Cervix
The cervix (PL: cervices) or cervix uteri (Latin, "neck of the uterus") is the lower part of the uterus (womb) in the human female reproductive system. The cervix is usually 2 to 3 cm long (~1 inch) and roughly cylindrical in shape, which changes during pregnancy. The narrow, central cervical canal runs along its entire length, connecting the uterine cavity and the lumen of the vagina.[1] The opening into the uterus is called the internal os, and the opening into the vagina is called the external os. The lower part of the cervix, known as the vaginal portion of the cervix (or ectocervix), bulges into the top of the vagina. The cervix has been documented anatomically since at least the time of Hippocrates, over 2,000 years ago.
| Cervix | |
|---|---|
 The human female reproductive system. The cervix is the lower narrower portion of the uterus. | |
| Details | |
| Precursor | Müllerian duct |
| Artery | Vaginal artery and uterine artery |
| Identifiers | |
| Latin | cervix uteri |
| MeSH | D002584 |
| TA98 | A09.1.03.010 |
| TA2 | 3508 |
| FMA | 17740 |
| Anatomical terminology | |
The cervical canal is a passage through which sperm must travel to fertilize an egg cell after sexual intercourse. Several methods of contraception, including cervical caps and cervical diaphragms, aim to block or prevent the passage of sperm through the cervical canal. Cervical mucus is used in several methods of fertility awareness, such as the Creighton model and Billings method, due to its changes in consistency throughout the menstrual period. During vaginal childbirth, the cervix must flatten and dilate to allow the fetus to progress along the birth canal. Midwives and doctors use the extent of the dilation of the cervix to assist decision-making during childbirth.
The cervical canal is lined with a single layer of column-shaped cells, while the ectocervix is covered with multiple layers of cells topped with flat cells. The two types of epithelia meet at the squamocolumnar junction. Infection with the human papillomavirus (HPV) can cause changes in the epithelium, which can lead to cancer of the cervix. Cervical cytology tests can often detect cervical cancer and its precursors, and enable early successful treatment. Ways to avoid HPV include avoiding sex, using condoms, and HPV vaccination. HPV vaccines, developed in the early 21st century, reduce the risk of cervical cancer by preventing infections from the main cancer-causing strains of HPV.[2]
Structure
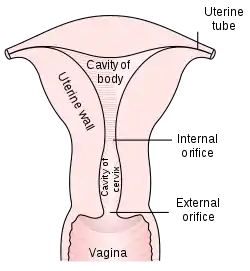
The cervix is part of the female reproductive system. Around 2–3 centimetres (0.8–1.2 in) in length,[3] it is the lower narrower part of the uterus continuous above with the broader upper part—or body—of the uterus.[4] The lower end of the cervix bulges through the anterior wall of the vagina, and is referred to as the vaginal portion of cervix (or ectocervix) while the rest of the cervix above the vagina is called the supravaginal portion of cervix.[4] A central canal, known as the cervical canal, runs along its length and connects the cavity of the body of the uterus with the lumen of the vagina.[4] The openings are known as the internal os and external orifice of the uterus (or external os), respectively.[4] The mucosa lining the cervical canal is known as the endocervix,[5] and the mucosa covering the ectocervix is known as the exocervix.[6] The cervix has an inner mucosal layer, a thick layer of smooth muscle, and posteriorly the supravaginal portion has a serosal covering consisting of connective tissue and overlying peritoneum.[4]

In front of the upper part of the cervix lies the bladder, separated from it by cellular connective tissue known as parametrium, which also extends over the sides of the cervix.[4] To the rear, the supravaginal cervix is covered by peritoneum, which runs onto the back of the vaginal wall and then turns upwards and onto the rectum, forming the recto-uterine pouch.[4] The cervix is more tightly connected to surrounding structures than the rest of the uterus.[7]
The cervical canal varies greatly in length and width between women or over the course of a woman's life,[3] and it can measure 8 mm (0.3 inch) at its widest diameter in premenopausal adults.[8] It is wider in the middle and narrower at each end. The anterior and posterior walls of the canal each have a vertical fold, from which ridges run diagonally upwards and laterally. These are known as palmate folds, due to their resemblance to a palm leaf. The anterior and posterior ridges are arranged in such a way that they interlock with each other and close the canal. They are often effaced after pregnancy.[7]
The ectocervix (also known as the vaginal portion of the cervix) has a convex, elliptical shape and projects into the cervix between the anterior and posterior vaginal fornices. On the rounded part of the ectocervix is a small, depressed external opening, connecting the cervix with the vagina. The size and shape of the ectocervix and the external opening (external os) can vary according to age, hormonal state, and whether childbirth has taken place. In women who have not had a vaginal delivery, the external opening is small and circular, and in women who have had a vaginal delivery, it is slit-like.[8] On average, the ectocervix is 3 cm (1.2 in) long and 2.5 cm (1 in) wide.[3]
Blood is supplied to the cervix by the descending branch of the uterine artery[9] and drains into the uterine vein.[10] The pelvic splanchnic nerves, emerging as S2–S3, transmit the sensation of pain from the cervix to the brain.[5] These nerves travel along the uterosacral ligaments, which pass from the uterus to the anterior sacrum.[9]
Three channels facilitate lymphatic drainage from the cervix.[11] The anterior and lateral cervix drains to nodes along the uterine arteries, travelling along the cardinal ligaments at the base of the broad ligament to the external iliac lymph nodes and ultimately the paraaortic lymph nodes. The posterior and lateral cervix drains along the uterine arteries to the internal iliac lymph nodes and ultimately the paraaortic lymph nodes, and the posterior section of the cervix drains to the obturator and presacral lymph nodes.[3][10][11] However, there are variations as lymphatic drainage from the cervix travels to different sets of pelvic nodes in some people. This has implications in scanning nodes for involvement in cervical cancer.[11]
After menstruation and directly under the influence of estrogen, the cervix undergoes a series of changes in position and texture. During most of the menstrual cycle, the cervix remains firm, and is positioned low and closed. However, as ovulation approaches, the cervix becomes softer and rises to open in response to the higher levels of estrogen present.[12] These changes are also accompanied by changes in cervical mucus,[13] described below.
