Salinispora
Salinispora is a genus of obligately aerobic, gram-positive, non-acid-fast bacteria belonging to the family of Micromonosporaceae. They are heterotrophic, non-motile, and obligately grow under high osmotic/ionic-strength conditions.[2][3][4] They are the first identified genus of gram-positive bacteria which has a high osmotic/ionic-strength requirement for survival.[2] They are widely abundant in tropical marine sediments and were first identified in 2002.[2] This genus of bacteria has potential biotechnological significance due to their production of novel secondary metabolites which can be used pharmaceutically.[5]
| Salinispora | |
|---|---|
 | |
| S. arenicola strain TM223-S2 | |
| Scientific classification | |
| Domain: | Bacteria |
| Phylum: | Actinomycetota |
| Class: | Actinomycetia |
| Order: | Micromonosporales |
| Family: | Micromonosporaceae |
| Genus: | Salinispora Maldonado et al. 2005 |
| Type species | |
| Salinispora arenicola Maldonado et al. 2005 | |
| Species[1] | |
| |
There are nine known species that fall within the genus of Salinispora including the better studied S. arenicola, S. tropica, and S. pacifica.[6][7] The clade that initially comprised only S. pacifica was further interrogated through comparative genomic analyses in 2020 to reveal six additional species.[6] The differentiation of these species is likely the result of niche differentiation rather than allopatric speciation due to the species co-occurrence.[8] Despite there being high sequence similarity among Salinispora genomes (>99% 16S rRNA sequence identity), species- and strain-based differences among biosynthetic gene clusters and products have been determined.[9][10]
Characteristics
Salinispora members are gram-positive, filamentous bacteria which form extensively branched hyphae with smooth surfaced spores that can occur in clusters or singles.[4] During sporulation, spores may disseminate from short spore-bearing protrusions (sporophores) or directly from the base (sessile).[3][4] They produce a range of pigments including dark brown, black, orange, and pink.[2] They likely spend a considerable amount of time in the resting stage as spores,[11][12] with a much larger abundance and distribution of spores relative to growing individuals. They have been shown in culture to preferentially grow at the upper sediment layers where blooms at the sediment-seawater interface have been observed.[12]
Salinispora is the first identified gram-positive bacterial genus which requires a high osmotic/ionic-strength environment to survive.[2] However, it has been shown that sodium concentrations experienced in marine environments can be replaced with potassium and lithium.[13] The required ionic-strength differs among Salinispora species, but is likely due to the loss of the mscL transporter in the cell membranes in all species.[13][14]
Species and strains within Salinispora produce a wide variety of secondary metabolites. It is likely that the biosynthetic gene clusters producing these secondary metabolites were initially acquired via horizontal gene transfer, potentially explaining the high sequence similarity among species and strains.[10][15] The wide array of different biosynthetic gene products may have also contributed to niche differentiation. The large production of species/strains’ particular secondary metabolite lends evidence to the importance of them in bacterial survival, and can potentially be used to identify specific species and strains within the genus Salinispora.[16][17]
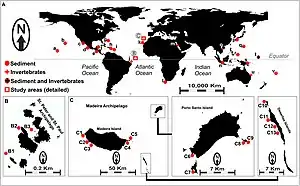
Distribution
Salinispora are commonly found in tropical and subtropical near-shore marine sediments of the Atlantic, Pacific, and Indian oceans.[11][18] They have been detected at depths up to 5699 m (undetermined whether actively growing or spores) and confirmed to be growing at depths up to 1100 m.[11][12] Whether their distribution may include higher latitudes or why they are limited to equatorial regions is not yet known. Additionally, their distribution may not be limited to sediments as they have also been isolated from sponges and seaweeds.
