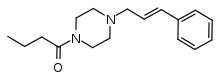
Bucinnazine
Bucinnazine (AP-237, 1-butyryl-4-cinnamylpiperazine) is an opioid analgesic drug that was widely used in China to treat pain in cancer patients as of 1986.[1] It is one of the most potent compounds among a series of piperazine-amides first synthesized and reported in Japan in the 1970s.[2][3][4] Bucinnazine has analgesic potency comparable to that of morphine but with a relatively higher therapeutic index.
 | |
| Clinical data | |
|---|---|
| Other names | AP-237 |
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C17H24N2O |
| Molar mass | 272.392 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
The drug was initially claimed to be a non-narcotic analgesic. However, subsequent studies have shown bucinnazine and similar acyl piperazines to be potent and selective agonists of μ-opioid receptor (MOR) with relatively low affinity for the δ-opioid receptor and the κ-opioid receptor.[5] In accordance with these studies, results from the intravenous self-administration experiments in rats showed that bucinnazine has a marked reinforcing effect with tolerance and dependence quickly developing.[1] In addition, the morphine antagonist naloxone reverses the effect of bucinnazine and precipitates withdrawal symptoms in bucinnazine treated rats further indicating a mechanism of analgesia mediated via selective agonist activity at μ-opioid receptors.
Derivatives
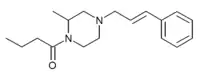
2-methyl-AP-237 has been sold on the grey market as a designer opioid, first identified by a police forensic laboratory in Slovenia in March 2019.[6][7][8]
See also
References
- Qing T, Zhi-Ji C, Wei-Ping W (1986). "Experimental Study on the Dependence-Producing Properties of Qiang Tong Ding (AP-237)". Chin. J. Clin. Pharmacol. (2).
- Nishimura, N.; Kiuchi, M.; Kanetake, Y.; Takahashi, T. (1970). "Clinical exaluation of a new analgesic agent Ap-237". Masui. The Japanese Journal of Anesthesiology. 19 (6): 653–6. PMID 4916908.
- Carrano, R. A.; Kimura, K. K.; McCurdy, D. H. (1975). "Analgesic and tolerance studies with AP-237, a new analgesic". Archives Internationales de Pharmacodynamie et de Therapie. 213 (1): 41–57. PMID 1156018.
- Carrano, R. A.; Kimura, K. K.; Landes, R. C.; McCurdy, D. H. (1975). "General pharmacology of a new analgesic-AP-237". Archives Internationales de Pharmacodynamie et de Therapie. 213 (1): 28–40. PMID 1156016.
- Barlocco, D.; Cignarella, G.; Greco, G.; Novellino, E. (1993). "Computer-aided structure-affinity relationships in a set of piperazine and 3,8-diazabicyclo3.2.1octane derivatives binding to the mu-opioid receptor". Journal of Computer-Aided Molecular Design. 7 (5): 557–71. Bibcode:1993JCAMD...7..557B. doi:10.1007/bf00124362. PMID 8294946. S2CID 23360530.
- Analytical Report 2-Methyl-AP-237. 19 March 2019
- Fogarty, Melissa F.; Vandeputte, Marthe M.; Krotulski, Alex J.; Papsun, Donna; Walton, Sara E.; Stove, Christophe P.; Logan, Barry K. (2022). "Toxicological and pharmacological characterization of novel cinnamylpiperazine synthetic opioids in humans and in vitro including 2-methyl AP-237 and AP-238". Archives of Toxicology. doi:10.1007/s00204-022-03257-7. ISSN 1432-0738. PMID 35275255.
- Giorgetti, A.; Brunetti, P.; Pelotti, S.; Auwärter, V. (August 2022). "Detection of AP‐237 and synthetic cannabinoids on an infused letter sent to a German prisoner". Drug Testing and Analysis. doi:10.1002/dta.3351. ISSN 1942-7603. PMID 35918775.