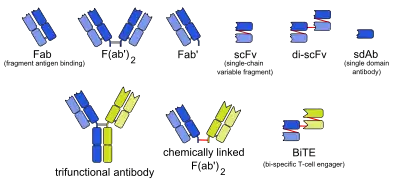
Small modular immunopharmaceutical
Small modular immunopharmaceuticals, or SMIPs for short, are artificial proteins that are intended for use as pharmaceutical drugs. They are largely built from parts of antibodies (immunoglobulins), and like them have a binding site for antigens that could be used for monoclonal antibody therapy. SMIPs have similar biological half-life and, being smaller than antibodies, are reasoned to have better tissue penetration properties. They were invented by Trubion and are now being developed by Emergent BioSolutions, which acquired Trubion in 2010.[1]
Structure
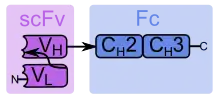
scFv: modified single-chain variable fragment
Fc: one half of a fragment crystallizable region
VH: heavy chain variable domain
VL: light chain variable domain
CH2: constant domain 2
CH3: constant domain 3 (effector domain)
Arrows: linker peptide (left) and hinge region (right)
N: N-terminus
C: C-terminus
SMIPs are single-chain proteins that comprise one binding region, one hinge region as a connector, and one effector domain. The binding region is a modified single-chain variable fragment (scFv), and the rest of the protein can be constructed from the fragment crystallizable region (Fc) and the hinge region of an immunoglobulin G1 (IgG1). Genetically modified cells produce SMIPs as antibody-like dimers, which are about 30% smaller than real antibodies.[2]
Like ordinary monoclonal antibodies, SMIPs are monospecific, meaning they recognize and attach to a single antigen target to initiate their biological activity. SMIP drug candidates are intended to target antigens with the same specificity and predictable biological activity as monoclonal antibodies. Examples are TRU-015, a CD20 targeting SMIP under research for rheumatoid arthritis,[3] and TRU-016, a CD37 targeting potential treatment for chronic lymphocytic leukemia and other B-cell cancers.[4][5]
Production
A monoclonal antibody targeting the desired antigen can be developed the classical way, using hybridoma technology. The scFv is then constructed from the antibody's variable regions. A large number of different hinge regions and effector domains are taken from libraries of immunoglobulins, and the combined proteins are produced in genetically modified (transfected) cells and screened for clones with useful properties like high binding specificity. The selected protein is multiplied in transfected cells suitable for medium- or large-scale production, for example Chinese hamster ovary cells, and purified by chromatography.[2]
References
- TruEmergent: SMIP Therapeutics Archived 2011-08-22 at the Wayback Machine
- Zhao, X.; Lapalombella, R.; Joshi, T.; Cheney, C.; Gowda, A.; Hayden-Ledbetter, M. S.; Baum, P. R.; Lin, T. S.; Jarjoura, D.; Lehman, A.; Kussewitt, D.; Lee, R. J.; Caligiuri, M. A.; Tridandapani, S.; Muthusamy, N.; Byrd, J. C. (2007). "Targeting CD37-positive lymphoid malignancies with a novel engineered small modular immunopharmaceutical". Blood. 110 (7): 2569–2577. doi:10.1182/blood-2006-12-062927. PMC 1988922. PMID 17440052.
- Rubbert-Roth, A. (2010). "TRU-015, a fusion protein derived from an anti-CD20 antibody, for the treatment of rheumatoid arthritis". Current Opinion in Molecular Therapeutics. 12 (1): 115–123. PMID 20140823.
- Robak, T.; Robak, P.; Smolewski, P. (2009). "TRU-016, a humanized anti-CD37 IgG fusion protein for the potential treatment of B-cell malignancies". Current Opinion in Investigational Drugs. 10 (12): 1383–1390. PMID 19943209.
- TruEmergent: TRU-016 for the Treatment of B-Cell Malignancies Archived 2011-08-22 at the Wayback Machine