The majority of the S. tropica isolates have been isolated in the Caribbean while the rest have been identified in all three oceans, with S. arenicola being the most geographically abundant.[19]
Pharmaceutical significance
Salinispora has been used as a model for analyzing genome sequence data in order to further uncover biosynthetic pathways among bacteria. This has been an integral part of research into using microbial natural products as leads for the discovery of traditional natural products and potential new drugs.[3] By viewing the evolutionary histories and diversity of the genus, researchers have been able to uncover mechanisms behind the strategies bacteria use to generate chemical diversity and produce diverse secondary metabolites.[3][17] These various new secondary metabolites in Salinispora species have been identified as potentially pharmaceutically valuable. Salinispora species can be distinguished by the natural products that each species produces.[20]
Salinispora arenicola secondary metabolites
Salinispora arenicola secondary metabolites have a broad range of pharmaceutical applications. There are over 20 natural compounds that can be isolated from Salinispora arenicola (e.g., arenamides, arenicolides, arenimycins or salinisporamycin). The potential applications for these compounds are extensive. Over nine compounds are related to cancer cells or cancer precursor inhibition; other compounds have shown antibiotic abilities for resistant bacteria, yeast and fungi. Cycloaspeptides could be a potential treatment for asthma, as it is slightly toxic to lung fibroblasts, the human lung cells responsible for inflammation. Other compounds could be used in treatments for heavy metal poisoning or cholesterol-lowering.[20]
Salinispora pacifica secondary metabolites
Although the list of Salinispora pacifica natural compounds identified is not as extensive as found in S. arenicola, the potential pharmaceutical use of these metabolites is of great interest. Some metabolites are effective at inhibiting cancer growth. Cyanosporasides,[21] for example, were found to be a potential inhibitor of human colon carcinoma. Other metabolites can be used as antioxidants or antibiotics.[20] This species also includes some metabolites that can act as immunosuppressants. Mycalamide A was found to be a potential antiviral, antitumor and even a possible HIV treatment, as it inhibits the activation of the preferred T cell host of HIV.[22] Pacificanones A and B [23] are other metabolites with immunosuppressant capacities and possible applications in the treatment of allergies.
Lomaiviticins A and B
Salinispora pacifica produces a cytotoxic family of secondary metabolites called lomaiviticins. They were the first compounds isolated from the genus Salinispora, with the structures being published in 2001. It was initially reported that the producing bacterial strain was a new Micromonospora species with "Micromonospora lomaivitiensis" as the proposed name. Further gene sequence analysis revealed that the strain was in fact S. pacifica.[5]
Lomaiviticins are aromatic polyketides in the angucycline family and share a diazo group similar to kinamycins.[24] There are two classes of this natural product: lomaiviticin A and lomaiviticin B. Both classes demonstrate potent activity that is damaging to DNA, and is observed to be highly cytotoxic against human cancer cells. Lomaiviticin A in particular generates the greatest activity and is the most abundant of the two.[25]
Due to their unique molecular architecture and biological activities, lomaiviticins are an ideal natural product for chemical synthesis. However, notwithstanding the interest shown by the synthetic chemistry community, total synthesis of lomaiviticins has not yet been achieved and the enzymatic chemistry associated with lomaiviticin assembly has not yet been extensively researched.[26] From studying their distinctive structure, it is theorized that the synthesis of this product will lead to many novel enzymatic transformations. In addition to driving the discovery of new analogs through metabolic engineering or chemoenzymatic synthesis, further research could uncover applicable tools for biocatalysis and metabolic engineering.[24][26]
Salinispora tropica secondary metabolites
The number of secondary metabolites identified and isolated from Salinispora tropica is fewer than the two other species. However, the pharmaceutical and clinical relevance of these metabolites is much higher. As of yet Salinosporamide is the most successful secondary metabolite of Salinisopora from the clinical point of view. It has already been moved into human trials, and it has shown to be a strong anti-cancer agent.[27] Salinispora tropica also produces antiprotealide, another anti-cancer agent which is potentially the strongest cancer inhibitor within the Salinospora secondary metabolite lists. This natural compound is a very potent cytotoxin for myeloma cells.[28]
References
- "Genus Salinispora". List of Prokaryotic names with Standing in Nomenclature (LPSN). Leibniz Institute DSMZ-German Collection of Microorganisms and Cell Cultures GmbH.
- Mincer TJ, Jensen PR, Kauffman CA, Fenical W (October 2002). "Widespread and persistent populations of a major new marine actinomycete taxon in ocean sediments". Applied and Environmental Microbiology. 68 (10): 5005–5011. Bibcode:2002ApEnM..68.5005M. doi:10.1128/aem.68.10.5005-5011.2002. PMC 126404. PMID 12324350.
- Maldonado LA, Fenical W, Jensen PR, Kauffman CA, Mincer TJ, Ward AC, et al. (September 2005). "Salinispora arenicola gen. nov., sp. nov. and Salinispora tropica sp. nov., obligate marine actinomycetes belonging to the family Micromonosporaceae". International Journal of Systematic and Evolutionary Microbiology. 55 (Pt 5): 1759–1766. doi:10.1099/ijs.0.63625-0. PMID 16166663.
