- Hamilton, Stuart E; Casey, Daniel (2016). "Creation of a high spatio-temporal resolution global database of continuous mangrove forest cover for the 21st century (CGMFC-21)". Global Ecology and Biogeography. 25 (6): 729–38. arXiv:1412.0722. doi:10.1111/geb.12449. S2CID 55999275.
- "The Secret Life of Mangroves - Television - Distribution - ZED". www.zed.fr.
- Charcoal is used as a cheap source of energy in developing countries for cooking purposes
- "The Secret Life of Mangroves - Television - Distribution - ZED". www.zed.fr. Archived from the original on 2022-04-06. Retrieved 2021-11-09.
- "Thailand – Trang Province – Taking Back the Mangroves with Community Management | The EcoTipping Points Project". Ecotippingpoints.org. Retrieved 2012-02-08.
- "Bees: An income generator and mangrove conservation tool for a community in Thailand". IUCN. December 14, 2016.
- "Harvesting honey for mangrove resilience in the Tsiribihina delta". The Mangrove Alliance. February 5, 2018.
- "Madagascar: What's good for the forest is good for the native silk industry". Mongabay Environmental News. August 16, 2019.
- Hamilton, S. & S. Collins (2013) Las respuestas a los medios de subsistencia deforestación de los manglares en las provincias del norte de Ecuador. Bosque 34:2
- "Tree News, Spring/Summer 2005, Publisher Felix Press". Treecouncil.org.uk. Retrieved 2012-02-08.
- Laffoley, D. & Baxter, J.M. (eds.) (2019). Ocean deoxygenation: Everyone's problem - Causes, impacts, consequences and solutions. IUCN, Switzerland.
- "2010a. ""World Atlas of Mangroves" Highlights the Importance of and Threats to Mangroves: Mangroves among World's Most Valuable Ecosystems." Press release. Arlington, Virginia". The Nature Conservancy. Archived from the original on 2010-07-17. Retrieved 2014-01-25.
- Warne, Kennedy (February 2007). "Mangroves: Forests of the Tide". National Geographic. Tim Laman, photographer. National Geographic Society. Retrieved 2010-08-08.
- Sato, Gordon; Abraham Fisseha; Simon Gebrekiros; Hassan Abdul Karim; Samuel Negassi; Martin Fischer; Emanuel Yemane; Johannes Teclemariam & Robert Riley (2005). "A novel approach to growing mangroves on the coastal mud flats of Eritrea with the potential for relieving regional poverty and hunger". Wetlands. 25 (3): 776–779. doi:10.1672/0277-5212(2005)025[0776:ANATGM]2.0.CO;2. S2CID 45705546.
- Guest, Peter (April 28, 2019). "Tropical forests are dying. Seed-slinging drones can save them". WIRED. Retrieved 11 August 2021.
- Pearce, Fred (May 2, 2019). "On Java's Coast, A Natural Approach to Holding Back the Waters". Yale E360. Retrieved 2019-05-15.
- Lee, Shing Yip; Hamilton, Stu; Barbier, Edward B.; Primavera, Jurgenne; Lewis, Roy R. (June 2019). "Better restoration policies are needed to conserve mangrove ecosystems". Nature Ecology & Evolution. 3 (6): 870–872. doi:10.1038/s41559-019-0861-y. ISSN 2397-334X. PMID 31036899. S2CID 139106235.
- Cherrington EA, Hernandez BE, Trejos NA, Smith OA, Anderson ER, Flores AI, Garcia BC (2010). Identification of Threatened and Resilient Mangroves in the Belize Barrier Reef System (PDF). Water Center for the Humid Tropics of Latin America and the Caribbean (CATHALAC) / Regional Visualization & Monitoring System (SERVIR) (Report). Technical report to the World Wildlife Fund. Archived from the original (PDF) on July 31, 2013.
- "Pelican_Cays_Review" (PDF). Retrieved 2012-02-08.
- "Cherrington, E.A., Ek, E., Cho, P., Howell, B.F., Hernandez, B.E., Anderson, E.R., Flores, A.I., Garcia, B.C., Sempris, E., and D.E. Irwin. 2010. "Forest Cover and Deforestation in Belize: 1980–2010." Water Center for the Humid Tropics of Latin America and the Caribbean. Panama City, Panama. 42 pp" (PDF). Archived from the original (PDF) on July 29, 2013.
- "Government of Belize (GOB). 2003. "Forests Act Subsidiary Laws." Chapter 213 in: Substantive Laws of Belize. Revised Edition 2003. Government Printer: Belmopan, Belize. 137 pp" (PDF). Archived from the original (PDF) on March 13, 2012.
- Cooper, E.; Burke, L.; Bood, N. (2009). "Coastal Capital: Belize. The Economic Contribution of Belize's Coral Reefs and Mangroves" (PDF). Washington, DC: World Resources Institute. Retrieved 2014-06-23.
- "Mapping Global Mangrove Forests", ORNL DAAC News, May 6, 2019, retrieved May 15, 2019
- Rasmussen, Carol; Carlowicz, Mike (February 25, 2019), New Satellite-Based Maps of Mangrove Heights (Text.Article), retrieved May 15, 2019
- Simard, M.; Fatoyinbo, L.; Smetanka, C.; Rivera-Monroy, V. H.; Castañeda-Moya, E.; Thomas, N.; Van der Stocken, T. (2018). "Mangrove canopy height globally related to precipitation, temperature and cyclone frequency". Nature Geoscience. 12 (1): 40–45. doi:10.1038/s41561-018-0279-1. hdl:2060/20190029179. S2CID 134827807.