Phenolphthalein
Phenolphthalein (/fɛˈnɒl(f)θəliːn/ feh-NOL(F)-thə-leen) is a chemical compound with the formula C20H14O4 and is often written as "HIn", "HPh", "phph" or simply "Ph" in shorthand notation. Phenolphthalein is often used as an indicator in acid–base titrations. For this application, it turns colorless in acidic solutions and pink in basic solutions. It belongs to the class of dyes known as phthalein dyes.
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
3,3-Bis(4-hydroxyphenyl)-2-benzofuran-1(3H)-one | |
| Other names
3,3-Bis(4-hydroxyphenyl)isobenzofuran-1(3H)-one | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.914 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C20H14O4 | |
| Molar mass | 318.328 g·mol−1 |
| Appearance | White powder |
| Density | 1.277 g/cm3 (32 °C (90 °F)) |
| Melting point | 258–263 °C (496–505 °F; 531–536 K)[1] |
| 400 mg/L | |
| Solubility in other solvents | Insoluble in benzene and hexane; very soluble in ethanol and ether; slightly soluble in DMSO |
| UV-vis (λmax) | 552 nm (1st) 374 nm (2nd)[1] |
| Pharmacology | |
| A06AB04 (WHO) | |
| Hazards | |
| GHS labelling: | |
 [1] [1] | |
| Danger | |
Hazard statements |
H341, H350, H361[1] |
Precautionary statements |
P201, P281, P308+P313[1] |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Phenolphthalein is slightly soluble in water and usually is dissolved in alcohols for use in experiments. It is a weak acid, which can lose H+ ions in solution. The nonionized phenolphthalein molecule is colorless and the double deprotonated phenolphthalein ion is fuchsia. Further loss of proton in higher pH occurs slowly and leads to a colorless form. Phenolphthalein ion in concentrated sulfuric acid is orange red due to sulfonation.[2]
Uses
pH indicator
Phenolphthalein's common use is as an indicator in acid-base titrations. It also serves as a component of universal indicator, together with methyl red, bromothymol blue, and thymol blue.[3]
Phenolphthalein adopts different forms in aqueous solution depending on the pH of the solution.[4][2][5][6] Inconsistency exists in the literature with regard to the hydrated forms of the compounds and the color in sulfuric acid. Wittke reported in 1983 that under strongly acidic conditions, it exists in protonated form (HIn+), providing an orange coloration. However, a later paper suggested that this color is due to sulfonation to phenolsulfonphthalein.[2]
Between strongly acidic and slightly basic conditions, the lactone form (HIn) is colorless. The doubly deprotonated (In2-) phenolate form (the anion form of phenol) gives the familiar pink color. In strongly basic solutions, phenolphthalein is converted to its In(OH)3− form, and its pink color undergoes a rather slow fading reaction[6] and becomes completely colorless when pH is greater than 13.
The pKA values of phenolphthalein was found to be 9.05, 9.50 and 12 while that of phenolsulfonphthalein are 1.2 and 7.70.[2]
| Species | H3In+ | H2In | In2− | In(OH)3− |
|---|---|---|---|---|
| Structure |  |  |  |  |
| Model |  | 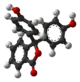 |  | 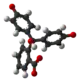 |
| pH | <−1 in H2SO4[4] | 0−8.3 | 8.3−10.0[7] | >10.0 |
| Conditions | strongly acidic | acidic or near-neutral | basic | strongly basic |
| Color | orange | colorless | pink to fuchsia | colorless |
| Image |  |  |
 |
| An animation of the pH dependent reaction mechanism: H3In+ → H2In → In2− → In(OH)3− |
Concrete carbonation
Phenolphthalein's pH sensitivity is exploited in other applications: concrete has naturally high pH due to the calcium hydroxide formed when Portland cement reacts with water. As the concrete reacts with carbon dioxide in the atmosphere, pH decreases to 8.5-9. When a 1% phenolphthalein solution is applied to normal concrete, it turns bright pink. However, if it remains colorless, it shows that the concrete has undergone carbonation. In a similar application, some spackling used to repair holes in drywall contains phenolphthalein. When applied, the basic spackling material retains a pink color; when the spackling has cured by reaction with atmospheric carbon dioxide, the pink color fades.[8]
Education
In a highly basic solution, phenolphthalein's slow change from pink to colorless as it is converted to its In(OH)3− form is used in chemistry classes for the study of reaction kinetics.
