Carbon tetrachloride
| |||
 | |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Tetrachloromethane | |||
| Other names
Benziform benzinoform carbon chloride carbon tet. Freon-10 Refrigerant-10 Halon-104 methane tetrachloride methyl tetrachloride perchloromethane Tetraform Tetrasol TCM | |||
| Identifiers | |||
CAS Number |
|||
3D model (JSmol) |
|||
Beilstein Reference |
1098295 | ||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.239 | ||
| EC Number |
| ||
Gmelin Reference |
2347 | ||
| KEGG | |||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1846 | ||
CompTox Dashboard (EPA) |
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula |
CCl4 | ||
| Molar mass | 153.81 g/mol | ||
| Appearance | Colourless liquid | ||
| Odor | Sweet, chloroform-like odor | ||
| Density |
| ||
| Melting point | −22.92 °C (−9.26 °F; 250.23 K) | ||
| Boiling point | 76.72 °C (170.10 °F; 349.87 K) | ||
Solubility in water |
| ||
| Solubility | Soluble in alcohol, ether, chloroform, benzene, naphtha, CS2, formic acid | ||
| log P | 2.64 | ||
| Vapor pressure | 11.94 kPa at 20 °C | ||
Henry's law constant (kH) |
2.76×10−2 atm·m3/mol | ||
Magnetic susceptibility (χ) |
−66.60×10−6 cm3/mol | ||
| Thermal conductivity | 0.1036 W/m·K (300 K)[1] | ||
Refractive index (nD) |
1.4607 | ||
| Viscosity | 0.86 mPa·s[2] | ||
Dipole moment |
0 D | ||
| Structure | |||
Crystal structure |
Monoclinic | ||
Coordination geometry |
Tetragonal | ||
Molecular shape |
Tetrahedral | ||
Dipole moment |
0 D | ||
| Thermochemistry | |||
Heat capacity (C) |
132.6 J/mol·K | ||
Std molar entropy (S |
214.42 J/mol·K | ||
Std enthalpy of formation (ΔfH⦵298) |
−139.3 kJ/mol | ||
Gibbs free energy (ΔfG˚) |
−686 kJ/mol | ||
| Hazards | |||
| GHS labelling: | |||
Pictograms |
   | ||
Signal word |
Danger | ||
Hazard statements |
H301, H311, H331, H351, H372, H412, H420 | ||
Precautionary statements |
P201, P202, P260, P261, P264, P270, P271, P273, P280, P281, P301+P310, P302+P352, P304+P340, P308+P313, P311, P312, P314, P321, P322, P330, P361, P363, P403+P233, P405, P501, P502 | ||
| NFPA 704 (fire diamond) | |||
| Flash point | <982 °C | ||
Autoignition temperature |
982 °C (1,800 °F; 1,255 K) | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
2350 mg/kg | ||
LC50 (median concentration) |
| ||
LCLo (lowest published) |
| ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
TWA 10 ppm C 25 ppm 200 ppm (5-minute maximum peak in any 4 hours)[4] | ||
REL (Recommended) |
Ca ST 2 ppm (12.6 mg/m3) [60-minute][4] | ||
IDLH (Immediate danger) |
200 ppm[4] | ||
| Safety data sheet (SDS) | ICSC 0024 | ||
| Related compounds | |||
Other anions |
Carbon tetrafluoride Carbon tetrabromide Carbon tetraiodide | ||
Other cations |
Silicon tetrachloride Germanium tetrachloride Tin tetrachloride Lead tetrachloride | ||
Related chloromethanes |
Chloromethane Dichloromethane Chloroform | ||
| Supplementary data page | |||
| Carbon tetrachloride (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Carbon tetrachloride, also known by many other names (such as tetrachloromethane, also recognised by the IUPAC, carbon tet in the cleaning industry, Halon-104 in firefighting, and Refrigerant-10 in HVACR) is an organic compound with the chemical formula CCl4. It is a colourless liquid with a "sweet" smell that can be detected at low levels. It is practically not flammable at lower temperatures. It was formerly widely used in fire extinguishers, as a precursor to refrigerants and as a cleaning agent, but has since been phased out because of environmental and safety concerns. Exposure to high concentrations of carbon tetrachloride (including vapor) can affect the central nervous system and degenerate the liver and kidneys. Prolonged exposure can be fatal.
