Liquid-based cytology
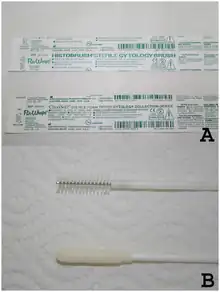
Liquid-based cytology is a method of preparing samples for examination in cytopathology. The sample is collected, normally by a small brush, in the same way as for a conventional smear test, but rather than the smear being transferred directly to a microscope slide, the sample is deposited into a small bottle of preservative liquid. At the laboratory, the liquid is treated to remove other elements such as mucus before a layer of cells is placed on a slide.[1] The technique improves specimen adequacy and HSIL detection.[2] The United Kingdom screening programmes changed their cervical screening method from the Pap test to liquid-based cytology in 2008.[3]
History
For many years, efforts have been made to develop methods that would enhance the sensitivity and specificity of the Papanicolaou smear (also called Pap smear). Emphasis has been placed on creating automated screening machines whose success depends on a representative sampling of cells on standardized slides containing a monolayer of well-stained, well-preserved cells.[4]
From this research and development, liquid-based gynecologic specimen collection has evolved. Its proponents argue that liquid-based preparations outperform conventional smears because of improved fixation, decreased obscuring factors, and standardization of cell transfer. Proponents point out that, in direct smears, the cells are not transferred in a representative fashion and that up to 90% of the material scraped from the cervix may be discarded with the sampling device. With liquid-based collection, the sampling will be representative and operator-dependent variation will not occur since processing is controlled by the laboratory.
SurePath (by BD) and ThinPrep (by Hologic) are two such systems currently approved by the United States Food and Drug Administration (FDA) for cervicovaginal testing. With both methods, the sample is collected in the conventional manner with one of the brush instruments but, instead of being spread onto a glass slide, it is transferred to a vial of transport medium. Multiple studies have shown that both systems improve specimen adequacy and HSIL detection.[2]
Methods
In the SurePath method, the sample is vortexed, strained, layered onto a density gradient, and centrifuged. Instruments required are a computer-controlled robotic pipette and a centrifuge. The cells form a circle 12.5 mm in diameter. The ThinPrep method requires an instrument and special polycarbonate filters. After the instrument immerses the filter into the vial, the filter is rotated to homogenize the sample. Cells are collected on the surface of the filter when a vacuum is applied. The filter is then pressed against a slide to transfer the cells into a 20 mm diameter circle.
Both methods result in a well-preserved approximate monolayer of cells with a background devoid of blood and mucus. However, the current high cost of these patented commercial systems, requiring cytotechnologists and pathologists to interpret the ThinPrep slides, has led to the development and evaluation of less costly alternatives.
One such method referred to as SpinThin, developed by Khalbuss et al. (2000), uses a modified electric toothbrush to release the cells into suspension from the collecting device. The cells are spun directly onto a 10 × 20 mm area of a glass slide using a Cytospin II cytocentrifuge with megafunnel. Results correlated well with conventional smears and follow-up histology.
Another method described by Johnson et al. (2000) places the cervical collection device into 15 ml of CytoRich Red (TriPath Imaging), a proprietary formula of buffering agents, emulsifiers, formaldehyde, and alcohol. After arrival in the laboratory, cell suspensions are vortexed, poured through tulle and centrifuged. Following centrifugation, the supernatant is discarded and the sediment is vortexed. A drop of sediment is placed into an 8 ml Hettich cytocentrifuge chamber prefilled with 2 ml of CytoRich Yellow (TriPath Imaging), a proprietary Saccomanno-like fixative that prevents dehydration and collapse of 3-dimensional structures when slides are air-dried, and then spun onto adhesive-coated slides. Advantages include batch processing and reusability of its funnel assembly, which decreases the bulk of disposable plastic that can significantly impact the environment as well as add to the cost of individual tests.[5]
References
- ↑ Liptak, Peter; Barnetson, Robert James (2012-02-14). "Liquid-based cervical cytology in the United Kingdom and South Africa". Continuing Medical Education. 30 (2): 45–48. ISSN 2078-5143.
- 1 2 Gibb, Randall K; Martens, Mark G (2011). "The Impact of Liquid-Based Cytology in Decreasing the Incidence of Cervical Cancer". Reviews in Obstetrics and Gynecology. 4 (Suppl 1): S2–S11. ISSN 1941-2797. PMC 3101960. PMID 21617785.
- ↑ Liquid Based Cytology (LBC), NHS cervical screening programme (accessed 28/07/2014)
- ↑ Wilbur, David C.; Bibbo, Marluce (2008), "Automation in Cervical Cytology", Comprehensive Cytopathology, Elsevier, pp. 1021–1042, doi:10.1016/b978-141604208-2.10034-x, ISBN 978-1-4160-4208-2, retrieved 2020-07-27
- ↑ "בדיקת פאפ". Saturday, 17 July 2021