Minimed Paradigm
MiniMed Paradigm is a series of insulin pumps manufactured by Medtronic for patients with diabetes mellitus. The pump operates with a single AA battery and uses a piston-plunger pump to infuse a programmed amount of insulin into the patient through a length of tubing. The Paradigm uses a one-way wireless radio frequency link to receive blood sugar measurements from select glucose meters. The Paradigm RT (Real Time) series adds the ability to receive data from a mated continuous blood-glucose monitor. Although the pump can use these measurements to assist in calculating a dose of insulin, no actual change in insulin delivery occurs without manual user-intervention. [1][2]

In the United States, the device is regulated by a branch of the Food and Drug Administration.[3]
Description
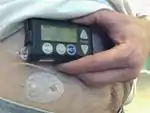
Insulin pumps are drug delivery devices used to treat patients with type 1 and type 2 diabetes. The Minimed Paradigm REAL-Time and Continuous Glucose Monitoring (CGM) system, which received FDA clearance in 2006, uses tubing and a reservoir with rapid-acting insulin. This "infusion set" is patient-connected via a catheter to the abdomen region. The infusion set can remain in the place for three days while the pump is clip-belt worn. There is a quick-disconnect feature for the tubing. The pump delivers insulin in two modes. In Basal rate mode, the delivery is continuous in small doses similar to a pancreas, for example 0.15 units per hour throughout the day. Basal rates are set to meet individual metabolic rates. In Bolus mode, the delivery is programmed to be a one-time delivery prior to eating or after an unexpected high, for example 18 units spread out to several hours. This type of continuous treatment is in contrast to traditional multiple daily injections (MDI) that use slower-acting insulin. Continuous treatment reduces glucose variability.[4]
The Paradigm system consists of two basic parts: an insulin pump and an optional glucose sensor CGM worn for up to 3 days. The disposable sensor is subcutaneously-placed to make glucose measurements[5] in interstitial fluid every 5 minutes and transmit the reading via low power radio frequency (ISM band) to the pump for realtime display. However, insulin therapy may be conducted without CGM and although there is not yet an automated insulin-regulation feedback mechanism between measure and infusion to control the amount and timing of insulin, this is clearly a future objective. So any change in basal or bolus is patient-driven by programming the pump using the Bolus Wizard. The latest model pumps are the MiniMed Paradigm 522 and 722 which differ in reservoir size, 176 versus 300 units, respectively. In 2007 the FDA approved a pediatric model for patients 7 to 17 years old.[6]
History
The development history of the Minimed pump goes back to the 1980s.[7]
- 1983 – 1st Pump MiniMed 502 (Eli Lilly makes synthetic insulin)[8]
- 1985 – MiniMed 504 Insulin Pump
- 1992 – Launch Of MiniMed 506 Insulin Pump
- 1996 – Introduction of MiniMed 507 Pump
- 1999 – Launch of the Model 507C
- 1999 – Introduction of MiniMed 508 Insulin Pump
- 2002 – Inauguration of the MiniMed Paradigm 511
- 2003 – 1st Wireless MiniMed Paradigm 512/712 (followed by 515/715)
- 2006 – MiniMed Paradigm REAL-Time 522/722
- 2010 – MiniMed Paradigm REAL-Time Revel 523/723[9]
FDA Classification
The Food and Drug Administration has at least six classifications for the various parts of the Minimed Paradigm System.