Development
As a component of the female reproductive system, the cervix is derived from the two paramesonephric ducts (also called Müllerian ducts), which develop around the sixth week of embryogenesis. During development, the outer parts of the two ducts fuse, forming a single urogenital canal that will become the vagina, cervix and uterus.[14] The cervix grows in size at a smaller rate than the body of the uterus, so the relative size of the cervix over time decreases, decreasing from being much larger than the body of the uterus in fetal life, twice as large during childhood, and decreasing to its adult size, smaller than the uterus, after puberty.[10] Previously it was thought that during fetal development, the original squamous epithelium of the cervix is derived from the urogenital sinus and the original columnar epithelium is derived from the paramesonephric duct. The point at which these two original epithelia meet is called the original squamocolumnar junction.[15]: 15–16 New studies show, however, that all the cervical as well as large part of the vaginal epithelium are derived from Müllerian duct tissue and that phenotypic differences might be due to other causes.[16]
Histology

The endocervical mucosa is about 3 mm (0.12 in) thick and lined with a single layer of columnar mucous cells. It contains numerous tubular mucous glands, which empty viscous alkaline mucus into the lumen.[4] In contrast, the ectocervix is covered with nonkeratinized stratified squamous epithelium,[4] which resembles the squamous epithelium lining the vagina.[17]: 41 The junction between these two types of epithelia is called the squamocolumnar junction.[17]: 408–11 Underlying both types of epithelium is a tough layer of collagen.[18] The mucosa of the endocervix is not shed during menstruation. The cervix has more fibrous tissue, including collagen and elastin, than the rest of the uterus.[4]
.jpg.webp) The squamocolumnar junction of the cervix, with abrupt transition: The ectocervix, with its stratified squamous epithelium, is visible on the left. Simple columnar epithelium, typical of the endocervix, is visible on the right. A layer of connective tissue is visible under both types of epithelium.
The squamocolumnar junction of the cervix, with abrupt transition: The ectocervix, with its stratified squamous epithelium, is visible on the left. Simple columnar epithelium, typical of the endocervix, is visible on the right. A layer of connective tissue is visible under both types of epithelium.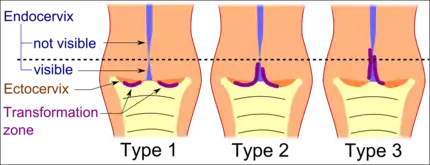 Transformation zone types:[19]
Transformation zone types:[19]
Type 1: Completely ectocervical (common under hormonal influence).
Type 2: Endocervical component but fully visible (common before puberty).
Type 3: Endocervical component, not fully visible (common after menopause).