- Jensen PR, Maldonado LA, Goodfellow M (2015-09-14). "Salinispora". Bergey's Manual of Systematics of Archaea and Bacteria: 1–10. doi:10.1002/9781118960608.gbm00151. ISBN 9781118960608.
- Jensen PR, Moore BS, Fenical W (May 2015). "The marine actinomycete genus Salinispora: a model organism for secondary metabolite discovery". Natural Product Reports. 32 (5): 738–751. doi:10.1039/C4NP00167B. PMC 4414829. PMID 25730728.
- Román-Ponce B, Millán-Aguiñaga N, Guillen-Matus D, Chase AB, Ginigini JG, Soapi K, et al. (August 2020). "Six novel species of the obligate marine actinobacterium Salinispora, Salinispora cortesiana sp. nov., Salinispora fenicalii sp. nov., Salinispora goodfellowii sp. nov., Salinispora mooreana sp. nov., Salinispora oceanensis sp. nov. and Salinispora vitiensis sp. nov., and emended description of the genus Salinispora". International Journal of Systematic and Evolutionary Microbiology. 70 (8): 4668–4682. doi:10.1099/ijsem.0.004330. PMC 7660244. PMID 32701422.
- Millán-Aguiñaga N, Chavarria KL, Ugalde JA, Letzel AC, Rouse GW, Jensen PR (June 2017). "Phylogenomic Insight into Salinispora (Bacteria, Actinobacteria) Species Designations". Scientific Reports. 7 (1): 3564. Bibcode:2017NatSR...7.3564M. doi:10.1038/s41598-017-02845-3. PMC 5472633. PMID 28620214.
- Jensen PR, Mafnas C (November 2006). "Biogeography of the marine actinomycete Salinispora". Environmental Microbiology. 8 (11): 1881–1888. doi:10.1111/j.1462-2920.2006.01093.x. PMID 17014488.
- Bauermeister A, Velasco-Alzate K, Dias T, Macedo H, Ferreira EG, Jimenez PC, et al. (2018-12-11). "Metabolomic Fingerprinting of Salinispora From Atlantic Oceanic Islands". Frontiers in Microbiology. 9: 3021. doi:10.3389/fmicb.2018.03021. PMC 6297358. PMID 30619120.
- Jensen PR, Williams PG, Oh DC, Zeigler L, Fenical W (February 2007). "Species-specific secondary metabolite production in marine actinomycetes of the genus Salinispora". Applied and Environmental Microbiology. 73 (4): 1146–1152. Bibcode:2007ApEnM..73.1146J. doi:10.1128/AEM.01891-06. PMC 1828645. PMID 17158611.
- Prieto-Davó A, Villarreal-Gómez LJ, Forschner-Dancause S, Bull AT, Stach JE, Smith DC, et al. (June 2013). "Targeted search for actinomycetes from nearshore and deep-sea marine sediments". FEMS Microbiology Ecology. 84 (3): 510–518. doi:10.1111/1574-6941.12082. PMC 3654085. PMID 23360553.
- Mincer TJ, Fenical W, Jensen PR (November 2005). "Culture-dependent and culture-independent diversity within the obligate marine actinomycete genus Salinispora". Applied and Environmental Microbiology. 71 (11): 7019–7028. Bibcode:2005ApEnM..71.7019M. doi:10.1128/AEM.71.11.7019-7028.2005. PMC 1287694. PMID 16269737.
- Tsueng G, Lam KS (May 2010). "A preliminary investigation on the growth requirement for monovalent cations, divalent cations and medium ionic strength of marine actinomycete Salinispora". Applied Microbiology and Biotechnology. 86 (5): 1525–1534. doi:10.1007/s00253-009-2424-7. PMID 20084507. S2CID 26775348.
- Bucarey SA, Penn K, Paul L, Fenical W, Jensen PR (June 2012). "Genetic complementation of the obligate marine actinobacterium Salinispora tropica with the large mechanosensitive channel gene mscL rescues cells from osmotic downshock". Applied and Environmental Microbiology. 78 (12): 4175–4182. Bibcode:2012ApEnM..78.4175B. doi:10.1128/aem.00577-12. PMC 3370544. PMID 22492446.
- Nett M, Moore BS (May 2009). "Exploration and engineering of biosynthetic pathways in the marine actinomycete Salinispora tropica". Pure and Applied Chemistry. 81 (6): 1075–1084. doi:10.1351/PAC-CON-08-08-08. ISSN 1365-3075. S2CID 97624337.
