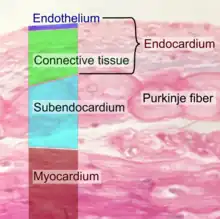
Endothelium
| Endothelium | |
|---|---|
 Diagram showing the location of endothelial cells | |
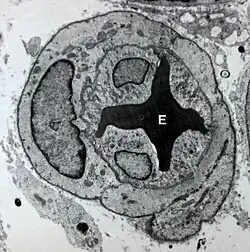 Transmission electron micrograph of a microvessel showing endothelial cells, which encircle an erythrocyte (E), forming the innermost layer of the vessel, the tunica intima. | |
| Details | |
| System | Circulatory system |
| Location | Lining of the inner surface of blood vessels and lymphatic vessels |
| Identifiers | |
| MeSH | D004727 |
| TH | H2.00.02.0.02003 |
| FMA | 63916 |
| Anatomical terms of microanatomy | |
Endothelium is a single layer of squamous endothelial cells that line the interior surface of blood vessels, and lymphatic vessels.[1] The endothelium forms an interface between circulating blood or lymph in the lumen and the rest of the vessel wall. Endothelial cells form the barrier between vessels and tissue and control the flow of substances and fluid into and out of a tissue.
Endothelial cells in direct contact with blood are called vascular endothelial cells whereas those in direct contact with lymph are known as lymphatic endothelial cells. Vascular endothelial cells line the entire circulatory system, from the heart to the smallest capillaries.
These cells have unique functions that include fluid filtration, such as in the glomerulus of the kidney, blood vessel tone, hemostasis, neutrophil recruitment, and hormone trafficking. Endothelium of the interior surfaces of the heart chambers is called endocardium. An impaired function can lead to serious health issues throughout the body.
Structure
The endothelium is a thin layer of single flat (squamous) cells that line the interior surface of blood vessels and lymphatic vessels.[1]
Endothelium is of mesodermal origin. Both blood and lymphatic capillaries are composed of a single layer of endothelial cells called a monolayer. In straight sections of a blood vessel, vascular endothelial cells typically align and elongate in the direction of fluid flow.[2][3]
Terminology
The foundational model of anatomy, an index of terms used to describe anatomical structures, makes a distinction between endothelial cells and epithelial cells on the basis of which tissues they develop from, and states that the presence of vimentin rather than keratin filaments separates these from epithelial cells.[4] Many considered the endothelium a specialized epithelial tissue.[5]
Function

The endothelium forms an interface between circulating blood or lymph in the lumen and the rest of the vessel wall. This forms a barrier between vessels and tissues and control the flow of substances and fluid into and out of a tissue. This controls the passage of materials and the transit of white blood cells into and out of the bloodstream. Excessive or prolonged increases in permeability of the endothelium, as in cases of chronic inflammation, may lead to tissue swelling (edema). Altered barrier function is also implicated in cancer extravasation.[6]
Endothelial cells are involved in many other aspects of vessel function, including:
- Blood clotting (thrombosis and fibrinolysis). The endothelium normally provides a surface on which blood does not clot, because it contains and expresses substances that prevent clotting, including heparan sulfate which acts as a cofactor for activating antithrombin, a protease that inactivates several factors in the coagulation cascade.
- Inflammation.[7] Endothelial cells actively signal to white blood cells of the immune system[8] during inflammation
- Formation of new blood vessels (angiogenesis).
- Constriction and enlargement of the blood vessel, called vasoconstriction and vasodilation, and hence the control of blood pressure
Blood vessel formation
The endothelium is involved in the formation of new blood vessels, called angiogenesis. Angiogenesis is a crucial process for development of organs in the embryo and fetus,[9] as well as repair of damaged areas. The process is triggered by decreased tissue oxygen (hypoxia) or insufficient oxygen tension leading to the new development of blood vessels lined with endothelial cells. Angiogenesis is regulated by signals that promote and decrease the process. These pro- and antiangiogenic signals including integrins, chemokines, angiopoietins, oxygen sensing agents, junctional molecules and endogenous inhibitors.[9] Angiopoietin-2 works with VEGF to facilitate cell proliferation and migration of endothelial cells.
The general outline of angiogenesis is
- activating signals binding to surface receptors of vascular endothelial cells.
- activated endothelial cells release proteases leading to the degradation of the basement membrane
- endothelial cells are freed to migrate from the existing blood vessels and begin to proliferate to form extensions towards the source of the angiogenic stimulus.
