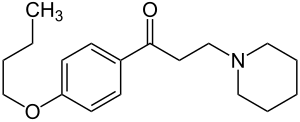
Dyclonine
 | |
| Names | |
|---|---|
| Trade names | Dyclone, Sucrets, others[1] |
| Other names | Dyclonine hydrochloride |
IUPAC name
| |
| Clinical data | |
| Drug class | Local anesthetic[2] |
| Main uses | Throat pain[2] |
| Side effects | Stinging, allergic reaction[2] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | Lozenge |
| External links | |
| AHFS/Drugs.com | Monograph |
| Legal | |
| Legal status |
|
| Chemical and physical data | |
| Formula | C18H27NO2 |
| Molar mass | 289.419 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Dyclonine, also known as dyclocaine, is a local anesthetic used for throat or mouth pain.[2] It is available as a cough drop.[2] Effects may begin within 10 minutes and last for up to 30 minutes.[2]
Common side effects include stinging.[2] Other side effects may include allergic reactions and jitteriness.[2] Safety in pregnancy is unclear.[3] It is neither an ester or an amide.[3]
Dyclonine was in medical use at least as early as the 1950s.[4] It is the available over-the-counter under the brand name Sucrets among others.[2]
History
The product Sucrets was introduced in Baltimore, Maryland, by Sharp & Dohme in 1932 and would eventually contain dyclonine.[5]
In 1966 the Federal Trade Commission ordered Merck and Company to discontinue the false claims of germ-killing and pain-relieving properties for its Sucrets and Children's Sucrets throat lozenges.[6] In 1977, it was acquired by Beecham, later merging with SmithKline Beckman in 1989 to form SmithKline Beecham. By 1994 the brand switched from a metal container to a plastic container.[5] SmithKline Beecham, after announcing a merger with GlaxoWellcome to form GlaxoSmithKline, sold the brand in 2000 to Insight Pharmaceuticals. In 2011, Sucrets reintroduced their product back into the familiar tin due to popular demand and nostalgia.
References
- ↑ Society, Swiss Pharmaceutical (2000). Index Nominum 2000: International Drug Directory. Taylor & Francis. p. 378. ISBN 978-3-88763-075-1. Archived from the original on 2022-01-11. Retrieved 2021-12-27.
- 1 2 3 4 5 6 7 8 9 "Dyclonine Monograph for Professionals". Drugs.com. Archived from the original on 25 January 2021. Retrieved 27 December 2021.
- 1 2 Logothetis, Demetra D. (15 March 2016). Local Anesthesia for the Dental Hygienist. Elsevier Health Sciences. p. 83. ISBN 978-0-323-43050-0. Archived from the original on 11 January 2022. Retrieved 27 December 2021.
- ↑ GREIFENSTEIN, FE; HARRIS LC, Jr; PARRY, JC (September 1956). "Dyclonine; a new local anesthetic agent: clinical evaluation". Anesthesiology. 17 (5): 648–52. doi:10.1097/00000542-195609000-00002. PMID 13355009.
- 1 2 "The Sucrets tin joins the age of plastics". USA Today. July 19, 1994. Archived from the original on 2017-08-22. Retrieved 2011-09-24.
Invented in Baltimore by Sharp & Dohme pharmaceutical in 1932, Sucrets have always been sold in the trademark metal box except for one 4 1⁄2-month period during the late 1960s when a tin shortage led to cardboard packaging, says [Frank Dzvonik].
- ↑ "F.T.C. Bids Merck Halt Claims That Lozenges Will Kill Germs". The New York Times. Associated Press. April 19, 1966. Archived from the original on 2012-11-07. Retrieved 2011-09-24.
External links
| Identifiers: |
|---|