Properties
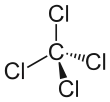
In the carbon tetrachloride molecule, four chlorine atoms are positioned symmetrically as corners in a tetrahedral configuration joined to a central carbon atom by single covalent bonds. Because of this symmetric geometry, CCl4 is non-polar. Methane gas has the same structure, making carbon tetrachloride a halomethane. As a solvent, it is well suited to dissolving other non-polar compounds such as fats, and oils. It can also dissolve iodine. It is somewhat volatile, giving off vapors with a smell characteristic of other chlorinated solvents, somewhat similar to the tetrachloroethylene smell reminiscent of dry cleaners' shops.
Solid tetrachloromethane has two polymorphs: crystalline II below −47.5 °C (225.6 K) and crystalline I above −47.5 °C.[5] At −47.3 °C it has monoclinic crystal structure with space group C2/c and lattice constants a = 20.3, b = 11.6, c = 19.9 (.10−1 nm), β = 111°.[6]
With a specific gravity greater than 1, carbon tetrachloride will be present as a dense nonaqueous phase liquid if sufficient quantities are spilled in the environment.
History and synthesis
Carbon tetrachloride was originally synthesized by the French chemist Henri Victor Regnault in 1839 by the reaction of chloroform with chlorine,[7] but now it is mainly produced from methane:
- CH4 + 4 Cl2 → CCl4 + 4 HCl
The production often utilizes by-products of other chlorination reactions, such as from the syntheses of dichloromethane and chloroform. Higher chlorocarbons are also subjected to "chlorinolysis":
- C2Cl6 + Cl2 → 2 CCl4
Prior to the 1950s, carbon tetrachloride was manufactured by the chlorination of carbon disulfide at 105 to 130 °C:[8]
- CS2 + 3Cl2 → CCl4 + S2Cl2
The production of carbon tetrachloride has steeply declined since the 1980s due to environmental concerns and the decreased demand for CFCs, which were derived from carbon tetrachloride. In 1992, production in the U.S./Europe/Japan was estimated at 720,000 tonnes.[8]
Safety
Carbon tetrachloride is one of the most potent hepatotoxins (toxic to the liver), so much so that it is widely used in scientific research to evaluate hepatoprotective agents.[9][10] Exposure to high concentrations of carbon tetrachloride (including vapor) can affect the central nervous system and degenerate the liver[10] and kidneys,[11] and prolonged exposure may lead to coma or death.[12] Chronic exposure to carbon tetrachloride can cause liver[13][14] and kidney damage and could result in cancer.[15] See safety data sheets.[16]
The effects of carbon tetrachloride on human health and the environment have been assessed under REACH in 2012 in the context of the substance evaluation by France.[17]
In 2008, a study of common cleaning products found the presence of carbon tetrachloride in "very high concentrations" (up to 101 mg/m3) as a result of manufacturers' mixing of surfactants or soap with sodium hypochlorite (bleach).[18]
Carbon tetrachloride is also both ozone-depleting[19] and a greenhouse gas.[20] However, since 1992[21] its atmospheric concentrations have been in decline for the reasons described above (see atmospheric concentration graphs in the gallery). CCl4 has an atmospheric lifetime of 85 years.[22]
At high temperatures in air, it decomposes or burns to produce poisonous phosgene.
Toxicological studies
Carbon tetrachloride is a suspected human carcinogen based on sufficient evidence of carcinogenicity from studies in experimental animals.[23] World Health Organization reports, carbon tetrachloride can induce hepatomas and hepatocellular carcinomas in mice and rats. The doses inducing hepatic tumours are higher than those inducing cell toxicity.[24] The International Agency for Research on Cancer (IARC) classified this compound in Group 2B, possibly carcinogenic to humans.[25]
Uses
In organic chemistry, carbon tetrachloride serves as a source of chlorine in the Appel reaction.
One specialty use of carbon tetrachloride is in stamp collecting, to reveal watermarks on postage stamps without damaging them. A small amount of the liquid is placed on the back of a stamp, sitting in a black glass or obsidian tray. The letters or design of the watermark can then be seen clearly.
Historical uses
Carbon tetrachloride was widely used as a dry cleaning solvent, as a refrigerant, and in lava lamps.[26] In the last case, carbon tetrachloride is a key ingredient that adds weight to the otherwise buoyant wax.