Host immune response
Endothelial cells express a variety of immune genes in an organ-specific manner.[10] These genes include critical immune mediators and proteins that facilitate cellular communication with hematopoietic immune cells.[11] Endothelial cells encode important features of the structural cell immune response in the epigenome and can therefore respond swiftly to immunological challenges. The contribution to host immunity by non-hematopoietic cells, such as endothelium, is called “structural immunity”.[12]
Clinical significance
Endothelial dysfunction, or the loss of proper endothelial function, is a hallmark for vascular diseases, and is often regarded as a key early event in the development of atherosclerosis.[13] Impaired endothelial function, causing hypertension and thrombosis, is often seen in patients with coronary artery disease, diabetes mellitus, hypertension, hypercholesterolemia, as well as in smokers. Endothelial dysfunction has also been shown to be predictive of future adverse cardiovascular events, and is also present in inflammatory disease such as rheumatoid arthritis and systemic lupus erythematosus.
Endothelial dysfunction is a result of changes in endothelial function.[14][15] After fat (lipid) accumulation and when stimulated by inflammation, endothelial cells become activated, which is characterized by the expression of molecules such as E-selectin, VCAM-1 and ICAM-1, which stimulate the adhesion of immune cells.[16] Additionally, transcription factors, which are substances which act to increase the production of proteins within cells, become activated; specifically AP-1 and NF-κB, leading to increased expression of cytokines such as IL-1, TNFα and IFNγ, which promotes inflammation.[17][18] This state of endothelial cells promotes accumulation of lipids and lipoproteins in the intima, leading to atherosclerosis, and the subsequent recruitment of white blood cells and platelets, as well as proliferation of smooth muscle cells, leading to the formation of a fatty streak. The lesions formed in the intima, and persistent inflammation lead to desquamation of endothelium, which disrupts the endothelial barrier, leading to injury and consequent dysfunction.[19] In contrast, inflammatory stimuli also activate NF-κB-induced expression of the deubiquitinase A20 (TNFAIP3), which has been shown to intrinsically repair the endothelial barrier.[20]
One of the main mechanisms of endothelial dysfunction is the diminishing of nitric oxide, often due to high levels of asymmetric dimethylarginine, which interfere with the normal L-arginine-stimulated nitric oxide synthesis and so leads to hypertension. The most prevailing mechanism of endothelial dysfunction is an increase in reactive oxygen species, which can impair nitric oxide production and activity via several mechanisms.[21] The signalling protein ERK5 is essential for maintaining normal endothelial cell function.[22] A further consequence of damage to the endothelium is the release of pathological quantities of von Willebrand factor, which promote platelet aggregation and adhesion to the subendothelium, and thus the formation of potentially fatal thrombi.
Angiosarcoma is cancer of the endothelium and is rare with only 300 cases per year in the US.[23] However it generally has poor prognosis with a five-year survival rate of 35%.[24]
Research
In cancer therapy the development and delivery of anti-angiogenic drugs is a very promising path and restoring vascular homeostasis holds great potential for the treatment of ischemic tissue diseases [25]
In April 2020, the presence of viral elements in endothelial cells of 3 patients who had died of COVID-19 was reported for the first time. The researchers from the University of Zurich and Harvard Medical School considered these findings to be a sign of a general endotheliitis in different organs, an inflammatory response of the endothelium to the infection that can lead or at least contribute to multi-organ failure in Covid-19 patients with comorbidities such as diabetes mellitus, hypertension and cardiovascular disease.[26][27]
History
In 1958, Todd demonstrated that endothelium in human blood vessels have fibrinolytic activity.[28][29]
See also
- Apelin
- Caveolae
- Cellular dewetting
- Endothelial activation
- Endothelial microparticle
- Endothelial progenitor cell
- Endothelium-derived relaxing factor (EDRF)
- Robert F. Furchgott (1998 Nobel prize for discovery of EDRF)
- Platelet activation
- Susac's syndrome
- Tunica intima
- VE-cadherin
- Weibel–Palade body
- Angiocrine growth factors
- Endothelial Cell Tropism
References
- 1 2 "Endothelium" at Dorland's Medical Dictionary
- ↑ Eskin SG, Ives CL, McIntire LV, Navarro LT (July 1984). "Response of cultured endothelial cells to steady flow". Microvascular Research. 28 (1): 87–94. doi:10.1016/0026-2862(84)90031-1. PMID 6748961.
