Centipede
Centipedes (from Neo-Latin centi-, "hundred", and Latin pes, pedis, "foot") are predatory arthropods belonging to the class Chilopoda (Ancient Greek χεῖλος, kheilos, lip, and Neo-Latin suffix -poda, "foot", describing the forcipules) of the subphylum Myriapoda, an arthropod group which includes millipedes and other multi-legged animals. Centipedes are elongated segmented (metameric) creatures with one pair of legs per body segment. All centipedes are venomous and can inflict painful stings, injecting their venom through pincer-like appendages known as forcipules or toxicognaths, which are actually modified legs instead of fangs. Despite the name, no centipede has exactly 100 pairs of legs; number of legs ranges from 15 pairs to 191 pairs, always an odd number.[1]
| Centipede | |
|---|---|
 | |
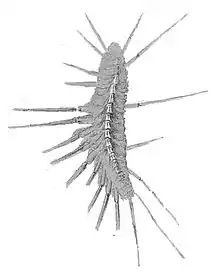
| Various centipedes (clockwise from top left): Thereuopoda clunifera, a Scutigeromorph; Lithobius forficatus, a Lithobiomorph; Geophilus, a Geophilomorph; and Scolopendra cataracta, a Scolopendromorph | |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Subphylum: | Myriapoda |
| Class: | Chilopoda Latreille, 1817 |
| Orders and suborders | |
| |
Centipedes are predominantly generalist carnivorous, hunting for a variety of prey items that can be overpowered.[2] They have a wide geographical range, which can be found in terrestrial habitats from tropical rainforests to deserts. Within these habitats, centipedes require a moist microhabitat because they lack the waxy cuticle of insects and arachnids, therefore causing them to rapidly lose water. Accordingly, they avoid direct sunlight by staying under cover or by being active at night.
Description
Centipedes have a rounded or flattened head, bearing a pair of antennae at the forward margin. They have a pair of elongated mandibles, and two pairs of maxillae. The first pair of maxillae form the lower lip, and bear short palps. The first pair of limbs stretch forward from the body over the mouth. These limbs, or forcipules, end in sharp claws and include venom glands that help the animal to kill or paralyze its prey.[3] Their size ranges from a few millimetres in the smaller lithobiomorphs and geophilomorphs to about 30 cm (12 in) in the largest scolopendromorphs.[4]
Sensory organs
Many species of centipedes lack eyes, but some possess a variable number of ocelli, sometimes clustered together to form true compound eyes. However, these eyes are only capable of discerning light from dark, and provide no true vision. In some species, the first pair of legs can function as sensory organs, similar to antennae; unlike the antennae of most other invertebrates, these point backwards. An unusual clustering of sensory organs found in some centipedes is the organ of Tömösváry. The organs, at the base of the antennae, consist of a disc-like structure and a central pore, with an encircling of sensitive cells. They are likely used for sensing vibrations, and may provide a weak form of hearing.[3]
Forcipules
Forcipules are unique to centipedes. The forcipules are modifications of the first pair of legs (the maxillipeds), forming a pincer-like appendage, just behind the head. Forcipules are not oral mouthparts, though they are used to subdue prey by injecting venom and gripping the prey animal. Venom glands run through a tube, from inside the head to the tip of each forcipule.[5]
Body
Behind the head, the body consists of at least fifteen segments. Most of the segments bear a single pair of legs; the maxillipeds project forward from the first body segment, while the final two segments are small and legless. Each pair of legs is slightly longer than the pair preceding them, ensuring that they do not overlap, which reduces the chance that they will collide and trip the animal. The last pair of legs may be as much as twice the length of the first pair. The final segment bears a telson, and includes the openings of the reproductive organs.[3]
Centipedes mainly use their antennae to seek out their prey. The digestive tract forms a simple tube, with digestive glands attached to the mouthparts. Like insects, centipedes breathe through a tracheal system, typically with a single opening, or spiracle, on each body segment. They excrete waste through a single pair of malpighian tubules.[3]
Ultimate legs