In prepubertal girls, the functional squamocolumnar junction is present just within the cervical canal.[17]: 411 Upon entering puberty, due to hormonal influence, and during pregnancy, the columnar epithelium extends outward over the ectocervix as the cervix everts.[15]: 106 Hence, this also causes the squamocolumnar junction to move outwards onto the vaginal portion of the cervix, where it is exposed to the acidic vaginal environment.[15]: 106 [17]: 411 The exposed columnar epithelium can undergo physiological metaplasia and change to tougher metaplastic squamous epithelium in days or weeks,[17]: 25 which is very similar to the original squamous epithelium when mature.[17]: 411 The new squamocolumnar junction is therefore internal to the original squamocolumnar junction, and the zone of unstable epithelium between the two junctions is called the transformation zone of the cervix.[17]: 411 Histologically, the transformation zone is generally defined as surface squamous epithelium with surface columnar epithelium or stromal glands/crypts, or both.[20]
After menopause, the uterine structures involute and the functional squamocolumnar junction moves into the cervical canal.[17]: 41
Nabothian cysts (or Nabothian follicles) form in the transformation zone where the lining of metaplastic epithelium has replaced mucous epithelium and caused a strangulation of the outlet of some of the mucous glands.[17]: 410–411 A buildup of mucus in the glands forms Nabothian cysts, usually less than about 5 mm (0.20 in) in diameter,[4] which are considered physiological rather than pathological.[17]: 411 Both gland openings and Nabothian cysts are helpful to identify the transformation zone.[15]: 106
Function
Fertility
The cervical canal is a pathway through which sperm enter the uterus after being induced by estradiol after sexual intercourse,[21] and some forms of artificial insemination.[22] Some sperm remains in cervical crypts, infoldings of the endocervix, which act as a reservoir, releasing sperm over several hours and maximising the chances of fertilisation.[23] A theory states the cervical and uterine contractions during orgasm draw semen into the uterus.[21] Although the "upsuck theory" has been generally accepted for some years, it has been disputed due to lack of evidence, small sample size, and methodological errors.[24][25]
Some methods of fertility awareness, such as the Creighton model and the Billings method involve estimating a woman's periods of fertility and infertility by observing physiological changes in her body. Among these changes are several involving the quality of her cervical mucus: the sensation it causes at the vulva, its elasticity (Spinnbarkeit), its transparency, and the presence of ferning.[12]
Cervical mucus
Several hundred glands in the endocervix produce 20–60 mg of cervical mucus a day, increasing to 600 mg around the time of ovulation. It is viscous because it contains large proteins known as mucins. The viscosity and water content varies during the menstrual cycle; mucus is composed of around 93% water, reaching 98% at midcycle. These changes allow it to function either as a barrier or a transport medium to spermatozoa. It contains electrolytes such as calcium, sodium, and potassium; organic components such as glucose, amino acids, and soluble proteins; trace elements including zinc, copper, iron, manganese, and selenium; free fatty acids; enzymes such as amylase; and prostaglandins.[13] Its consistency is determined by the influence of the hormones estrogen and progesterone. At midcycle around the time of ovulation—a period of high estrogen levels— the mucus is thin and serous to allow sperm to enter the uterus and is more alkaline and hence more hospitable to sperm.[23] It is also higher in electrolytes, which results in the "ferning" pattern that can be observed in drying mucus under low magnification; as the mucus dries, the salts crystallize, resembling the leaves of a fern.[12] The mucus has a stretchy character described as Spinnbarkeit most prominent around the time of ovulation.[26]
At other times in the cycle, the mucus is thick and more acidic due to the effects of progesterone.[23] This "infertile" mucus acts as a barrier to keep sperm from entering the uterus.[27] Women taking an oral contraceptive pill also have thick mucus from the effects of progesterone.[23] Thick mucus also prevents pathogens from interfering with a nascent pregnancy.[28]
A cervical mucus plug, called the operculum, forms inside the cervical canal during pregnancy. This provides a protective seal for the uterus against the entry of pathogens and against leakage of uterine fluids. The mucus plug is also known to have antibacterial properties. This plug is released as the cervix dilates, either during the first stage of childbirth or shortly before.[29] It is visible as a blood-tinged mucous discharge.[30]
Childbirth
.svg.png.webp)
The cervix plays a major role in childbirth. As the fetus descends within the uterus in preparation for birth, the presenting part, usually the head, rests on and is supported by the cervix.[31] As labour progresses, the cervix becomes softer and shorter, begins to dilate, and withdraws to face the anterior of the body.[32] The support the cervix provides to the fetal head starts to give way when the uterus begins its contractions. During childbirth, the cervix must dilate to a diameter of more than 10 cm (3.9 in) to accommodate the head of the fetus as it descends from the uterus to the vagina. In becoming wider, the cervix also becomes shorter, a phenomenon known as effacement.[31]