| Product code | Generic part name | Regulation number | Device classification code | Description |
|---|---|---|---|---|
| MDS | CGM | null | Premarket approval[10] | Invasive glucose sensor |
| FMF | Drug reservoir | 21CFR888.5860[11] | standards | Piston syringe |
| NBW | Glucose meter | 21CFR862.1345[12] | standards | Glucose test system |
| LZG/FRN | Infusion/insulin pump | 21CFR888.5725[13] | standards | Infusion pump |
| FPA | Infusion set | 21CFR888.5440[14] | standards | Intravascular administration set |
| KZH | Infusion set insertion system | 21CFR880.6920[15] | standards | Syringer needle introducer |
Competitors devices
Competitive devices includes Deltec Cozmo, Animas Ping, Tandem Diabetes Care, Inc., Insulet OmniPod, Accu-chek Spirit Combo, and Sooil DiabecareIIS.[16]
Future devices
Currently, Medtronic has the following research and development projects in its pipeline: Next Generation REAL-Time Continuous Glucose Monitoring System; Next Generation Insulin Pump; Pre-filled Insulin Reservoirs; Implantable Insulin Pump; and Artificial pancreas (Semi-Automated System & Closed-Loop System).[9] The industry trend in portable devices has piggybacked on the success of wireless technology[17] but not on the success of other disciplines, such as dynamical system, Cybernetics and adaptive systems, for root cause solutions
to close the "true" loop.[18] While the slow-responding dynamics in the physiology of glucose regulation is not beyond the mathematics of PID controllers, 50 years of patient-perspective "advancement" says it is beyond the corporate, congressional, and lobbying leaders. Also, the accuracy of existing continuous glucose monitoring systems poses a problem for an artificial pancreas.
References
- ADA - CSII Diabetes Care 2004; 27: S110.
- ADA - Implications of the DCCT study Diabetes Care 2002; 25: 25-27.
- ADA - Implications of the UKPD Study Diabetes Care 2002; 25: 28-32.
- Bell D.S.H., Fernando O. (2000). "Improved glycemic control with use of CSII compared with MDI therapy". Endocrine Practice. 6 (5): 257–360. doi:10.4158/EP.6.5.357. PMID 11141585.
- Binder C., Lauritzen T., Faber O., Pramming S. (1984). "Insulin pharmacokinetics". Diabetes Care. 7 (2): 188–99. doi:10.2337/diacare.7.2.188. PMID 6376015. S2CID 29287604.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Bode B.W., Steed R.D., Davidson P.C. (1996). "Reduction in severe hypoglycemia with longterm CSII in type 1 diabetes". Diabetes Care. 19 (4): 324–7. doi:10.2337/diacare.19.4.324. PMID 8729154. S2CID 29779558.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Bode BW; et al. (1999). "Continuous glucose monitoring used to adjust diabetes therapy improves glycosylated hemoglobin: A pilot study". Diabetes Research and Clin Practice. 46 (3): 183–90. doi:10.1016/S0168-8227(99)00113-8. PMID 10624783. Diabetes Technol Ther. 2004;6(2):105-13.
- Boland E.A., Grey M.; et al. (1999). "CSII - a new way to lower risk of severe hypoglycemia, improve metabolic control, and enhance coping in adolescents with type 1 diabetes". Diabetes Care. 22 (11): 1779–84. doi:10.2337/diacare.22.11.1779. PMID 10546007.
- Carlton F.B. (2000). "Recent advances in the pharmacologic management of diabetes mellitus". Emergency Medicine Clinics of North America. 18 (4): 745–53. doi:10.1016/S0733-8627(05)70156-5. PMID 11130936.
- Chantelau E., Schiffers T., Schutze J., Hansen B. (1997). "Effect of patient-selected intensive insulin therapy on quality of life". Patient Education and Counseling. 38 (2): 167–713. doi:10.1016/S0738-3991(96)00964-0. PMID 9128618.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Crawford L.M., Sinha R.N., Odell R.M., Comi R.J. (2000). "Efficacy of insulin pump therapy: mealtime delivery is the key factor". Endocrine Practice. 6 (3): 239–43. doi:10.4158/EP.6.3.239. PMID 11421538.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - The DCCT Research Group. JAMA. 1997;227(5):374-4 Skyler J.S. Diabtes Technol Ther. 2000.
- The DCCT Research Group. Progression of retinopathy with intensive versus conventional treatment in the diabetes control and complications trial. 1994.
- The DCCT/EDIC Study Research Group. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. NEJM 2005; 353(25): 2643-2653.
- The Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Report of the expert committee on the diagnosis and classification of diabetes mellitus Diabetes Care 2001; 24: 1.