Just as the first pair of legs are modified into forcipules, the back legs are modified into "ultimate legs", also called anal legs, caudal legs, and terminal legs.[6] Their use varies between species, but does not include locomotion.[7][8] The ultimate legs may be elongated and thin, thickened, or pincer-like.[8] They are frequently sexually dimorphic, and may play a role in mating rituals.[7][8] Because glandular pores occur more frequently on ultimate legs than on the "walking" legs, they may serve a sensory role.[7][8] They are sometimes used in defensive postures, and some species use them to capture prey, defend themselves against predators, or suspend themselves from objects such as branches, using the legs as pincers.[7] Several species use their ultimate legs upon encountering another centipede, trying to grab the body of the other centipede.[9]
Members of the genus Alipes can stridulate their leaf-like ultimate legs to distract or threaten predators.[9] Rhysida immarginata togoensis makes a faint creaking sound when it swings its ultimate legs.[9]
Distinction from millipedes
Scholars have noted that disinformation exists about the difference between millipedes and centipedes, and they seek to provide more generalized information for education purposes.[1] Both groups of myriapods have long, multi-segmented bodies, many legs, a single pair of antennae, and the presence of postantennal organs. Centipedes have one pair of legs per segment, while millipedes have two. Their heads differ in that millipedes have short, elbowed antennae, a pair of robust mandibles and a single pair of maxillae fused into a lip; centipedes have long, threadlike antennae, a pair of small mandibles, two pairs of maxillae and a pair of large venom claws.[10]

| Trait | Millipedes | Centipedes |
|---|---|---|
| Legs | Two pairs on most body segments; attached to underside of body | One pair per body segment; attached to sides of body; last pair extends backwards |
| Locomotion | Generally adapted for burrowing or inhabiting small crevices; slow-moving | Generally adapted for running, except for the burrowing soil centipedes |
| Feeding | Primarily detritivores, some herbivores, few carnivores; no venom | Primarily carnivores with front legs modified into venomous fangs |
| Spiracles | On underside of body | On the sides or top of body |
| Reproductive openings | Third body segment | Last body segment |
| Reproductive behaviour | Male generally inserts spermatophore into female, using his gonopods | Male produces spermatophore that is usually picked up by female |
Lifecycle
Reproduction

Centipede reproduction does not involve copulation. Males deposit a spermatophore for the female to take up. In temperate areas, egg laying occurs in spring and summer. A few parthenogenetic species are known.[11] Females provide parental care, both by curling their bodies around eggs and young, and by grooming them, probably to remove fungi and bacteria.[12]
Centipedes are longer-lived than insects; the European Lithobius forficatus may live for 5 to 6 years,[13] and the wide-ranging Scolopendra subspinipes can live for over 10 years.[14] The combination of a small number of eggs laid, long gestation period, and long time of development to reproduction has led authors to label lithobiomorph centipedes as K-selected.[15]
Development
Centipedes grow their legs at different points in their development. In the primitive condition, seen in the Lithobiomorpha, Scutigeromorpha, and Craterostigmomorpha, development is anamorphic: more segments and pairs of legs are grown between moults.[16] For example, Scutigera coleoptrata, the house centipede, hatches with only four pairs of legs and in successive moults has 5, 7, 9, 11, 15, 15, 15 and 15 pairs respectively, before becoming a sexually mature adult. Life stages with fewer than 15 pairs of legs are called larval stadia (there are about five stages). After the full complement of legs is achieved, the now postlarval stadia (about five more stages) develop gonopods, sensory pores, more antennal segments, and more ocelli. All mature lithobiomorph centipedes have 15 leg-bearing segments.[17] The Craterostigmomorpha only have one phase of anamorphosis, with embryos having 12 pairs, and adults 15.[18]
The clade Epimorpha, consisting of the orders Geophilomorpha and Scolopendromorpha, is epimorphic, meaning that all pairs of legs are developed in the embryonic stages, and offspring do not develop more legs between moults. This clade contains the longest centipedes; the maximum number of thoracic segments varies within species, often on a geographical basis, and in most cases, females bear more legs than males. The number of leg-bearing segments in these groups varies from 15 to 191, but the developmental mode of their creation is constrained so that they are always added in pairs. The total number of pairs begins and remains odd throughout development, so there are never exactly 100 pairs, despite the group's common name.[18][19][20][21]