Along with other factors, midwives and doctors use the extent of cervical dilation to assist decision making during childbirth.[33][34] Generally, the active first stage of labour, when the uterine contractions become strong and regular,[33] begins when the cervical dilation is more than 3–5 cm (1.2–2.0 in).[35][36] The second phase of labor begins when the cervix has dilated to 10 cm (4 in), which is regarded as its fullest dilation,[31] and is when active pushing and contractions push the baby along the birth canal leading to the birth of the baby.[34] The number of past vaginal deliveries is a strong factor in influencing how rapidly the cervix is able to dilate in labour.[31] The time taken for the cervix to dilate and efface is one factor used in reporting systems such as the Bishop score, used to recommend whether interventions such as a forceps delivery, induction, or Caesarean section should be used in childbirth.[31]
Cervical incompetence is a condition in which shortening of the cervix due to dilation and thinning occurs, before term pregnancy. Short cervical length is the strongest predictor of preterm birth.[32]
Contraception
Several methods of contraception involve the cervix. Cervical diaphragms are reusable, firm-rimmed plastic devices inserted by a woman prior to intercourse that cover the cervix. Pressure against the walls of the vagina maintain the position of the diaphragm, and it acts as a physical barrier to prevent the entry of sperm into the uterus, preventing fertilisation. Cervical caps are a similar method, although they are smaller and adhere to the cervix by suction. Diaphragms and caps are often used in conjunction with spermicides.[37] In one year, 12% of women using the diaphragm will undergo an unintended pregnancy, and with optimal use this falls to 6%.[38] Efficacy rates are lower for the cap, with 18% of women undergoing an unintended pregnancy, and 10–13% with optimal use.[39] Most types of progestogen-only pills are effective as a contraceptive because they thicken cervical mucus, making it difficult for sperm to pass along the cervical canal.[40] In addition, they may also sometimes prevent ovulation.[40] In contrast, contraceptive pills that contain both oestrogen and progesterone, the combined oral contraceptive pills, work mainly by preventing ovulation.[41] They also thicken cervical mucus and thin the lining of the uterus, enhancing their effectiveness.[41]
Clinical significance
Cancer
In 2008, cervical cancer was the third-most common cancer in women worldwide, with rates varying geographically from less than one to more than 50 cases per 100,000 women.[42] It is a leading cause of cancer-related death in poor countries, where delayed diagnosis leading to poor outcomes is common.[43] The introduction of routine screening has resulted in fewer cases of (and deaths from) cervical cancer, however this has mainly taken place in developed countries. Most developing countries have limited or no screening, and 85% of the global burden occurring there.[44]
Cervical cancer nearly always involves human papillomavirus (HPV) infection.[45][46] HPV is a virus with numerous strains, several of which predispose to precancerous changes in the cervical epithelium, particularly in the transformation zone, which is the most common area for cervical cancer to start.[47] HPV vaccines, such as Gardasil and Cervarix, reduce the incidence of cervical cancer, by inoculating against the viral strains involved in cancer development.[48]
Potentially precancerous changes in the cervix can be detected by cervical screening, using methods including a Pap smear (also called a cervical smear), in which epithelial cells are scraped from the surface of the cervix and examined under a microscope.[48] The colposcope, an instrument used to see a magnified view of the cervix, was invented in 1925. The Pap smear was developed by Georgios Papanikolaou in 1928.[49] A LEEP procedure using a heated loop of platinum to excise a patch of cervical tissue was developed by Aurel Babes in 1927.[50] In some parts of the developed world including the UK, the Pap test has been superseded with liquid-based cytology.[51]
A cheap, cost-effective and practical alternative in poorer countries is visual inspection with acetic acid (VIA).[43] Instituting and sustaining cytology-based programs in these regions can be difficult, due to the need for trained personnel, equipment and facilities and difficulties in follow-up. With VIA, results and treatment can be available on the same day. As a screening test, VIA is comparable to cervical cytology in accurately identifying precancerous lesions.[52]
A result of dysplasia is usually further investigated, such as by taking a cone biopsy, which may also remove the cancerous lesion.[48] Cervical intraepithelial neoplasia is a possible result of the biopsy and represents dysplastic changes that may eventually progress to invasive cancer.[53] Most cases of cervical cancer are detected in this way, without having caused any symptoms. When symptoms occur, they may include vaginal bleeding, discharge, or discomfort.[54]
Inflammation
Inflammation of the cervix is referred to as cervicitis. This inflammation may be of the endocervix or ectocervix.[55] When associated with the endocervix, it is associated with a mucous vaginal discharge and sexually transmitted infections such as chlamydia and gonorrhoea.[56] As many as half of pregnant women having a gonorrheal infection of the cervix are asymptomatic.[57] Other causes include overgrowth of the commensal flora of the vagina.[46] When associated with the ectocervix, inflammation may be caused by the herpes simplex virus. Inflammation is often investigated through directly visualising the cervix using a speculum, which may appear whiteish due to exudate, and by taking a Pap smear and examining for causal bacteria. Special tests may be used to identify particular bacteria. If the inflammation is due to a bacterium, then antibiotics may be given as treatment.[56]
