Deferiprone
Deferiprone, sold under the brand name Ferriprox among others, is a medication that chelates iron and is used to treat iron overload in thalassaemia major.[3] It was first approved and indicated for use in treating thalassaemia major in 1994[4] and had been licensed for use in the European Union for many years while awaiting approval in Canada and in the United States.[3] On October 14, 2011, it was approved for use in the US under the FDA's accelerated approval program.[5][6]
 | |
| Clinical data | |
|---|---|
| Trade names | Ferriprox |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a612016 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Glucuronidation |
| Elimination half-life | 2 to 3 hours |
| Excretion | Kidney (75 to 90% in 24 hours) |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.157.470 |
| Chemical and physical data | |
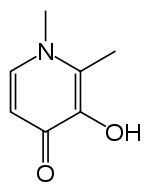
| Formula | C7H9NO2 |
| Molar mass | 139.154 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
The most common side effects include red-brown urine (showing that iron is being removed through the urine), nausea (feeling sick), abdominal pain (stomach ache) and vomiting.[7][5] Less common but more serious side effects are agranulocytosis (very low levels of granulocytes, a type of white blood cell) and neutropenia (low levels of neutrophils, a type of white blood cell that fights infections).[7][5]
Medical uses
Deferiprone monotherapy is indicated in the European Union for the treatment of iron overload in those with thalassaemia major when current chelation therapy is contraindicated or inadequate.[7]
Deferiprone in combination with another chelator is indicated in the European Union in those with thalassaemia major when monotherapy with any iron chelator is ineffective, or when prevention or treatment of life-threatening consequences of iron overload (mainly cardiac overload) justifies rapid or intensive correction.[7]
Controversy
Deferiprone was at the center of a protracted struggle between Nancy Olivieri, a Canadian haematologist and researcher, and the Hospital for Sick Children and the pharmaceutical company Apotex, that started in 1996, and delayed approval of the drug in North America.[8] Olivieri's data suggested deferiprone leads to progressive hepatic fibrosis.[9][10][11]
History
Deferiprone was approved for medical use in the European Union in August 1999.[7]
It was approved for medical use in the United States in October 2011.[5][6] Generic versions were approved in August 2019.[12]
The safety and effectiveness of deferiprone is based on an analysis of data from twelve clinical studies in 236 participants.[5] Participants in the study did not respond to prior iron chelation therapy.[5] Deferiprone was considered a successful treatment for participants who experienced at least a 20 percent decrease in serum ferritin, a protein that stores iron in the body for later use.[5] Half of the participants in the study experienced at least a 20 percent decrease in ferritin levels.[5]
References
- "Deferiprone (Ferriprox) Use During Pregnancy". Drugs.com. 30 March 2020. Retrieved 20 May 2020.
- "Ferriprox 100 mg/ml oral solution - Summary of Product Characteristics (SmPC)". (emc). 26 November 2019. Retrieved 20 May 2020.
- Savulescu J (February 2004). "Thalassaemia major: the murky story of deferiprone". BMJ. 328 (7436): 358–9. doi:10.1136/bmj.328.7436.358. PMC 341373. PMID 14962851.
- Staff. "Cipla's History". Cipla. Archived from the original on 2015-10-27.
- "FDA Approves Ferripox (deferiprone) to Treat Patients with Excess Iron in the Body". U.S. Food and Drug Administration (FDA) (Press release). 14 October 2011. Archived from the original on 10 October 2016.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - "Drug Approval Package: Ferriprox (deferiprone) Tablet NDA #021825". U.S. Food and Drug Administration (FDA). 30 November 2011. Retrieved 20 May 2020.
- "Ferriprox EPAR". European Medicines Agency (EMA). Retrieved 20 May 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- Viens AM, Savulescu J (February 2004). "Introduction to The Olivieri symposium". Journal of Medical Ethics. 30 (1): 1–7. doi:10.1136/jme.2003.006577. PMC 1757126. PMID 14872065.
- Brittenham GM, Nathan DG, Olivieri NF, Porter JB, Pippard M, Vichinsky EP, Weatherall DJ (June 2003). "Deferiprone and hepatic fibrosis". Blood. 101 (12): 5089–90, author reply 5090–1. doi:10.1182/blood-2002-10-3173. PMID 12788794.
- Wanless IR, Sweeney G, Dhillon AP, Guido M, Piga A, Galanello R, et al. (September 2002). "Lack of progressive hepatic fibrosis during long-term therapy with deferiprone in subjects with transfusion-dependent beta-thalassemia". Blood. 100 (5): 1566–9. doi:10.1182/blood-2002-01-0306. PMID 12176871.
- Cribb R (2019-02-27). "UHN patients given unlicensed drug that led to diabetes, liver dysfunction and one death, study finds". The Star. Toronto. Retrieved 2019-02-27.
- "Deferiprone: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Retrieved 20 May 2020.
External links
- "Deferiprone". Drug Information Portal. U.S. National Library of Medicine.