Centipede segments are developed in two phases. Firstly, the head gives rise to a fixed but odd number of segments, driven by Hox genes as in all arthropods.[22][23] Secondly, pairs of segments are added at the tail (posterior) end by the creation of a prepattern unit, a double segment, which is then always divided into two. The repeated creation of these prepattern units is driven by an oscillator clock, implemented with the Notch signalling pathway. The segments are homologous with the legs of other arthropods such as trilobites; it would be sufficient for the Notch clock to run faster, as it does in snakes, to create more legs.[22]
Ecology
Diet
Centipedes are predominantly generalist predators, which means they are adapted to eat a broad range of prey.[24] Common prey items include lumbricid earthworms, dipteran fly larvae, collembolans, and other centipedes.[25] They are carnivorous; study of gut contents suggests that plant material is an unimportant part of their diets, although they eat vegetable matter when starved during laboratory experiments.[24]
Species of Scolopendromorph, noticeably members from the genera Scolopendra and Ethmostigmus, are able to hunt for substantial prey items, including large invertebrates and sizable vertebrates, which could be larger than the myriapod itself.[26][2] For instance, Scolopendra gigantea (the Amazonian giant centipede) preys on tarantulas, scorpions, lizards, frogs, birds, mice, snakes, and even bats, catching them in midflight.[27] Three species (Scolopendra cataracta, S. paradoxa, and S. alcyona) are amphibious, believed to hunt aquatic or amphibious invertebrates.[28][29]
Predators

Many larger animals prey upon centipedes, such as mongooses, mice, salamanders, beetles and some specialist snake species.[30] They form an important item of diet for many species and the staple diet of some such as the African ant Amblyopone pluto, which feeds solely on geophilomorph centipedes,[31] and the South African Cape black-headed snake Aparallactus capensis.[30]
Defences
Some Geophilomorph, Lithobiomorph, and Scolopendromorph centipedes produce sticky, toxic secretions to defend themselves. The various secretions ward off or entangle predators.[32][33] Scolopendromorph secretions contain hydrogen cyanide.[34] Among Geophilomorphs, the secretions of Geophilus vittatus are sticky and odorous, and contain hydrogen cyanide.[35]
The giant desert centipede of Arizona, Scolopendra polymorpha, has a black head and tail, and an orange body; this conspicuous pattern may be aposematic, an honest signal of the animal's toxicity.[12] Many species raise and splay their ultimate legs and display the spines found on the legs in a defensive threat posture.[9]
Habitat and behaviour
Because centipedes lack the waxy water-resistant cuticle of other arthropods, they are more susceptible to water loss via evaporation.[36] Thus, centipedes are most commonly found in high-humidity environments to avoid dehydration,[37] and are mostly nocturnal.[38]
Centipedes live in many different habitats including in soil and leaf litter; they are found in environments as varied as tropical rain forests,[25] deserts,[39] and caves.[40] Some geophilomorphs are adapted to littoral habitats, where they feed on barnacles.[41]
Threatened species
As of the 2019 IUCN Red List, there are two vulnerable and one endangered species of centipede: the Serpent Island centipede (Scolopendra abnormis), Turk's earth centipede (Nothogeophilus turki), and the Seychelles long-legged centipede (Seychellonema gerlachi), the first two of which are vulnerable and the last endangered.[42][43][44]
Evolution
Fossil history
The fossil record of centipedes extends back to 430 million years ago, during the Late Silurian,[45][46] though they are rare throughout the Paleozoic.[47] Three species, two scutigeromorphs and one scolopendromorph, have been described from the Mazon Creek fossil beds, which are Carboniferous, 309–307 mya.[48] More species appear in the Mesozoic, including scolopendromorphs and scutigeromorphs in the Cretaceous.[47][49]
External phylogeny
The following cladogram shows the position of the Chilopoda within the arthropods as of 2019:[50][51]
| Arthropoda |
| ||||||||||||||||||
Internal phylogeny
Within the myriapods, centipedes are believed to be the first of the extant classes to branch from the last common ancestor. The five orders of centipedes are: Craterostigmomorpha, Geophilomorpha, Lithobiomorpha, Scolopendromorpha, and Scutigeromorpha. These orders are united into the clade Chilopoda by the following synapomorphies:[52]
- The first postcephalic appendage is modified to venom claws.