Anatomical abnormalities
Cervical stenosis is an abnormally narrow cervical canal, typically associated with trauma caused by removal of tissue for investigation or treatment of cancer, or cervical cancer itself.[46][58] Diethylstilbestrol, used from 1938 to 1971 to prevent preterm labour and miscarriage, is also strongly associated with the development of cervical stenosis and other abnormalities in the daughters of the exposed women. Other abnormalities include: vaginal adenosis, in which the squamous epithelium of the ectocervix becomes columnar; cancers such as clear cell adenocarcinomas; cervical ridges and hoods; and development of a cockscomb cervix appearance,[59] which is the condition wherein, as the name suggests, the cervix of the uterus is shaped like a cockscomb. About one third of women born to diethylstilbestrol-treated mothers (i.e. in-utero exposure) develop a cockscomb cervix.[60]
Enlarged folds or ridges of cervical stroma (fibrous tissues) and epithelium constitute a cockscomb cervix.[61] Similarly, cockscomb polyps lining the cervix are usually considered or grouped into the same overarching description. It is in and of itself considered a benign abnormality; its presence, however is usually indicative of DES exposure, and as such women who experience these abnormalities should be aware of their increased risk of associated pathologies.[62][63][64]
Cervical agenesis is a rare congenital condition in which the cervix completely fails to develop, often associated with the concurrent failure of the vagina to develop.[65] Other congenital cervical abnormalities exist, often associated with abnormalities of the vagina and uterus. The cervix may be duplicated in situations such as bicornuate uterus and uterine didelphys.[66]
Cervical polyps, which are benign overgrowths of endocervical tissue, if present, may cause bleeding, or a benign overgrowth may be present in the cervical canal.[46] Cervical ectropion refers to the horizontal overgrowth of the endocervical columnar lining in a one-cell-thick layer over the ectocervix.[56]
In mammals
Female marsupials have paired uteri and cervices.[67][68] Most eutherian (placental) mammal species have a single cervix and single, bipartite or bicornuate uterus. Lagomorphs, rodents, aardvarks and hyraxes have a duplex uterus and two cervices.[69] Lagomorphs and rodents share many morphological characteristics and are grouped together in the clade Glires. Anteaters of the family myrmecophagidae are unusual in that they lack a defined cervix; they are thought to have lost the characteristic rather than other mammals developing a cervix on more than one lineage.[70] In domestic pigs, the cervix contains a series of five interdigitating pads that hold the boar's corkscrew-shaped penis during copulation.[71]
Etymology and pronunciation
The word cervix (/ˈsɜːrvɪks/) came to English from Latin, where it means "neck", and like its Germanic counterpart, it can refer not only to the neck [of the body] but also to an analogous narrowed part of an object. The cervix uteri (neck of the uterus) is thus the uterine cervix, but in English the word cervix used alone usually refers to it. Thus the adjective cervical may refer either to the neck (as in cervical vertebrae or cervical lymph nodes) or to the uterine cervix (as in cervical cap or cervical cancer).
Latin cervix came from the Proto-Indo-European root ker-, referring to a "structure that projects". Thus, the word cervix is linguistically related to the English word "horn", the Persian word for "head" (Persian: سر sar), the Greek word for "head" (Greek: κορυφή koruphe), and the Welsh and Romanian words for "deer" (Welsh: carw, Romanian: cerb).[72][73]
The cervix was documented in anatomical literature in at least the time of Hippocrates; cervical cancer was first described more than 2,000 years ago, with descriptions provided by both Hippocrates and Aretaeus.[49] However, there was some variation in word sense among early writers, who used the term to refer to both the cervix and the internal uterine orifice.[74] The first attested use of the word to refer to the cervix of the uterus was in 1702.[72]
References
Citations
- Myers KM, Feltovich H, Mazza E, Vink J, Bajka M, Wapner RJ, et al. (June 2015). "The mechanical role of the cervix in pregnancy". Journal of Biomechanics. Reproductive Biomechanics. 48 (9): 1511–1523. doi:10.1016/j.jbiomech.2015.02.065. PMC 4459908. PMID 25841293.
- "Human Papillomavirus (HPV) Vaccines". National Cancer Institute. Bethesda, MD. 29 December 2011. Retrieved 18 June 2014.
- Kurman RJ, ed. (1994). Blaustein's Pathology of the Female Genital Tract (4th ed.). New York, NY: Springer New York. pp. 185–201. ISBN 978-1-4757-3889-6.
- Gray H (1995). Williams PL (ed.). Gray's Anatomy (38th ed.). Churchill Livingstone. pp. 1870–73. ISBN 0-443-04560-7.
- Drake RL, Vogl W, Mitchell AW (2005). Gray's Anatomy for Students. Illustrations by Richardson P, Tibbitts R. Philadelphia, PA: Elsevier/Churchill Livingstone. pp. 415, 423. ISBN 978-0-8089-2306-0.
- Ovalle WK, Nahirney PC (2013). "Female Reproductive System". Netter's Essential Histology. Illustrations by Frank H. Netter, contributing illustrators, Joe Chovan, et al. (2nd ed.). Philadelphia, PA: Elsevier/Saunders. p. 416. ISBN 978-1-4557-0631-0.
- Gardner E, Gray DJ, O'Rahilly R (1969) [1960]. Anatomy: A Regional Study of Human Structure (3rd ed.). Philadelphia, PA: W.B. Saunders. pp. 495–98.
- Kurman RJ, ed. (2002). Blaustein's Pathology of the Female Genital Tract (5th ed.). Springer. p. 207.