Entertainment
Phenolphthalein is used in toys, for example as a component of disappearing inks, or disappearing dye on the "Hollywood Hair" Barbie hair. In the ink, it is mixed with sodium hydroxide, which reacts with carbon dioxide in the air. This reaction leads to the pH falling below the color change threshold as hydrogen ions are released by the reaction:
To develop the hair and "magic" graphical patterns, the ink is sprayed with a solution of hydroxide, which leads to the appearance of the hidden graphics by the same mechanism described above for color change in alkaline solution. The pattern will eventually disappear again because of the reaction with carbon dioxide. Thymolphthalein is used for the same purpose and in the same way, when a blue color is desired.[9]
Medical uses
Phenolphthalein has been used for over a century as a laxative, but is now being removed from over-the-counter laxatives[10] because of concerns over carcinogenicity.[11][12] Laxative products formerly containing phenolphalein have often been reformulated to have alternate active ingredients: Feen-a-Mint[13] switched to bisacodyl and Ex-Lax[14] was switched to a senna extract.
Thymolphthalein is a related laxative made from thymol.
Despite concerns regarding its carcinogenicity, the use of phenolphthalein as a laxative is unlikely to cause ovarian cancer.[15] Phenolphthalein has been found to inhibit human cellular calcium influx via store-operated calcium entry (SOCE, see Calcium release activated channel § Structure). This is effected by its inhibiting thrombin and thapsigargin, two activators of SOCE that increase intracellular free calcium.[16]
Phenolphthalein has been added to the European Chemicals Agency's candidate list for Substances of Very High Concern (SVHC).[17]
A reduced form of phenolphthalein, phenolphthalin, which is colorless, is used in a test to identify substances thought to contain blood, commonly known as the Kastle–Meyer test. A dry sample is collected with a swab or filter paper. A few drops of alcohol, then a few drops of phenolphthalin, and finally a few drops of hydrogen peroxide are dripped onto the sample. If the sample contains hemoglobin, it will turn pink immediately upon addition of the peroxide, because of the generation of phenolphthalein. A positive test indicates the sample contains hemoglobin and, therefore, is likely blood. A false positive can result from the presence of substances with catalytic activity similar to hemoglobin. This test is not destructive to the sample; it can be kept and used in further tests. This test has the same reaction with blood from any animal whose blood contains hemoglobin, including almost all vertebrates; further testing would be required to determine whether it originated from a human.
Synthesis
Phenolphthalein can be synthesized by condensation of phthalic anhydride with two equivalents of phenol under acidic conditions. It was discovered in 1871 by Adolf von Baeyer.[18][19][20]

The reaction can also be catalyzed by a mixture of zinc chloride and thionyl chloride.[22]
See also
- Bromothymol blue
- Litmus
- Methyl orange
- pH indicator
- Universal indicator
References
- "Phenolphthalein". Archived from the original on 8 November 2015. Retrieved 7 October 2014.
- Tamura, Zenzo (December 1996). "Spectrophotometric Analysis of the Relationship between Dissociation and Coloration, and of the Structural Formulas of Phenolphthalein in Aqueous Solution". Analytical Sciences. 12 (6): 927–930. doi:10.2116/analsci.12.927.
- "Universal Indicator". ISCID Encyclopedia of Science and Philosophy. Archived from the original on September 25, 2006.
- Wittke, Georg (1 March 1983). "Reactions of phenolphthalein at various pH values". Journal of Chemical Education. 60 (3): 239. Bibcode:1983JChEd..60..239W. doi:10.1021/ed060p239. ISSN 0021-9584.
- Abdel-Hamid, Refat (1 September 1998). "The Adsorption Behavior of Phenolphthalein at a Mercury Electrode in Water-Ethanol Solutions". Monatshefte für Chemie - Chemical Monthly. 129 (8): 817–826. doi:10.1007/PL00013491. S2CID 92339258.
- Kunimoto, Ko-Ki (February 2001). "Molecular structure and vibrational spectra of phenolphthalein and its dianion". Spectrochimica Acta Part A. 57 (2): 265–271. Bibcode:2001AcSpA..57..265K. doi:10.1016/S1386-1425(00)00371-1. PMID 11206560.