- The embryonic cuticle on second maxilliped has an egg tooth.
- The trochanter–prefemur joint is fixed.
- A spiral ridge occurs on the nucleus of the spermatozoon.
The Chilopoda are then split into two clades: the Notostigmophora including the Scutigeromorpha and the Pleurostigmophora including the other four orders. The following physical and developmental traits can be used to separate members of the Pleurostigmomorpha from Notostigmomorpha:[53][54]
- The spiracles are located on the sides of the centipede (in Notostigmomorphs, they are located dorsally).
- The spiracles are deep, more complex, and always present in pairs.
- The head is somewhat flatter.
- The centipedes can develop through either anamorphosis or epimorphosis.
It was previously believed that Chilopoda was split into Anamorpha (Lithobiomorpha and Scutigeromorpha) and Epimorpha (Geophilomorpha and Scolopendromorpha), based on developmental modes, with the relationship of the Craterostigmomorpha being uncertain. Recent phylogenetic analyses using combined molecular and morphological characters supports the previous phylogeny. The Epimorpha still exist as a monophyletic group within the Pleurostigmophora, but the Anamorpha are paraphyletic, as shown in the cladogram:[52]
| Chilopoda |
|
"Anamorpha" | ||||||||||||||||||||||||
Evolution of venoms
All centipedes are venomous. Over the first 50 million years of the clade's evolutionary history, centipede venoms appear to have consisted of a simple cocktail of about four different components, and differentiation into specific venom types appears to have only occurred after the currently recognized five orders had developed. The evolution of the venom includes horizontal gene transfer, involving bacteria, fungi and oomycetes.[55]
Interaction with humans
As food

As a food item, certain large centipedes are consumed in China, usually skewered and grilled or deep fried. They are often seen in street vendors’ stalls in large cities, including Donghuamen and Wangfujing markets in Beijing.[56][57]
Large centipedes are steeped in alcohol to make centipede vodka.[58]
Hazard
Some species of centipedes can be hazardous to humans because of their bite. While a bite to an adult human is usually very painful and may cause severe swelling, chills, fever, and weakness, it is unlikely to be fatal. Bites can be dangerous to small children and those with allergies to bee stings. The venomous bite of larger centipedes can induce anaphylactic shock in such people. Smaller centipedes are generally incapable of piercing human skin.[59]
Even small centipedes that cannot pierce human skin are considered frightening by some humans due to their dozens of legs moving at the same time and their tendency to dart swiftly out of the darkness towards one's feet.[60] A 19th-century Tibetan poet warned his fellow Buddhists, "if you enjoy frightening others, you will be reborn as a centipede."[61]
References
- Shelley, Rowland M. (1999). "Centipedes and millipedes with emphasis on North American fauna". The Kansas School Naturalist. 45 (3): 1–16. Archived from the original on 2016-11-12. Retrieved 2013-10-14.