- Daftary SN, Chakravari S (2011). Manual of Obstretics, 3/e. Elsevier. pp. 1–16. ISBN 978-81-312-2556-1.
- Ellis H (2011). "Anatomy of the uterus". Anaesthesia & Intensive Care Medicine. 12 (3): 99–101. doi:10.1016/j.mpaic.2010.11.005.
- Mould TA, Chow C (2005). "The Vascular, Neural and Lymphatic Anatomy of the Cervix". In Jordan JA, Singer A (eds.). The Cervix (2nd ed.). Oxford, United Kingdom: Blackwell Publishing. pp. 41–47. ISBN 9781405131377.
- Weschler T (2006). Taking charge of your fertility : the definitive guide to natural birth control, pregnancy achievement, and reproductive health (Revised ed.). New York, NY: Collins. pp. 59, 64. ISBN 978-0-06-088190-0.
- Sharif K, Olufowobi O (2006). "The structure chemistry and physics of human cervical mucus". In Jordan J, Singer A, Jones H, Shafi M (eds.). The Cervix (2nd ed.). Malden, MA: Blackwell Publishing. pp. 157–68. ISBN 978-1-4051-3137-7.
- Schoenwolf GC, Bleyl SB, Brauer PR, Francis-West PH (2009). ""Development of the Urogenital system"". Larsen's human embryology (4th ed.). Philadelphia, PA: Churchill Livingstone/Elsevier. ISBN 978-0-443-06811-9.
- McLean JM (November 2006). "Morphogenesis and Differentiation of the cervicovaginal epithelium". In Jordan J, Singer A, Jones H, Shafi M (eds.). The Cervix (2nd ed.). Wiley-Blackwell. ISBN 978-1-4051-3137-7.
- Reich O, Fritsch H (October 2014). "The developmental origin of cervical and vaginal epithelium and their clinical consequences: a systematic review". Journal of Lower Genital Tract Disease. 18 (4): 358–360. doi:10.1097/LGT.0000000000000023. PMID 24977630. S2CID 3060493.
- Beckmann CR, Herbert W, Laube D, Ling F, Smith R (March 2013). Obstetrics and Gynecology (7th ed.). pp. 408–11. ISBN 9781451144314.
- Young B (2006). Wheater's functional histology : a text and colour atlas (5th ed.). Edinburgh, United Kingdom: Churchill Livingstone/Elsevier. p. 376. ISBN 978-0-443-06850-8.
- International Federation for Cervical Pathology and Colposcopy (IFCPC) classification. References:
-"Transformation zone (TZ) and cervical excision types". Royal College of Pathologists of Australasia.
- Jordan J, Arbyn M, Martin-Hirsch P, Schenck U, Baldauf JJ, Da Silva D, et al. (December 2008). "European guidelines for quality assurance in cervical cancer screening: recommendations for clinical management of abnormal cervical cytology, part 1". Cytopathology. 19 (6): 342–354. doi:10.1111/j.1365-2303.2008.00623.x. PMID 19040546. S2CID 16462929. - Mukonoweshuro P, Oriowolo A, Smith M (June 2005). "Audit of the histological definition of cervical transformation zone". Journal of Clinical Pathology. 58 (6): 671. PMC 1770692. PMID 15917428.
- Guyton AC, Hall JE (2005). Textbook of Medical Physiology (11th ed.). Philadelphia, PA: W.B. Saunders. p. 1027. ISBN 978-0-7216-0240-0.
- "Demystifying IUI, ICI, IVI and IVF". Seattle Sperm Bank. 4 January 2014. Retrieved 9 November 2014.
- Brannigan RE, Lipshultz LI (2008). "Sperm Transport and Capacitation". The Global Library of Women's Medicine. doi:10.3843/GLOWM.10316.
- Levin RJ (November 2011). "The human female orgasm: a critical evaluation of its proposed reproductive functions". Sexual and Relationship Therapy. 26 (4): 301–14. doi:10.1080/14681994.2011.649692. S2CID 143550619.
- Borrow AP, Cameron NM (March 2012). "The role of oxytocin in mating and pregnancy". Hormones and Behavior. 61 (3): 266–276. doi:10.1016/j.yhbeh.2011.11.001. PMID 22107910. S2CID 45783934.
- Anderson M, Karasz A, Friedland S (November 2004). "Are vaginal symptoms ever normal? a review of the literature". MedGenMed. 6 (4): 49. PMC 1480553. PMID 15775876.
- Westinore A, Billings E (1998). The Billings Method: Controlling Fertility Without Drugs or Devices. Toronto, ON: Life Cycle Books. p. 37. ISBN 0-919225-17-9.
- Wagner G, Levin RJ (September 1980). "Electrolytes in vaginal fluid during the menstrual cycle of coitally active and inactive women". Journal of Reproduction and Fertility. 60 (1): 17–27. doi:10.1530/jrf.0.0600017. PMID 7431318.
