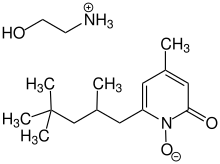
Piroctone olamine
 | |
| Identifiers | |
|---|---|
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.065.957 |
| MeSH | Piroctone+olamine |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C16H30N2O3 |
| Molar mass | 298.421 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Piroctone olamine (INN; also known as piroctone ethanolamine;) is a compound sometimes used in the treatment of fungal infections.[1] Piroctone olamine is the ethanolamine salt of the hydroxamic acid derivative piroctone.
It is often used in anti-dandruff shampoo as a replacement for the commonly used compound zinc pyrithione. It is structurally similar to ciclopirox and pyrithione, containing a substituted pyridine (pyridinone) group which inhibits ergosterol synthesis.
References
- ↑ Dubini F, Bellotti MG, Frangi A, Monti D, Saccomani L (2005). "In vitro antimycotic activity and nail permeation models of a piroctone olamine containing transungual water soluble technology". Arzneimittel-Forschung. 55 (8): 478–83. doi:10.1055/s-0031-1296892. PMID 16149717.
This article is issued from Offline. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.