- P.G. Guizze1, Samuel; Knysak1, Irene; C. Barbaro, Katia; Karam-Gemael, Manoela; Chagas-Jr, Amazonas (2016-11-26). "Predatory behavior of three centipede species of the order Scolopendromorpha (Arthropoda: Myriapoda: Chilopoda)". Zoologia. 33 (6): e20160026. doi:10.1590/S1984-4689zool-20160026.
- Barnes, Robert D. (1982). Invertebrate Zoology. Philadelphia, Pennsylvania: Holt-Saunders International. pp. 810–816. ISBN 978-0-03-056747-6.
- Minelli, A.; Golovatch, S.I. (2017). "Myriapods". Reference Module in Life Sciences. Elsevier. doi:10.1016/b978-0-12-809633-8.02259-7.
- Dugon, Michel M.; Black, Alexander; Arthur, Wallace (2012-05-01). "Variation and specialisation of the forcipular apparatus of centipedes (Arthropoda: Chilopoda): A comparative morphometric and microscopic investigation of an evolutionary novelty". Arthropod Structure & Development. 41 (3): 231–243. doi:10.1016/j.asd.2012.02.001. ISSN 1467-8039. PMID 22370199.
- Bonato, Lucio; Edgecombe, Gregory; Lewis, John; Minelli, Alessandro; Pereira, Luis; Shelley, Rowland; Zapparoli, Marzio (2010-11-18). "A common terminology for the external anatomy of centipedes (Chilopoda)". ZooKeys (69): 17–51. doi:10.3897/zookeys.69.737. ISSN 1313-2970. PMC 3088443. PMID 21594038.
- Kenning, Matthes; Müller, Carsten H. G.; Sombke, Andy (2017-11-14). "The ultimate legs of Chilopoda (Myriapoda): a review on their morphological disparity and functional variability". PeerJ. 5: e4023. doi:10.7717/peerj.4023. ISSN 2167-8359. PMC 5691793. PMID 29158971.
- Kenning, Matthes; Schendel, Vanessa; Müller, Carsten H. G.; Sombke, Andy (2019). "Comparative morphology of ultimate and walking legs in the centipede Lithobius forficatus (Myriapoda) with functional implications". Zoological Letters. 5: 3. doi:10.1186/s40851-018-0115-x. ISSN 2056-306X. PMC 6330759. PMID 30656061.
- Kronmüller, Christian; Lewis, John G.J. (2015-06-30). "On the function of the ultimate legs of some Scolopendridae (Chilopoda, Scolopendromorpha)". ZooKeys (510): 269–278. doi:10.3897/zookeys.510.8674. ISSN 1313-2970. PMC 4523778. PMID 26257548.
- Blower, John Gordon (1985). Millipedes: Keys and Notes for the Identification of the Species. Brill Archive. p. 1. ISBN 978-90-04-07698-3.
- Lewis 2007, pp. 252–298.
- Lizotte, Renée. "Centipedes & Millipedes". Arizona-Sonora Desert Museum. Retrieved 2 May 2023.
- Lewis 2007, p. 328.
- Yates III, Julian R. (December 1992). "Scolopendra subspinipes (Leach)". University of Hawaii EXTension ENTOmology & UH-CTAHR Integrated Pest Management Program: Knowledge Master. Retrieved 2016-12-24.
- Albert, A. M. (1979). "Chilopoda as part of the predatory macroarthropod fauna in forests: abundance, life-cycle, biomass, and metabolism". In Camatini, Marina (ed.). Myriapod biology. Academic Press. pp. 215–231. ISBN 978-0-12-155750-8.
- Fusco, Giuseppe (December 2005). "Trunk segment numbers and sequential segmentation in myriapods". Evolution & Development. 7 (6): 608–617. doi:10.1111/j.1525-142X.2005.05064.x. PMID 16336414. S2CID 21401688. Retrieved 25 August 2020.
- Lewis 2007, p. 27.
- Edgecombe, Gregory D.; Giribet, Gonzalo (2007). "Evolutionary biology of centipedes (Myriapoda: Chilopoda)". Annual Review of Entomology. 52 (1): 151–170. doi:10.1146/annurev.ento.52.110405.091326. PMID 16872257.
- Arthur, Wallace (2002). "The interaction between developmental bias and natural selection from centipede segmentation to a general hypothesis". Heredity. 89 (4): 239–246. doi:10.1038/sj.hdy.6800139. PMID 12242638.
- Arthur, Wallace; Chapman, Ariel D. (2005). "The centipede Strigamia maritima: what it can tell us about development and evolution of segmentation". BioEssays. 27 (6): 653–660. doi:10.1002/bies.20234. PMID 15892117.
- Minelli, Alessandro (2009). "Evolutionary developmental biology does not offer a significant challenge to the neo-Darwinian paradigm". In Ayala, Francisco J.; Arp, Robert (eds.). Contemporary Debates in Philosophy of Biology. John Wiley and Sons. pp. 213–226. ISBN 978-1-4051-5999-9.
- Held, Lewis I. (2014). "Why the centipede has odd segments". How the Snake Lost its Legs. Curious Tales from the Frontier of Evo-Devo. Cambridge University Press. pp. 69, 120. ISBN 978-1-107-62139-8.
- Hughes, Cynthia L.; Kaufman, Thomas C. (2002). "Exploring the myriapod body plan: expression patterns of the ten Hox genes in a centipede" (PDF). Development. 129 (5): 1225–1238. doi:10.1242/dev.129.5.1225. PMID 11874918.
- Lewis 2007, p. 168.
- Klarner, Bernhard; Winkelmann, Helge; Krashevska, Valentyna; Maraun, Mark; Widyastuti, Rahayu; Scheu, Stefan (2017-08-01). "Trophic niches, diversity and community composition of invertebrate top predators (Chilopoda) as affected by conversion of tropical lowland rainforest in Sumatra (Indonesia)". PLOS ONE. 12 (8): e0180915. Bibcode:2017PLoSO..1280915K. doi:10.1371/journal.pone.0180915. ISSN 1932-6203. PMC 5538669. PMID 28763453.
- Halpin, Luke R.; Terrington, Daniel I.; Jones, Holly P.; Mott, Rowan; Wong, Wei Wen; Dow, David C.; Carlile, Nicholas; Clarke, Rohan H. (2021-08-03). "Arthropod Predation of Vertebrates Structures Trophic Dynamics in Island Ecosystems". The American Naturalist. 198 (4): 540–550. doi:10.1086/715702. PMID 34559614.
- Molinari, Jesús; Gutiérrez, Eliécer E.; de Ascenção, Antonio A.; et al. (2005). "Predation by giant centipedes, Scolopendra gigantea, on three species of bats in a Venezuelan cave" (PDF). Caribbean Journal of Science. 4 (2): 340–346. Archived from the original (PDF) on 2010-10-09. Retrieved 2011-02-20.
- Holmes, O. (1 July 2016). "Giant swimming, venomous centipede discovered by accident in world-first". The Guardian. Retrieved 1 July 2016.
- Sho, T. (12 April 2021). "A new amphibious species of the genus Scolopendra Linnaeus, 1758 (Scolopendromorpha, Scolopendridae) from the Ryukyu Archipelago and Taiwan". Biotaxa. Retrieved 2022-01-10.>
- Lewis 2007, pp. 354–356.
- Hölldobler, Bert; Wilson, Edward O. (1990). "The specialized predators". The Ants. Harvard University Press. pp. 557–572. ISBN 978-0-674-04075-5.
- Rosenberg, Jörg; Müller, Carsten (2009-12-15). "Morphology in Chilopoda – a survey". Soil Organisms. 81 (3): 1–55–1–55. ISSN 2509-9523.
- Eisner, Thomas; Eisner, Maria; Siegler, Melody (2005). Secret weapons: defenses of insects, spiders, scorpions and other many-legged creatures. Cambridge, Massachusetts: The Belknap Press. pp. 33–36. ISBN 978-0-674-01882-2.