- Bose U, Hewavitharana AK, Vidgen ME, Ng YK, Shaw PN, Fuerst JA, Hodson MP (2014-03-12). Chaturvedi V (ed.). "Discovering the recondite secondary metabolome spectrum of Salinispora species: a study of inter-species diversity". PLOS ONE. 9 (3): e91488. Bibcode:2014PLoSO...991488B. doi:10.1371/journal.pone.0091488. PMC 3951395. PMID 24621594.
- Ziemert N, Lechner A, Wietz M, Millán-Aguiñaga N, Chavarria KL, Jensen PR (March 2014). "Diversity and evolution of secondary metabolism in the marine actinomycete genus Salinispora". Proceedings of the National Academy of Sciences of the United States of America. 111 (12): E1130–E1139. Bibcode:2014PNAS..111E1130Z. doi:10.1073/pnas.1324161111. PMC 3970525. PMID 24616526.
- Jensen PR, Mafnas C (November 2006). "Biogeography of the marine actinomycete Salinispora". Environmental Microbiology. 8 (11): 1881–1888. doi:10.1111/j.1462-2920.2006.01093.x. PMID 17014488.
- Freel KC, Edlund A, Jensen PR (February 2012). "Microdiversity and evidence for high dispersal rates in the marine actinomycete 'Salinispora pacifica'". Environmental Microbiology. 14 (2): 480–493. doi:10.1111/j.1462-2920.2011.02641.x. PMC 3272088. PMID 22117917.
- Kim H, Kim S, Kim M, Lee C, Yang I, Nam SJ (December 2020). "Bioactive natural products from the genus Salinospora: a review". Archives of Pharmacal Research. 43 (12): 1230–1258. doi:10.1007/s12272-020-01288-1. PMID 33237436. S2CID 227166135.
- Kersten RD, Lane AL, Nett M, Richter TK, Duggan BM, Dorrestein PC, Moore BS (May 2013). "Bioactivity-guided genome mining reveals the lomaiviticin biosynthetic gene cluster in Salinispora tropica". ChemBioChem. 14 (8): 955–962. doi:10.1002/cbic.201300147. PMC 3755882. PMID 23649992.
- Yang KX, Ji DS, Zheng H, Gu Y, Xu PF (March 2022). "Organocatalytic inverse-electron-demand Diels-Alder reaction between 5-alkenyl thiazolones and β,γ-unsaturated carbonyl compounds". Chemical Communications. 58 (26): 4227–4230. doi:10.1039/D2CC00457G. PMID 35285468. S2CID 247275008.
- Oh DC, Gontang EA, Kauffman CA, Jensen PR, Fenical W (April 2008). "Salinipyrones and pacificanones, mixed-precursor polyketides from the marine actinomycete Salinispora pacifica". Journal of Natural Products. 71 (4): 570–575. doi:10.1021/np0705155. PMC 2820078. PMID 18321059.
- Janso JE, Haltli BA, Eustáquio AS, Kulowski K, Waldman AJ, Zha L, et al. (July 2014). "Discovery of the lomaiviticin biosynthetic gene cluster in Salinispora pacifica.". Tetrahedron. Tetrahedron Young Investigator Award 2014 A divergent strategy enables the synthesis of three architecturally distinct ent-kauranoids from γ-cyclogeraniol Sarah E. Reisman. 70 (27–28): 4156–4164. doi:10.1016/j.tet.2014.03.009. PMC 4101813. PMID 25045187.
- He H, Ding WD, Bernan VS, Richardson AD, Ireland CM, Greenstein M, et al. (June 2001). "Lomaiviticins A and B, potent antitumor antibiotics from Micromonospora lomaivitiensis". Journal of the American Chemical Society. 123 (22): 5362–5363. doi:10.1021/ja010129o. PMID 11457405.
- Achanta S (2009). Studies toward the synthesis of lomaiviticins A and B and englerin A (Ph.D. thesis). Saint Louis, Mo: Washington University. OCLC 671642940.
- Chauhan D, Catley L, Li G, Podar K, Hideshima T, Velankar M, et al. (November 2005). "A novel orally active proteasome inhibitor induces apoptosis in multiple myeloma cells with mechanisms distinct from Bortezomib". Cancer Cell. 8 (5): 407–419. doi:10.1016/j.ccr.2005.10.013. PMID 16286248.
- Manam RR, Macherla VR, Tsueng G, Dring CW, Weiss J, Neuteboom ST, et al. (February 2009). "Antiprotealide is a natural product". Journal of Natural Products. 72 (2): 295–297. doi:10.1021/np800578e. PMID 19133779.