- ↑ Langille BL, Adamson SL (April 1981). "Relationship between blood flow direction and endothelial cell orientation at arterial branch sites in rabbits and mice". Circulation Research. 48 (4): 481–8. doi:10.1161/01.RES.48.4.481. PMID 7460219.
- ↑ "Endothelial cell". BioPortal. Stanford University. Archived from the original on 2013-10-02. Retrieved 2013-09-28.
- ↑ Kovacic JC, Mercader N, Torres M, Boehm M, Fuster V (April 2012). "Epithelial-to-mesenchymal and endothelial-to-mesenchymal transition: from cardiovascular development to disease". Circulation. 125 (14): 1795–808. doi:10.1161/circulationaha.111.040352. PMC 3333843. PMID 22492947.
- ↑ Escribano J, Chen MB, Moeendarbary E, Cao X, Shenoy V, Garcia-Aznar JM, et al. (May 2019). "Balance of mechanical forces drives endothelial gap formation and may facilitate cancer and immune-cell extravasation". PLOS Computational Biology. 15 (5): e1006395. arXiv:1811.09326. Bibcode:2019PLSCB..15E6395E. doi:10.1371/journal.pcbi.1006395. PMC 6497229. PMID 31048903.
- ↑ Li X, Fang P, Li Y, Kuo YM, Andrews AJ, Nanayakkara G, et al. (June 2016). "Mitochondrial Reactive Oxygen Species Mediate Lysophosphatidylcholine-Induced Endothelial Cell Activation". Arteriosclerosis, Thrombosis, and Vascular Biology. 36 (6): 1090–100. doi:10.1161/ATVBAHA.115.306964. PMC 4882253. PMID 27127201.
- ↑ Vestweber D (November 2015). "How leukocytes cross the vascular endothelium". Nature Reviews. Immunology. 15 (11): 692–704. doi:10.1038/nri3908. PMID 26471775. S2CID 29703333.
- 1 2 Bouïs D, Kusumanto Y, Meijer C, Mulder NH, Hospers GA (February 2006). "A review on pro- and anti-angiogenic factors as targets of clinical intervention". Pharmacological Research. 53 (2): 89–103. doi:10.1016/j.phrs.2005.10.006. PMID 16321545.
- ↑ Krausgruber T, Fortelny N, Fife-Gernedl V, Senekowitsch M, Schuster LC, Lercher A, et al. (July 2020). "Structural cells are key regulators of organ-specific immune responses". Nature. 583 (7815): 296–302. Bibcode:2020Natur.583..296K. doi:10.1038/s41586-020-2424-4. PMC 7610345. PMID 32612232. S2CID 220295181.
- ↑ Armingol E, Officer A, Harismendy O, Lewis NE (November 2020). "Deciphering cell-cell interactions and communication from gene expression". Nature Reviews. Genetics. doi:10.1038/s41576-020-00292-x. PMC 7649713. PMID 33168968.
- ↑ Minton K (September 2020). "A gene atlas of 'structural immunity'". Nature Reviews. Immunology. 20 (9): 518–519. doi:10.1038/s41577-020-0398-y. PMID 32661408. S2CID 220491226.
- ↑ Botts SR, Fish JE, Howe KL (December 2021). "Dysfunctional Vascular Endothelium as a Driver of Atherosclerosis: Emerging Insights Into Pathogenesis and Treatment". Frontiers in Pharmacology. 12: 787541. doi:10.3389/fphar.2021.787541. PMC 8727904. PMID 35002720.
- ↑ Iantorno M, Campia U, Di Daniele N, Nistico S, Forleo GB, Cardillo C, Tesauro M (April 2014). "Obesity, inflammation and endothelial dysfunction". Journal of Biological Regulators and Homeostatic Agents. 28 (2): 169–76. PMID 25001649.
- ↑ Reriani MK, Lerman LO, Lerman A (June 2010). "Endothelial function as a functional expression of cardiovascular risk factors". Biomarkers in Medicine. 4 (3): 351–60. doi:10.2217/bmm.10.61. PMC 2911781. PMID 20550469.
- ↑ Lopez-Garcia E, Hu FB (August 2004). "Nutrition and the endothelium". Current Diabetes Reports. 4 (4): 253–9. doi:10.1007/s11892-004-0076-7. PMID 15265466. S2CID 24878288.