Solvent
It once was a popular solvent in organic chemistry, but, because of its adverse health effects, it is rarely used today.[9] It is sometimes useful as a solvent for infrared spectroscopy, because there are no significant absorption bands above 1600 cm−1. Because carbon tetrachloride does not have any hydrogen atoms, it was historically used in proton NMR spectroscopy. In addition to being toxic, its dissolving power is low.[27] Its use in NMR spectroscopy has been largely superseded by deuterated solvents. Use of carbon tetrachloride in determination of oil has been replaced by various other solvents, such as tetrachloroethylene.[9] Because it has no C–H bonds, carbon tetrachloride does not easily undergo free-radical reactions. It is a useful solvent for halogenations either by the elemental halogen or by a halogenation reagent such as N-bromosuccinimide (these conditions are known as Wohl–Ziegler bromination).
Fire suppression


In 1910, the Pyrene Manufacturing Company of Delaware filed a patent to use carbon tetrachloride to extinguish fires.[28] The liquid was vaporized by the heat of combustion and extinguished flames, an early form of gaseous fire suppression. At the time it was believed the gas simply displaced oxygen in the area near the fire, but later research found that the gas actually inhibits the chemical chain reaction of the combustion process.
In 1911, Pyrene patented a small, portable extinguisher that used the chemical.[29] The extinguisher consisted of a brass bottle with an integrated hand-pump that was used to expel a jet of liquid toward the fire. As the container was unpressurized, it could easily be refilled after use.[30] Carbon tetrachloride was suitable for liquid and electrical fires and the extinguishers were often carried on aircraft or motor vehicles. However as early as 1920, there were reports of fatalities caused by the chemical when used to fight a fire in a confined space.[31]
In the first half of the 20th century, another common fire extinguisher was a single-use, sealed glass globe known as a "fire grenade", filled with either carbon tetrachloride or salt water. The bulb could be thrown at the base of the flames to quench the fire. The carbon tetrachloride type could also be installed in a spring-loaded wall fixture with a solder-based restraint. When the solder melted by high heat, the spring would either break the globe or launch it out of the bracket, allowing the extinguishing agent to be automatically dispersed into the fire.[32]
A well-known brand of fire grenade was the "Red Comet", which was variously manufactured with other fire-fighting equipment in the Denver, Colorado area by the Red Comet Manufacturing Company from its founding in 1919 until manufacturing operations were closed in the early 1980s.[33]
Refrigerants
Prior to the Montreal Protocol, large quantities of carbon tetrachloride were used to produce the chlorofluorocarbon refrigerants R-11 (trichlorofluoromethane) and R-12 (dichlorodifluoromethane). However, these refrigerants play a role in ozone depletion and have been phased out. Carbon tetrachloride is still used to manufacture less destructive refrigerants. Carbon tetrachloride made from heavy chlorine-37 has been used in the detection of neutrinos.
Fumagant
Carbon tetrachloride was widely used as an fumigant to kill insect pests in stored grain.[34] It was employed in a mixture known as 80/20, that was 80% carbon tetrachloride and 20% Carbon disulfide.[35] The United States Environmental Protection Agency banned its use in 1985.[36]
Gallery
 Hemispheric and Global mean concentrations of CCl4 (NOAA/ESRL).
Hemispheric and Global mean concentrations of CCl4 (NOAA/ESRL). Time-series of atmospheric concentrations of CCl4 (Walker et al., 2000).
Time-series of atmospheric concentrations of CCl4 (Walker et al., 2000).
References
- ↑ Touloukian, Y.S., Liley, P.E., and Saxena, S.C. Thermophysical properties of matter - the TPRC data series. Volume 3. Thermal conductivity - nonmetallic liquids and gases. Data book. 1970.
- ↑ Reid, Robert C.; Prausnitz, John M.; Poling, Bruce E. (1987), The Properties of Gases and Liquids, McGraw-Hill Book Company, p. 442, ISBN 0-07-051799-1
- 1 2 "Carbon tetrachloride". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- 1 2 3 NIOSH Pocket Guide to Chemical Hazards. "#0107". National Institute for Occupational Safety and Health (NIOSH).
- ↑ "Carbon Tetrachloride". webbook.nist.gov. Archived from the original on 30 June 2017. Retrieved 28 April 2018.
- ↑ F. Brezina, J. Mollin, R. Pastorek, Z. Sindelar. Chemicke tabulky anorganickych sloucenin (Chemical tables of inorganic compounds). SNTL, 1986.
- ↑ V. Regnault (1839) "Sur les chlorures de carbone CCl et CCl2" (On the chlorides of carbon CCl and CCl2 ), Annales de Chimie et de Physique, vol. 70, pages 104-107. Reprinted in German as: V. Regnault (1839). "Ueber die Chlorverbindungen des Kohlenstoffs, C2Cl2 und CCl2". Annalen der Pharmacie. 30 (3): 350–352. doi:10.1002/jlac.18390300310.
- 1 2 Manfred Rossberg, Wilhelm Lendle, Gerhard Pfleiderer, Adolf Tögel, Eberhard-Ludwig Dreher, Ernst Langer, Heinz Jaerts, Peter Kleinschmidt, Heinz Strack, Richard Cook, Uwe Beck, Karl-August Lipper, Theodore R. Torkelson, Eckhard Löser, Klaus K. Beutel, "Chlorinated Hydrocarbons" in Ullmann's Encyclopedia of Industrial Chemistry, 2006 Wiley-VCH, Weinheim. doi:10.1002/14356007.a06_233.pub2
- 1 2 3 Use of Ozone Depleting Substances in Laboratories. TemaNord 516/2003. Archived February 27, 2008, at the Wayback Machine
- 1 2
Seifert W. F., Bosma A., Brouwer A. et al. (January 1994). "Vitamin A deficiency potentiates carbon tetrachloride-induced liver fibrosis in rats". Hepatology. 19 (1): 193–201. doi:10.1002/hep.1840190129. PMID 8276355. S2CID 205863459.
{{cite journal}}: CS1 maint: uses authors parameter (link) - ↑
Liu K. X., Kato Y., Yamazaki M., Higuchi O., Nakamura T., Sugiyama Y. (April 1993). "Decrease in the hepatic clearance of hepatocyte growth factor in carbon tetrachloride-intoxicated rats". Hepatology. 17 (4): 651–60. doi:10.1002/hep.1840170420. PMID 8477970. S2CID 25794501.
{{cite journal}}: CS1 maint: uses authors parameter (link) - ↑ Recknagel R. O.; Glende E. A.; Dolak J. A.; Waller R. L. (1989). "Mechanism of Carbon-tetrachloride Toxicity". Pharmacology & Therapeutics. 43 (43): 139–154. doi:10.1016/0163-7258(89)90050-8. PMID 2675128.
- ↑ Recknagel R. O. (June 1967). "Carbon tetrachloride hepatotoxicity". Pharmacol. Rev. 19 (2): 145–208. PMID 4859860.
- ↑ Masuda Y. (October 2006). "[Learning toxicology from carbon tetrachloride-induced hepatotoxicity]". Yakugaku Zasshi (in Japanese). 126 (10): 885–99. doi:10.1248/yakushi.126.885. PMID 17016019.
- ↑ Rood A. S., McGavran P. D., Aanenson J. W., Till J. E. (August 2001). "Stochastic estimates of exposure and cancer risk from carbon tetrachloride released to the air from the rocky flats plant". Risk Anal. 21 (4): 675–95. doi:10.1111/0272-4332.214143. PMID 11726020.
{{cite journal}}: CS1 maint: uses authors parameter (link) - ↑ Material Safety Data Sheet, Carbon tetrachloride Archived 2010-09-13 at the Wayback Machine at Fisher Scientific.
- ↑ "Substance evaluation - CoRAP - ECHA". echa.europa.eu. Archived from the original on 20 August 2016. Retrieved 28 April 2018.
- ↑ Odabasi M. (2008). "Halogenated Volatile Organic Compounds from the Use of Chlorine-Bleach-Containing Household Products". Environmental Science & Technology. 42 (5): 1445–51. Bibcode:2008EnST...42.1445O. doi:10.1021/es702355u. PMID 18441786.
- ↑ Fraser P. (1997). "Chemistry of stratospheric ozone and ozone depletion". Australian Meteorological Magazine. 46 (3): 185–193.
- ↑ Evans W. F. J., Puckrin E. (1996). "A measurement of the greenhouse radiation associated with carbon tetrachloride (CCl4)". Geophysical Research Letters. 23 (14): 1769–72. Bibcode:1996GeoRL..23.1769E. doi:10.1029/96GL01258.
{{cite journal}}: CS1 maint: uses authors parameter (link) - ↑ Walker, S. J.; Weiss R. F. & Salameh P. K. (2000). "Reconstructed histories of the annual mean atmospheric mole fractions for the halocarbons CFC-11, CFC-12, CFC-113 and carbon tetrachloride". Journal of Geophysical Research. 105 (C6): 14285–96. Bibcode:2000JGR...10514285W. doi:10.1029/1999JC900273.
- ↑ The Atlas of Climate Change (2006) by Kirstin Dow and Thomas E. Downing ISBN 978-0-520-25558-6
- ↑ "Report on Carcinogens, Fourteenth Edition - Carbon Tetrachloride" (PDF). ntp.niehs.nih.gov.
- ↑ "Environmental Health Criteria 208: CARBON TETRACHLORIDE" (PDF). who.int.
- ↑ "Public Health Statement for Carbon Tetrachloride (Tetracloruro de Carbono)". atsdr.cdc.gov.
- ↑ Doherty R. E. (2000). "A History of the Production and Use of Carbon Tetrachloride, Tetrachloroethylene, Trichloroethylene and 1,1,1-Trichloroethane in the United States: Part 1—Historical Background; Carbon Tetrachloride and Tetrachloroethylene". Environmental Forensics. 1 (2): 69–81. doi:10.1006/enfo.2000.0010. S2CID 97680726.
- ↑ W. Reusch. "Introduction to Nuclear Magnetic Resonance Spectroscopy". Virtual Textbook of Organic Chemistry. Michigan State University. Archived from the original on August 31, 2006.
- ↑ U.S. Patent 1,010,870, filed April 5, 1910.
- ↑ U.S. Patent 1,105,263, filed Jan 7, 1911.
- ↑ "Pyrene Fire Extinguishers". Vintage Fire Extinguishers. Archived from the original on 25 March 2010. Retrieved 23 December 2009.
- ↑ Fieldner, A. C.; Katz, S. H.; Kinney, S. P.; Longfellow, E. S. (1920-10-01). "Poisonous gases from carbon tetrachloride fire extinguishers". Journal of the Franklin Institute. 190 (4): 543–565. doi:10.1016/S0016-0032(20)91494-1. Retrieved 2022-02-03.
- ↑ Burke, Robert (2007-11-06). Fire Protection: Systems and Response. CRC Press. p. 209. ISBN 978-0-203-48499-9.
- ↑ "Red Comet Manufacturing Company". City of Littleton, CO. Archived from the original on 1 October 2016. Retrieved 30 September 2016.
- ↑ "ACSH Explains: What's The Story On Carbon Tetrachloride?". American Council on Science and Health. 2018-08-09. Retrieved 2022-02-03.
- ↑ Peters, H. A.; Levine, R. L.; Matthews, C. G.; Sauter, S.; Chapman, L. (1986). "Synergistic neurotoxicity of carbon tetrachloride/carbon disulfide (80/20 fumigants) and other pesticides in grain storage workers". Acta Pharmacologica et Toxicologica. 59: 535–546. doi:10.1111/j.1600-0773.1986.tb02820.x. PMID 3535379. Retrieved 2022-02-03.
- ↑ Darst, Guy (1985-02-12). "Manufacturers Take Grain Fumigant Off Market in Face of EPA Testing". AP News. Retrieved 2022-02-03.
External links
- International Chemical Safety Card 0024
- NIOSH Pocket Guide to Chemical Hazards. "#0107". National Institute for Occupational Safety and Health (NIOSH).
- "Carbon Tetrachloride (Group 2B)". International Agency for Research on Cancer (IARC) – Summaries & Evaluations. 71: 401. 1999.
- IARC Monograph: "Carbon Tetrachloride"
- Toxicological profile for carbon tetrachloride
- Environmental health criteria for carbon tetrachloride
- Carbon tetrachloride MSDS at Hazardous Chemical Database
- Substance profile at ntp.niehs.nih.gov
- ChemSub Online: Carbon tetrachloride