ITCH
ITCH is a HECT domain E3 ubiquitin ligase that is ablated in non-agouti-lethal 18H (aka Itchy) mice.[5][6] Itchy mice develop a severe immunological phenotype after birth that includes hyperplasia of lymphoid and hematopoietic cells, and stomach and lung inflammation.[7][8] In humans ITCH deficiency causes altered physical growth, craniofacial morphology defects, defective muscle development, and aberrant immune system function.[9] ITCH contains a C2 domain, proline-rich region, WW domains, HECT domain, and multiple amino acids that are phosphorylated and ubiquitinated.[10]
Regulation by phosphorylation
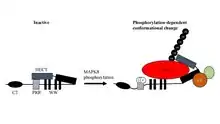
ITCH is regulated by MAPK8.[11] MAPK8 regulates JUNB protein turnover by MAPK8-dependent phosphorylation of ITCH and a subsequent conformational change in ITCH. This mechanism is discrete from the direct activation of Jun family transcription factors by direct phosphorylation. ITCH serves as a paradigm for our understanding of the regulation of the ubiquitylation machinery by direct protein phosphorylation of its components. Importantly, this regulatory process controls the balance of Th2 cytokine secretion by negatively regulating JUNB levels and Interleukin 4 transcription.[12]
Interaction partners
Itch has been shown to interact with a number of proteins,[13] including:
References
- GRCh38: Ensembl release 89: ENSG00000078747 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000027598 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Perry WL, Hustad CM, Swing DA, O'Sullivan TN, Jenkins NA, Copeland NG (February 1998). "The itchy locus encodes a novel ubiquitin protein ligase that is disrupted in a18H mice". Nature Genetics. 18 (2): 143–146. doi:10.1038/ng0298-143. PMID 9462742. S2CID 9438916.
- "Q96J02 | SWISS-MODEL Repository".
- Melino G, Gallagher E, Aqeilan RI, Knight R, Peschiaroli A, Rossi M, et al. (July 2008). "Itch: a HECT-type E3 ligase regulating immunity, skin and cancer". Cell Death and Differentiation. 15 (7): 1103–1112. doi:10.1038/cdd.2008.60. PMID 18552861. S2CID 2626150.
- Aki D, Zhang W, Liu YC (July 2015). "The E3 ligase Itch in immune regulation and beyond". Immunological Reviews. 266 (1): 6–26. doi:10.1111/imr.12301. PMID 26085204. S2CID 42110767.
- Lohr NJ, Molleston JP, Strauss KA, Torres-Martinez W, Sherman EA, Squires RH, et al. (March 2010). "Human ITCH E3 ubiquitin ligase deficiency causes syndromic multisystem autoimmune disease". American Journal of Human Genetics. 86 (3): 447–453. doi:10.1016/j.ajhg.2010.01.028. PMC 2833372. PMID 20170897.
- "ITCH (human)". www.phosphosite.org. Retrieved 2020-10-27.
- Karin M, Gallagher E (2005). "From JNK to pay dirt: jun kinases, their biochemistry, physiology and clinical importance". IUBMB Life. 57 (4–5): 283–295. doi:10.1080/15216540500097111. PMID 16036612. S2CID 25508987.
- Gao M, Karin M (September 2005). "Regulating the regulators: control of protein ubiquitination and ubiquitin-like modifications by extracellular stimuli". Molecular Cell. 19 (5): 581–593. doi:10.1016/j.molcel.2005.08.017. PMID 16137616.
- "ITCH itchy E3 ubiquitin protein ligase [Homo sapiens (human)] - Gene - NCBI". www.ncbi.nlm.nih.gov. Retrieved 2021-12-29.
- Bhandari D, Robia SL, Marchese A (March 2009). "The E3 ubiquitin ligase atrophin interacting protein 4 binds directly to the chemokine receptor CXCR4 via a novel WW domain-mediated interaction". Molecular Biology of the Cell. 20 (5): 1324–1339. doi:10.1091/mbc.e08-03-0308. PMC 2649280. PMID 19116316.
- Fang D, Kerppola TK (October 2004). "Ubiquitin-mediated fluorescence complementation reveals that Jun ubiquitinated by Itch/AIP4 is localized to lysosomes". Proceedings of the National Academy of Sciences of the United States of America. 101 (41): 14782–14787. doi:10.1073/pnas.0404445101. PMC 522008. PMID 15469925.
- Ahn YH, Kurie JM (October 2009). "MKK4/SEK1 is negatively regulated through a feedback loop involving the E3 ubiquitin ligase itch". The Journal of Biological Chemistry. 284 (43): 29399–29404. doi:10.1074/jbc.M109.044958. PMC 2785572. PMID 19737936.
- Enzler T, Chang X, Facchinetti V, Melino G, Karin M, Su B, Gallagher E (September 2009). "MEKK1 binds HECT E3 ligase Itch by its amino-terminal RING motif to regulate Th2 cytokine gene expression". Journal of Immunology. 183 (6): 3831–3838. doi:10.4049/jimmunol.0803412. PMID 19710465. S2CID 12554570.
- Kathania M, Zeng M, Yadav VN, Moghaddam SJ, Yang B, Venuprasad K (March 2015). "Ndfip1 regulates itch ligase activity and airway inflammation via UbcH7". Journal of Immunology. 194 (5): 2160–2167. doi:10.4049/jimmunol.1402742. PMID 25632008. S2CID 22850215.
- Gallagher E, Gao M, Liu YC, Karin M (February 2006). "Activation of the E3 ubiquitin ligase Itch through a phosphorylation-induced conformational change". Proceedings of the National Academy of Sciences of the United States of America. 103 (6): 1717–1722. doi:10.1073/pnas.0510664103. PMC 1413664. PMID 16446428.
- Oberst A, Malatesta M, Aqeilan RI, Rossi M, Salomoni P, Murillas R, et al. (July 2007). "The Nedd4-binding partner 1 (N4BP1) protein is an inhibitor of the E3 ligase Itch". Proceedings of the National Academy of Sciences of the United States of America. 104 (27): 11280–11285. doi:10.1073/pnas.0701773104. PMC 2040890. PMID 17592138.
- Puca L, Chastagner P, Meas-Yedid V, Israël A, Brou C (October 2013). "Α-arrestin 1 (ARRDC1) and β-arrestins cooperate to mediate Notch degradation in mammals". Journal of Cell Science. 126 (Pt 19): 4457–4468. doi:10.1242/jcs.130500. PMID 23886940. S2CID 9783441.
- Rossi M, Aqeilan RI, Neale M, Candi E, Salomoni P, Knight RA, et al. (August 2006). "The E3 ubiquitin ligase Itch controls the protein stability of p63". Proceedings of the National Academy of Sciences of the United States of America. 103 (34): 12753–12758. doi:10.1073/pnas.0603449103. PMC 1550770. PMID 16908849.
- Rossi M, De Laurenzi V, Munarriz E, Green DR, Liu YC, Vousden KH, et al. (February 2005). "The ubiquitin-protein ligase Itch regulates p73 stability". The EMBO Journal. 24 (4): 836–848. doi:10.1038/sj.emboj.7600444. PMC 549609. PMID 15678106.
External links
- ITCH+protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)



