Concept
Version 10
Created by Boundless
Bond Order
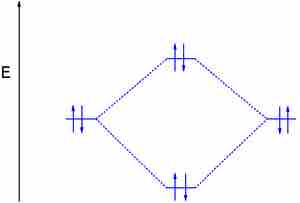
The electron configuration of dihelium
If the molecule He2 were to exist, the 4s electrons would have to fully occupy both the bonding and antibonding levels, giving a bond order of zero. Dihelium does not exist.
Source
Boundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources: