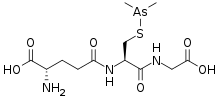
Darinaparsin
 | |
| Clinical data | |
|---|---|
| Trade names | Darvias; Zinapar |
| Other names | SP-02; ZIO-101; DMAs(III)G |
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.210.066 |
| Chemical and physical data | |
| Formula | C12H22AsN3O6S |
| Molar mass | 411.30 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Darinaparsin (trade names Darvias and Zinapar) is a drug for the treatment of various types of cancer.[1] It is an arsenic-containing derivative of glutathione.[2]
The mechanism of action of darinaparsin is proposed to involve disruption of mitochondrial function, increased production of reactive oxygen species, and modulation of intracellular signal transduction pathways, thereby inducing cell cycle arrest and apoptosis in cancer cells.[3]
History
Darinaparsin and related compounds were first studied in the 1970s at Texas A&M University. It wasn't until 1998, when a connection between organoarsenic compounds and their potential use in cancer chemotherapy was reported, that interest in darinaparsin as a pharmaceutical drug began.[4] Darinaparsin was licensed to Ziopharm Oncology and then Solasia Pharma for drug development.[4] In 2018, Solasia Pharma licensed Darinaparsin to HB Human BioScience SAS of Bogota Colombia.[5] HB Human BioScience SAS has began the registration process in Colombia, Peru, Ecuador, Venezuela, Chile, Panama, Costa Rica, and Guatemala, while offering Darinaparsin on a compassionate use (expanded access) basis.[5]
Darinaparsin was granted Orphan Drug Designation in the US and Europe as a treatment for peripheral T-cell lymphoma (PTCL).[6]
In Japan, Darinaparsin was approved for relapsed or refractory PTCL in June 2022.[7]
References
- ↑ "Darinaparsin - Solasia Pharma". Adis Insight. Springer Nature Switzerland AG.
- ↑ "Darinaparsin". NCI Drug Dictionary. National Cancer Institute.
- ↑ "SP-02". Solasia.
- 1 2 Quintás-Cardama A, Verstovsek S, Freireich E, Kantarjian H, Chen YW, Zingaro R (December 2008). "Chemical and clinical development of darinaparsin, a novel organic arsenic derivative". Anti-Cancer Agents in Medicinal Chemistry. 8 (8): 904–909. doi:10.2174/187152008786847666. PMID 19075572.
- 1 2 "Announcement Concerning Signing of License and Distribution Agreement for SP-02 (darinaparsin injection) in Latin America" (PDF). Solasia. August 17, 2018. Retrieved November 10, 2023.
- ↑ "Darinaparsin". Inxight Drugs. National Institutes of Health.
- ↑ "Solasia Pharma: DARVIAS® Injection 135mg (Generic Name: DARINAPARSIN /Development Code: SP-02) Approved in Japan" (Press release). June 19, 2022.