- Maschwitz, U.; Lauschke, Ursula; Würmli, M. (1979-11-01). "Hydrogen cyanide-producing glands in a scolopender, Asanada n.sp. (Chilopoda, scolopendridae)". Journal of Chemical Ecology. 5 (6): 901–907. doi:10.1007/BF00990212. ISSN 1573-1561. S2CID 29951682.
- Jones, Tappey H.; Conner, William E.; Meinwald, Jerrold; Eisner, Hans E.; Eisner, Thomas (1976). "Benzoyl cyanide and mandelonitrile in the cyanogenetic secretion of a centipede". Journal of Chemical Ecology. 2 (4): 421–429. doi:10.1007/bf00988807. ISSN 0098-0331. S2CID 20051462.
- "Insects, Spiders, Centipedes, Millipedes". National Park Service. 2017-10-17. Retrieved 2023-05-10.
- Lewbart, Gregory A. (2006). Invertebrate Medicine. Blackwell Publishing. p. 198. ISBN 978-0-8138-1844-3.
- Hasan, Shadi; Hassan, Kamal (2004-12-15). "Proteinuria associated with centipede bite". Pediatric Nephrology. 20 (4): 550–551. doi:10.1007/s00467-004-1685-8. ISSN 0931-041X. PMID 15599772. S2CID 3120582.
- "Animal Fact Sheet: Desert Centipede". Arizona-Sonora Desert Museum. Retrieved 2 May 2023.
- Dunning, Hayley (1 July 2015). "Cave centipede from hell is the deepest-dwelling ever discovered". Natural History Museum. Retrieved 2 May 2023.
- Lewis, J. G. E. (1961). "The life history and ecology of the littoral centipede Strigamia maritima (Leach)". Proceedings of the Zoological Society of London. 137 (2): 221–248. doi:10.1111/j.1469-7998.1961.tb05900.x.
- Pearce-Kelly, P. 1996. Scolopendra abnormis. The IUCN Red List of Threatened Species 1996: e.T20042A9138315. doi:10.2305/IUCN.UK.1996.RLTS.T20042A9138315.en. Accessed on 10 January 2023.
- Macadam, C. 2022. Nothogeophilus turki. The IUCN Red List of Threatened Species 2022: e.T123669218A123674269. doi:10.2305/IUCN.UK.2022-1.RLTS.T123669218A123674269.en. Accessed on 10 January 2023.
- Gerlach, J. 2014. Seychellonema gerlachi. The IUCN Red List of Threatened Species 2014: e.T201495A15280805. doi:10.2305/IUCN.UK.2014-1.RLTS.T201495A15280805.en. Accessed on 10 January 2023.
- Shear, W. A. (1992). "Early life on land". American Scientist. 80 (5): 444–456. Bibcode:1992AmSci..80..444G.
- Jeram, Andrew J.; Selden, Paul A.; Edwards, Dianne (2 November 1990). "Land Animals in the Silurian: Arachnids and Myriapods from Shropshire, England" (PDF). Science. 250 (4981): 658–661. Bibcode:1990Sci...250..658J. doi:10.1126/science.250.4981.658. PMID 17810866.
- Heather M. Wilson (2003). "A new scolopendromorph centipede (Myriapoda: Chilopoda) from the Lower Cretaceous (Aptian) of Brazil". Journal of Paleontology. 77 (1): 73–77. doi:10.1666/0022-3360(2003)077<0073:ANSCMC>2.0.CO;2. JSTOR 4094718. S2CID 130297755.
- Shear, William A.; Bonamo, Patricia M. (1990). "Fossil centipedes from the Devonian of New York State, U.S.A.". In Minelli, Alessandro (ed.). Proceedings of the 7th International Congress of Myriapodology. Brill Publishers. pp. 89–96. ISBN 978-90-04-08972-3.