- Rathod, Balraj B.; Murthy, Sahana; Bandyopadhyay, Subhajit (20 February 2019). "Is this Solution Pink Enough? A Smartphone Tutor to Resolve the Eternal Question in Phenolphthalein-Based Titration". Journal of Chemical Education. 96 (3): 486–494. Bibcode:2019JChEd..96..486R. doi:10.1021/acs.jchemed.8b00708. S2CID 104475433.
- US 6531528, Ronald D. Kurp, "Ready to use spackle/repair product containing dryness indicator", published 2003-03-11, assigned to Dap Products Inc.
- "Toystore" (PDF). Archived (PDF) from the original on 2021-07-02. Retrieved 2022-02-22.
- Spiller, H. A.; Winter, M. L.; Weber, J. A.; Krenzelok, E. P.; Anderson, D. L.; Ryan, M. L. (May 2003). "Skin Breakdown and Blisters from Senna-Containing Laxatives in Young Children". The Annals of Pharmacotherapy. 37 (5): 636–639. doi:10.1345/aph.1C439. PMID 12708936. S2CID 31837142.
- Dunnick, J. K.; Hailey, J. R. (1996). "Phenolphthalein Exposure Causes Multiple Carcinogenic Effects in Experimental Model Systems" (PDF). Cancer Research. 56 (21): 4922–4926. PMID 8895745. Archived (PDF) from the original on 2022-02-17. Retrieved 2022-02-22.
- Tice, R. R.; Furedi-Machacek, M.; Satterfield, D.; Udumudi, A.; Vasquez, M.; Dunnick, J. K. (1998). "Measurement of Micronucleated Erythrocytes and DNA Damage during Chronic Ingestion of Phenolphthalein in Transgenic Female Mice Heterozygous for the p53 Gene". Environmental and Molecular Mutagenesis. 31 (2): 113–124. doi:10.1002/(SICI)1098-2280(1998)31:2<113::AID-EM3>3.0.CO;2-N. PMID 9544189.
- "Phenolphthalein". Archived from the original on 2020-08-06. Retrieved 2020-04-25.
- Stolberg, Sheryl Gay (August 30, 1997). "3 Versions of Ex-Lax Are Recalled After F.D.A. Proposes Ban on Ingredient". Archived from the original on May 7, 2021. Retrieved April 25, 2020.
- Cooper, G. S.; Longnecker, M. P.; Peters, R. K. (2004). "Ovarian Cancer Risk and Use of Phenolphthalein-Containing Laxatives". Pharmacoepidemiology and Drug Safety. 13 (1): 35–39. doi:10.1002/pds.824. PMID 14971121. S2CID 24783082. Archived from the original on 2022-01-26. Retrieved 2019-06-26.
- Dobrydneva, Y.; Wilson, E.; Abelt, C. J.; Blackmore, P. F. (2009). "Phenolphthalein as a Prototype Drug for a Group of Structurally Related Calcium Channel Blockers in Human Platelets". Journal of Cardiovascular Pharmacology. 53 (3): 231–240. doi:10.1097/FJC.0b013e31819b5494. PMID 19247192. S2CID 21833487.
- "Phenolphthalein - Substance Information - ECHA". echa.europa.eu. Archived from the original on 2018-06-12. Retrieved 2017-11-06.
- Baeyer, A. (1871). "Ueber eine neue Klasse von Farbstoffen". Berichte der Deutschen Chemischen Gesellschaft. 4 (2): 555–558. doi:10.1002/cber.18710040209. Archived from the original on 2021-02-14. Retrieved 2020-09-02.
- Baeyer, A. (1871). "Ueber die Phenolfarbstoffe". Berichte der Deutschen Chemischen Gesellschaft. 4 (2): 658–665. doi:10.1002/cber.18710040247. Archived from the original on 2021-10-11. Retrieved 2019-06-26.
- Baeyer, A. (1871). "Ueber die Phenolfarbstoffe". Polytechnisches Journal. 201 (89): 358–362. Archived from the original on 2021-10-11. Retrieved 2022-02-22.
- Max Hubacher, U.S. Patent 2,192,485 (1940 to Ex Lax Inc)
- U.S. Patent 2,522,939
