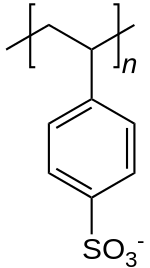
Polystyrene sulfonate
 | |
| Names | |
|---|---|
| Trade names | Sodium salt: Kayexalate, Kionex, Resonium A Calcium salt: Calcium Resonium, Sorbisterit, Resikali |
IUPAC name
| |
| Clinical data | |
| Drug class | Cation exchange resin[1] |
| Main uses | High blood potassium[1] |
| Side effects | Loss of appetite, gastrointestinal upset, constipation, low blood calcium[1] |
| WHO AWaRe | UnlinkedWikibase error: ⧼unlinkedwikibase-error-statements-entity-not-set⧽ |
| Pregnancy category |
|
| Routes of use | By mouth, retention enema |
| External links | |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682108 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | None |
| Metabolism | None |
| Excretion | Faeces (100%) |
| Chemical and physical data | |
| Formula | [C8H7SO3−] n |
Polystyrene sulfonates are a group of medications used to treat high blood potassium.[1][2] Effects generally take hours to days.[1] It may be taken by mouth or used rectally.[1]
Common side effects include loss of appetite, gastrointestinal upset, constipation, and low blood calcium.[1] Serious side effects may include intestinal injury, electrolyte abnormalities, and heart failure.[1] It works as a cation exchange resin.[1]
Sodium polystyrene sulfonate was approved for medical use in the United States in 1958.[1] In the United States the sodium salt costs about 60 USD for 450 grams as of 2021.[3] In the United Kingdom this amount of the sodium salt costs about £81 while the calcium salt costs about £125.[2]
Medical uses
Polystyrene sulfonate is usually supplied in either the sodium or calcium form. It is used as a potassium binder in acute and chronic kidney disease for people with hyperkalemia (abnormal high blood serum potassium levels).[4] However, it is unclear if it is beneficial and there is concern about possible side effects when it is combined with sorbitol.[5]
Dosage
Polystyrene sulfonates are given by mouth with a meal or rectally by retention enema.[6]
In adults the sodium and calcium salts are taken as a dose of 15 grams by mouth one to four times per day.[1][2] Rectally a dose of 30 to 50 grams may be used.[1][2]
Side effects
Intestinal disturbances are common, including loss of appetite, nausea, vomiting, and constipation. In rare cases, it has been associated with colonic necrosis.[7] Changes in electrolyte blood levels such as hypomagnesemia, hypocalcemia, and hypokalemia may occur.[8] Polystyrene sulfonates should not be used in people with obstructive bowel disease and in newborns with reduced gut motility.[9]
Intestinal injury
A total of 58 cases of intestinal injury including necrosis of the colon have been reported with polystyrene sulfonate as of 2013.[10] Well more cases have been reported when used in combination with sorbitol and other cases have occurred when used alone.[10]
The evidence supporting an association of polystyrene sulfonate and intestinal necrosis is low.[11]
Interactions
Polystyrene sulfonates can bind to various drugs within the digestive tract and thus lower their absorption and effectiveness. Common examples include lithium, thyroxine, and digitalis. In September 2017, the FDA recommended separating the dosing of polystyrene sulfonate from any other oral medications by at least three hours to avoid any potential interactions.[12]
Mechanism of action
Hyperkalemia
Polystyrene sulfonates release sodium or calcium ions in the stomach in exchange for hydrogen ions. When the resin reaches the large intestine the hydrogen ions are exchanged for free potassium ions; the resin is then eliminated in the feces. The net effect is lowering the amount of potassium available for absorption into the blood and increasing the amount that is excreted via the feces. The effect is a reduction of potassium levels in the body, at a capacity of 1 mEq of potassium exchanged per 1 g of resin.[9][13]
Production and chemical structure
Polystyrene sulfonic acid, the acid whose salts are the polystyrene sulfonates, has the idealized formula (CH2CHC6H4SO3H)n. The material is prepared by sulfonation of polystyrene:
- (CH2CHC6H5)n + n SO3 → (CH2CHC6H4SO3H)n
Several methods exist for this conversion, which can lead to varying degree of sulfonation. Usually the polystyrene is crosslinked, which keeps the polymer from dissolving. Since the sulfonic acid group (SO3H) is strongly acidic, this polymer neutralizes bases. In this way, various salts of the polymer can be prepared, leading to sodium, calcium, and other salts:
- (CH2CHC6H4SO3H)n + n NaOH → (CH2CHC6H4SO3Na)n + n H2O
These ion-containing polymers are called ionomers.