- Wilson, Heather M. (2001). "First Mesozoic Scutigeromorph Centipede, from the Lower Cretaceous of Brazil". Palaeontology. Wiley. 44 (3): 489–495. Bibcode:2001Palgy..44..489W. doi:10.1111/1475-4983.00188. ISSN 0031-0239.
- Lozano-Fernandez, Jesus; Giacomelli, Mattia; Fleming, James F. (4 July 2019). "Pancrustacean Evolution Illuminated by Taxon-Rich Genomic-Scale Data Sets with an Expanded Remipede Sampling". Genome Biology and Evolution. 11 (8): 2055–2070. doi:10.1093/gbe/evz097. PMC 6684935. PMID 31270537.
- Giribet, Gonzalo; Edgecombe, Gregory D. (2019). "The Phylogeny and Evolutionary History of Arthropods". Current Biology. 29 (12): R592–R602. doi:10.1016/j.cub.2019.04.057. PMID 31211983. S2CID 189926344.
- Edgecombe, G. D.; Giribet, G. (2002). "Myriapod phylogeny and the relationships of Chilopoda" (PDF). In Bousquets, J. Llorente; Morrone, J. J. (eds.). Biodiversidad, Taxonomía y Biogeografia de Artrópodos de México: Hacia una Síntesis de su Conocimiento, Volumen III. Universidad Nacional Autónoma de México. pp. 143–168.
- Svetnik, Ilja (2019). Red List and DNA barcoding of Carinthian and Styrian centipedes (Chilopoda) (Thesis).
- Shelley, Rowland M. (March 1999). "Centipedes and Millipedes with Emphasis on North America Fauna". Kansas School Naturalist. Emporia State University. 45 (3). ISSN 0022-877X. Archived from the original on 12 Nov 2016.
- Undheim, E. A.; Jenner, R. A. (2021). "Phylogenetic analyses suggest centipede venom arsenals were repeatedly stocked by horizontal gene transfer". Nature Communications. 12 (1): 818. Bibcode:2021NatCo..12..818U. doi:10.1038/s41467-021-21093-8. PMC 7864903. PMID 33547293.
- Pak, Mijune. "Eating Bugs/Insects in Donghuamen Night Market!". Follow me Foodie. Retrieved 14 November 2022.
- "101 Strangest Foods Around the World". Archived from the original on 2016-11-16. Retrieved 2015-06-28.
- Monks, Kieron (27 July 2015). "Centipede vodka and fried crickets: Is this the future of food?". CNN. Retrieved 14 November 2022.
- Bush, Sean P.; King, Bradley O.; Norris, Robert L.; Stockwell, Scott A. (2001). "Centipede envenomation". Wilderness & Environmental Medicine. 12 (2): 93–99. doi:10.1580/1080-6032(2001)012[0093:CE]2.0.CO;2. PMID 11434497.
- Jacobs, Steven J. (19 October 2009). House Centipede (PDF). Pennsylvania State University. Archived from the original (PDF) on 9 October 2011. Retrieved 26 July 2012.
In 1902, C.L. Marlatt, an entomologist with the United States Department of Agriculture writes in Circular #48 - The House Centipede: It may often be seen darting across floors with great speed, occasionally stopping suddenly and remaining absolutely motionless, presently to resume its rapid movements, often darting directly at residents, particularly women, evidently with a desire to conceal itself beneath their dresses, creating much consternation.
- Zabs-Dkar Tshogs-Drug-Ran-Grol. The Life of Shabkar: The Autobiography of a Tibetan Yogin. Albany: SUNY Press, 1994. p. 295.
Sources
- Lewis, J. G. E. (2007). The Biology of Centipedes. Cambridge University Press. ISBN 978-0-521-03411-1.
External links
- Chilobase, a web resource for Chilopoda taxonomy Archived 2017-11-06 at the Wayback Machine
- Debunking of some centipede myths, American Tarantula Society
- Centipedes of Australia
- Chilopoda Archived 2009-04-16 at the Wayback Machine, Tree of Life Web Project
- What do you call a centipede?