- Kaufman FR (2001). "A Pilot Study of the Continuous Glucose Monitoring System: Clinical decisions and glycemic control after its use in pediatric type 1 diabetic subjects". Diabetes Care. 24 (12): 2030–4. doi:10.2337/diacare.24.12.2030. PMID 11723078.
- Floyd J.C., Cornell R.G.; et al. (1993). "A prospective study identifying risk factors for discontinuance of insulin pump therapy". Diabetes Care. 16 (11): 1470–8. doi:10.2337/diacare.16.11.1470. PMID 8299436. S2CID 23783836.
- Haakens K., Hanssen K.F.; et al. (1990). "CSII, MDI and conventional insulin therapy in self-selecting insulin-dependent diabetic patients. A comparison of metabolic control acute complications and patient preferences". J Intern Med. 228 (5): 457–464. doi:10.1111/j.1365-2796.1990.tb00263.x. PMID 2254715. S2CID 20197231.
- Ludvigsson J; et al. (2003). "Continuous Subcutaneous Glucose Monitoring Improved Metabolic Control in Pediatric Patients With Type 1 Diabetes: A Controlled Crossover Study". Pediatrics. 111 (5): 933–8. doi:10.1542/peds.111.5.933. PMID 12728068. S2CID 30709714.
- Marcus A.O., Fernandez M.P. (1996). "Insulin pump therapy; acceptable alternative to injecting therapy". Postgraduate Medicine. 99: 3.
- Mudaliar S., Edelman S.V. (2001). "Insulin therapy in type 2 diabetes". Endocrinology and Metabolism Clinics. 39 (4): 935–82. doi:10.1016/s0889-8529(05)70222-x. PMID 11727406.
- Pitzer KR; et al. (2001). "Detection of Hypoglycemia With the GlucoWatch Biographer". Diabetes Care. 24 (5): 881–885. doi:10.2337/diacare.24.5.881. PMID 11347748.
- Tsui E.Y.L, Chiasson J.L; et al. (1998). "Counterregulatory hormone responses after long-term CSII with lispro insulin". Diabetes Care. 21 (1): 93–6. doi:10.2337/diacare.21.1.93. PMID 9538976. S2CID 21217439.
Notes
- "New Diabetes Device Adds Up". Cbsnews.com.
- "Insulin Pumpers Minimed 511 Review". Insulin-pumpers.org.
- "Devices@FDA". Accessdata.fda.gov.
- "Medical and Drug Policies" (PDF).
- "Accuracy of Real-Time Continuous Glucose Monitoring in the MiniMed Paradigm System – DiabetesPro – American Diabetes Association". Archived from the original on 2007-10-18. Retrieved 2009-07-20.
- "Ingentaconnect Insulin Pump Therapy in Childhood Diabetes Mellitus: Guidelines F". Archived from the original on 2011-06-07. Retrieved 2009-07-19.
- "About Us | Medtronic Diabetes". Medtronicdiabetes.com. 14 December 2012.
- "Biotechnology Timeline 1977 - 1999". Archived from the original on 2009-04-25. Retrieved 2009-07-30.
- "Overview | Medtronic". Newsroom.medtronic.com.
- Health, Center for Devices and Radiological (July 9, 2019). "Premarket Approval (PMA)". FDA.
- "CFR - Code of Federal Regulations Title 21". Accessdata.fda.gov.
- "CFR - Code of Federal Regulations Title 21". Accessdata.fda.gov.
- "CFR - Code of Federal Regulations Title 21". Accessdata.fda.gov.
- "CFR - Code of Federal Regulations Title 21". Accessdata.fda.gov.
- "CFR - Code of Federal Regulations Title 21". Accessdata.fda.gov.
- "Comparison of Current Insulin Pumps". Diabetesnet.com. November 22, 2010.
- "Metapress | A Fast Growing Resource for Young Entrepreneurs". December 14, 2017.
- "Patent US7029455 - Devices, systems and methods for patient infusion …". Archived from the original on 24 January 2013.