Alternative sulfonation methods
Double substitutions of the phenyl rings are known to occur, even with conversions well below 100%. Crosslinking reactions are also found, where condensation of two sulfonic acid groups yields a sulfonyl crosslink. On the other hand, the use of milder conditions such as acetyl sulfate leads to incomplete sulfonation. Recently, the atom transfer radical polymerization (ATRP) of protected styrene sulfonates has been reported,[14][15] leading to well defined linear polymers, as well as more complicated molecular architectures.[16]
Chemical uses
They are also used to remove potassium, calcium, and sodium from solutions in technical applications.
Polystyrene sulfonates are useful because of their ion exchange properties.[17] Linear ionic polymers are generally water-soluble, whereas cross-linked materials (called resins) do not dissolve in water. These polymers are classified as polysalts and ionomers.[17]
Water softening
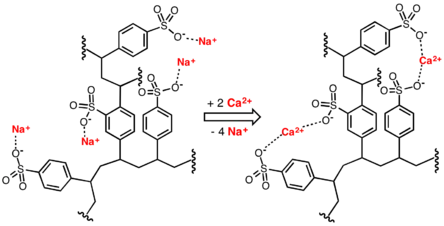
Water softening is achieved by percolating hard water through a bed of the sodium form of cross-linked polystyrene sulfonate. The hard ions such as calcium (Ca2+) and magnesium (Mg2+) adhere to the sulfonate groups, displacing sodium ions. The resulting solution of sodium ions is softened.
Other uses
Sodium polystyrene sulfonate is used as a superplastifier in cement, as a dye improving agent for cotton, and as proton exchange membranes in fuel cell applications. In its acid form, the resin is used as a solid acid catalyst in organic synthesis.[18]
History
A sodium potassium polystyrene sulfonate was developed in the 2000s to treat Clostridium difficile associated diarrhea under the name Tolevamer,[19] but it was never marketed.
See also
- Sodium zirconium cyclosilicate, another potassium binder
- Patiromer
References
- 1 2 3 4 5 6 7 8 9 10 11 12 "Sodium Polystyrene Sulfonate Monograph for Professionals". Drugs.com. Archived from the original on 21 October 2020. Retrieved 25 October 2019.
- 1 2 3 4 BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 1117. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ↑ "Sodium polystyrene sulfonate Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 25 October 2019. Retrieved 12 October 2021.
- ↑ MedlinePlus Encyclopedia: High potassium level
- ↑ Sterns RH, Rojas M, Bernstein P, Chennupati S (May 2010). "Ion-exchange resins for the treatment of hyperkalemia: are they safe and effective?". J. Am. Soc. Nephrol. 21 (5): 733–5. doi:10.1681/ASN.2010010079. PMID 20167700.
- ↑ "Polystyrene sulfonate". Martindale: The Complete Drug Reference. Medicines Complete. Archived from the original on 28 August 2021. Retrieved 27 November 2009.
- ↑ Rogers FB, Li SC (August 2001). "Acute colonic necrosis associated with sodium polystyrene sulfonate (Kayexalate) enemas in a critically ill patient: case report and review of the literature". J Trauma. 51 (2): 395–7. doi:10.1097/00005373-200108000-00031. PMID 11493807.