- Becher N, Adams Waldorf K, Hein M, Uldbjerg N (May 2009). "The cervical mucus plug: structured review of the literature". Acta Obstetricia et Gynecologica Scandinavica. 88 (5): 502–513. doi:10.1080/00016340902852898. PMID 19330570. S2CID 23738950.
- Lowdermilk DL, Perry SE (2006). Maternity Nursing (7th ed.). Edinburgh, United Kingdom: Elsevier Mosby. p. 394. ISBN 978-0-323-03366-4.
- Cunningham F, Leveno K, Bloom S, Hauth J, Gilstrap L, Wenstrom K (2005). Williams obstetrics (22nd ed.). New York; Toronto: McGraw-Hill Professional. pp. 157–60, 537–39. ISBN 0-07-141315-4.
- Goldenberg RL, Culhane JF, Iams JD, Romero R (January 2008). "Epidemiology and causes of preterm birth". Lancet. 371 (9606): 75–84. doi:10.1016/S0140-6736(08)60074-4. PMC 7134569. PMID 18177778.
- <NICE (2007). Section 1.6, Normal labour: first stage
- NICE (2007). Section 1.7, Normal labour: second stage
- ACOG (2012). "Obstetric Data Definitions Issues and Rationale for Change" (PDF). Revitalize. Archived from the original (PDF) on 6 November 2013. Retrieved 4 November 2014.
- Su M, Hannah WJ, Willan A, Ross S, Hannah ME (October 2004). "Planned caesarean section decreases the risk of adverse perinatal outcome due to both labour and delivery complications in the Term Breech Trial". BJOG. 111 (10): 1065–1074. doi:10.1111/j.1471-0528.2004.00266.x. PMID 15383108. S2CID 10086313.
- NSW Family Planning (2009). Contraception : healthy choices : a contraceptive clinic in a book (2nd ed.). Sydney, New South Wales: UNSW Press. pp. 27–37. ISBN 978-1-74223-136-5.
- Trussell J (May 2011). "Contraceptive failure in the United States". Contraception. 83 (5): 397–404. doi:10.1016/j.contraception.2011.01.021. PMC 3638209. PMID 21477680.
- Trussell J, Strickler J, Vaughan B (May–Jun 1993). "Contraceptive efficacy of the diaphragm, the sponge and the cervical cap". Family Planning Perspectives. 25 (3): 100–5, 135. doi:10.2307/2136156. JSTOR 2136156. PMID 8354373.
- Your Guide to the progesterone-one pill (PDF). Family Planning Association (UK). pp. 3–4. ISBN 978-1-908249-53-1. Archived (PDF) from the original on 2014-03-27. Retrieved 9 November 2014.
- Your Guide to the combined pill (PDF). Family Planning Association (UK). January 2014. p. 4. ISBN 978-1-908249-50-0. Archived (PDF) from the original on 2013-11-02. Retrieved 9 November 2014.
- Arbyn M, Castellsagué X, de Sanjosé S, Bruni L, Saraiya M, Bray F, Ferlay J (December 2011). "Worldwide burden of cervical cancer in 2008". Annals of Oncology. 22 (12): 2675–2686. doi:10.1093/annonc/mdr015. PMID 21471563.
- World Health Organization (February 2014). "Fact sheet No. 297: Cancer". Retrieved 23 July 2014.
- Vaccarella S, Lortet-Tieulent J, Plummer M, Franceschi S, Bray F (October 2013). "Worldwide trends in cervical cancer incidence: impact of screening against changes in disease risk factors". European Journal of Cancer. 49 (15): 3262–3273. doi:10.1016/j.ejca.2013.04.024. PMID 23751569.
- Wahl CE (2007). Hardcore pathology. Philadelphia, PA: Lippincott Williams & Wilkins. p. 72. ISBN 9781405104982.
- Mitchell RS, Kumar V, Robbins SL, Abbas AK, Fausto N (2007). Robbins basic pathology (8th ed.). Saunders/Elsevier. pp. 716–21. ISBN 978-1-4160-2973-1.
- Lowe A, Stevens JS (2005). Human histology (3rd ed.). Philadelphia, PA; Toronto, ON: Elsevier Mosby. pp. 350–51. ISBN 0-323-03663-5.
- Fauci AS, Braunwald E, Kasper DL, Hauser SL, Longo DL, Jameson JL, Loscalzo J, eds. (2008). Harrison's Principles of Internal Medicine (17th ed.). New York [etc.]: McGraw-Hill Medical. pp. 608–09. ISBN 978-0-07-147692-8.
- Gasparini R, Panatto D (May 2009). "Cervical cancer: from Hippocrates through Rigoni-Stern to zur Hausen". Vaccine. 27 (Suppl 1): A4–A5. doi:10.1016/j.vaccine.2008.11.069. PMID 19480961.