- ↑ Blake GJ, Ridker PM (October 2002). "Inflammatory bio-markers and cardiovascular risk prediction". Journal of Internal Medicine. 252 (4): 283–94. doi:10.1046/j.1365-2796.2002.01019.x. PMID 12366601. S2CID 26400610.
- ↑ Mizuno Y, Jacob RF, Mason RP (2011). "Inflammation and the development of atherosclerosis". Journal of Atherosclerosis and Thrombosis. 18 (5): 351–8. doi:10.5551/jat.7591. PMID 21427505.
- ↑ Mäyränpää MI, Heikkilä HM, Lindstedt KA, Walls AF, Kovanen PT (November 2006). "Desquamation of human coronary artery endothelium by human mast cell proteases: implications for plaque erosion". Coronary Artery Disease. 17 (7): 611–21. doi:10.1097/01.mca.0000224420.67304.4d. PMID 17047445. S2CID 1884596.
- ↑ Soni D, Wang DM, Regmi SC, Mittal M, Vogel SM, Schlüter D, Tiruppathi C (May 2018). "Deubiquitinase function of A20 maintains and repairs endothelial barrier after lung vascular injury". Cell Death Discovery. 4 (60): 60. doi:10.1038/s41420-018-0056-3. PMC 5955943. PMID 29796309.
- ↑ Deanfield J, Donald A, Ferri C, Giannattasio C, Halcox J, Halligan S, Lerman A, Mancia G, Oliver JJ, Pessina AC, Rizzoni D, Rossi GP, Salvetti A, Schiffrin EL, Taddei S, Webb DJ (January 2005). "Endothelial function and dysfunction. Part I: Methodological issues for assessment in the different vascular beds: a statement by the Working Group on Endothelin and Endothelial Factors of the European Society of Hypertension". Journal of Hypertension. 23 (1): 7–17. doi:10.1097/00004872-200501000-00004. PMID 15643116.
- ↑ Roberts OL, Holmes K, Müller J, Cross DA, Cross MJ (December 2009). "ERK5 and the regulation of endothelial cell function". Biochemical Society Transactions. 37 (Pt 6): 1254–9. doi:10.1042/BST0371254. PMID 19909257.
- ↑ "Angiosarcoma - National Cancer Institute". www.cancer.gov. 2019-02-27. Retrieved 2021-08-10.
- ↑ Young, Robin J.; Brown, Nicola J.; Reed, Malcolm W.; Hughes, David; Woll, Penella J. (October 2010). "Angiosarcoma". The Lancet. Oncology. 11 (10): 983–991. doi:10.1016/S1470-2045(10)70023-1. ISSN 1474-5488. PMID 20537949.
- ↑ Van Hove AH, Benoit DS (2015). "Depot-Based Delivery Systems for Pro-Angiogenic Peptides: A Review". Frontiers in Bioengineering and Biotechnology. 3: 102. doi:10.3389/fbioe.2015.00102. PMC 4504170. PMID 26236708.
- ↑ Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS, Mehra MR, Schuepbach RA, Ruschitzka F, Moch H (May 2020). "Endothelial cell infection and endotheliitis in COVID-19". Lancet. 395 (10234): 1417–1418. doi:10.1016/S0140-6736(20)30937-5. PMC 7172722. PMID 32325026.
- ↑ Sardu C, Gambardella J, Morelli MB, Wang X, Marfella R, Santulli G (May 2020). "Hypertension, Thrombosis, Kidney Failure, and Diabetes: Is COVID-19 an Endothelial Disease? A Comprehensive Evaluation of Clinical and Basic Evidence". J Clin Med. 9 (5). doi:10.3390/jcm9051417. PMC 7290769. PMID 32403217.
- ↑ Todd AS (February 1958). "Fibrinolysis autographs". Nature. 181 (4607): 495–6. Bibcode:1958Natur.181..495T. doi:10.1038/181495b0. eISSN 1476-4687. PMID 13517190. S2CID 4219257.
- ↑ Todd AS (September 1964). "=Localization of fibrinolytic activity in tissues". British Medical Bulletin. 20 (3): 210–2. doi:10.1093/oxfordjournals.bmb.a070333. eISSN 1471-8391. PMID 14209761.
External links
- Anatomy photo: Circulatory/vessels/capillaries1/capillaries3 - Comparative Organology at University of California, Davis, "Capillaries, non-fenestrated (EM, Low)"
- Histology image: 21402ooa – Histology Learning System at Boston University
- Endothelium and inflammation
- Platelet Activation, University of Washington