- ↑ KAYEXALATE (sodium polystyrene sulfonate). FULL PRESCRIBING INFORMATION Revised 07/2017 Archived 2021-07-10 at the Wayback Machine Retrieved 2018-10-21
- 1 2 FDA Professional Drug Information for Kayexalate.
- 1 2 Harel, Z; Harel, S; Shah, PS; Wald, R; Perl, J; Bell, CM (March 2013). "Gastrointestinal adverse events with sodium polystyrene sulfonate (Kayexalate) use: a systematic review". The American Journal of Medicine. 126 (3): 264.e9–24. doi:10.1016/j.amjmed.2012.08.016. PMID 23321430.
- ↑ Holleck, JL; Roberts, AE; Marhoffer, EA; Grimshaw, AA; Gunderson, CG (August 2021). "Risk of Intestinal Necrosis With Sodium Polystyrene Sulfonate: A Systematic Review and Meta-analysis". Journal of hospital medicine. 16 (8): 489–494. doi:10.12788/jhm.3655. PMID 34328838.
- ↑ Commissioner, Office of the. "Safety Alerts for Human Medical Products - Kayexalate (sodium polystyrene sulfonate): Drug Safety Communication - FDA Recommends Separating Dosing". www.fda.gov. Retrieved 2017-09-19.
- ↑ Chaitman, Martin; Dixit, Deepali; Bridgeman, Mary Barna (January 2016). "Potassium-Binding Agents for the Clinical Management of Hyperkalemia". Pharmacy and Therapeutics. 41 (1): 43–50. ISSN 1052-1372. PMC 4699486. PMID 26765867.
- ↑ Sikkema, FD; Comellas-Aragonès, M; Fokkink, RG; Verduin, BJ; Cornelissen, JJ; Nolte, RJ (2007). "Monodisperse polymer-virus hybrid nanoparticles". Organic & Biomolecular Chemistry. 5 (1): 54–7. doi:10.1039/b613890j. PMID 17164905.
- ↑ Lienkamp, Karen; Schnell, Ingo; Groehn, Franziska; Wegner, Gerhard (2006). "Polymerization of Styrene Sulfonate Ethyl Ester by ATRP: Synthesis and Characterization of Macromonomers for Suzuki Polycondensation". Macromolecular Chemistry and Physics. 207 (22): 2066. doi:10.1002/macp.200600322.
- ↑ Lienkamp, Karen; Ruthard, Christian; Lieser, GüNter; Berger, RüDiger; Groehn, Franziska; Wegner, Gerhard (2006). "Polymerization of Styrene Sulfonate Ethyl Ester and Styrene Sulfonate Dodecyl Ester by ATRP: Synthesis and Characterization of Polymer Brushes". Macromolecular Chemistry and Physics. 207 (22): 2050. doi:10.1002/macp.200600321. S2CID 98278283. Archived from the original on 2021-08-28. Retrieved 2021-07-12.
- 1 2 François Dardel and Thomas V. Arden "Ion Exchangers" in Ullmann's Encyclopedia of Industrial Chemistry, 2008, Wiley-VCH, Weinheim. doi:10.1002/14356007.a14_393.pub2
- ↑ Erik Gálvez, Pedro Romea, and Fèlix Urpí (2009). "Stereoselective Synthesis of anti α-Methyl-β-Methoxy Carboxylic Compounds". Organic Syntheses. 86: 81.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Hinkson, Paul L.; Dinardo, Carol; DeCiero, Daniel; Klinger, Jeffrey D.; Barker, Robert H. (2008). "Tolevamer, an Anionic Polymer, Neutralizes Toxins Produced by the BI/027 Strains of Clostridium difficile". Antimicrobial Agents and Chemotherapy. 52 (6): 2190–2195. doi:10.1128/AAC.00041-08. PMC 2415796. PMID 18391047.
External links
| Identifiers: |
|---|