- Diamantis A, Magiorkinis E, Androutsos G (November 2010). "Different strokes: Pap-test and Babes method are not one and the same". Diagnostic Cytopathology. 38 (11): 857–859. doi:10.1002/dc.21347. PMID 20973044. S2CID 823546.
- Gray W, Kocjan G, eds. (2010). Diagnostic Cytopathology. Churchill Livingstone. p. 613. ISBN 9780702048951.
- Sherris J, Wittet S, Kleine A, Sellors J, Luciani S, Sankaranarayanan R, Barone MA (September 2009). "Evidence-based, alternative cervical cancer screening approaches in low-resource settings". International Perspectives on Sexual and Reproductive Health. 35 (3): 147–154. doi:10.1363/3514709. PMID 19805020.
- Cannistra SA, Niloff JM (April 1996). "Cancer of the uterine cervix". The New England Journal of Medicine. 334 (16): 1030–1038. doi:10.1056/NEJM199604183341606. PMID 8598842.
- Colledge NR, Walker BR, Ralston SH, eds. (2010). Davidson's Principles and Practice of Medicine. Illustrated by Britton R (21st ed.). Edinburgh: Churchill Livingstone/Elsevier. p. 276. ISBN 978-0-7020-3084-0.
- Stamm W (2013). The Practitioner's Handbook for the Management of Sexually Transmitted Diseases. Seattle STD/HIV Prevention Training Center. pp. Chapter 7: Cervicitis. Archived from the original on 2013-06-22.
- Fauci AS, Braunwald E, Kasper DL, Hauser SL, Longo DL, Jameson JL, Loscalzo J, eds. (2008). Harrison's Principles of Internal Medicine (17th ed.). New York [etc.]: McGraw-Hill Medical. pp. 828–29. ISBN 978-0-07-147692-8.
- Kenner C (2014). Comprehensive neonatal nursing care (5th ed.). New York: Springer Publishing Company, LLC. ISBN 9780826109750. Access provided by the University of Pittsburgh.
- Valle RF, Sankpal R, Marlow JL, Cohen L (2002). "Cervical Stenosis: A Challenging Clinical Entity". Journal of Gynecologic Surgery. 18 (4): 129–43. doi:10.1089/104240602762555939.
- Casey PM, Long ME, Marnach ML (February 2011). "Abnormal cervical appearance: what to do, when to worry?". Mayo Clinic Proceedings. 86 (2): 147–50, quiz 151. doi:10.4065/mcp.2010.0512. PMC 3031439. PMID 21270291.
- Casey PM, Long ME, Marnach ML (February 2011). "Abnormal cervical appearance: what to do, when to worry?". Mayo Clinic Proceedings. 86 (2): 147–50, quiz 151. doi:10.4065/mcp.2010.0512. PMC 3031439. PMID 21270291.
- "Diethylstilbestrol (DES) Cervix". National Cancer Institute Visuals. National Cancer Institute. Retrieved 14 May 2015.
- Wingfield M (June 1991). "The daughters of stilboestrol". BMJ. 302 (6790): 1414–1415. doi:10.1136/bmj.302.6790.1414. PMC 1670127. PMID 2070103.
- Mittendorf R (June 1995). "Teratogen update: carcinogenesis and teratogenesis associated with exposure to diethylstilbestrol (DES) in utero". Teratology. 51 (6): 435–445. doi:10.1002/tera.1420510609. PMID 7502243.
- Herbst AL, Poskanzer DC, Robboy SJ, Friedlander L, Scully RE (February 1975). "Prenatal exposure to stilbestrol. A prospective comparison of exposed female offspring with unexposed controls". The New England Journal of Medicine. 292 (7): 334–339. doi:10.1056/NEJM197502132920704. PMID 1117962.
- Fujimoto VY, Miller JH, Klein NA, Soules MR (December 1997). "Congenital cervical atresia: report of seven cases and review of the literature". American Journal of Obstetrics and Gynecology. 177 (6): 1419–1425. doi:10.1016/S0002-9378(97)70085-1. PMID 9423745.
- Patton PE, Novy MJ, Lee DM, Hickok LR (June 2004). "The diagnosis and reproductive outcome after surgical treatment of the complete septate uterus, duplicated cervix and vaginal septum". American Journal of Obstetrics and Gynecology. 190 (6): 1669–75, discussion p.1675–78. doi:10.1016/j.ajog.2004.02.046. PMID 15284765.
- Tyndale-Biscoe CH (2005). Life of Marsupials. Csiro Publishing. ISBN 978-0-643-06257-3.
- Tyndale-Biscoe H, Renfree M (30 January 1987). Reproductive Physiology of Marsupials. Cambridge University Press. ISBN 978-0-521-33792-2.
- Feldhamer GA, Drickamer LC, Vessey SH, Merritt JF, Krajewski C (2007). Mammalogy: Adaptation, Diversity, Ecology. Baltimore, MD: JHU Press. p. 198. ISBN 9780801886959.
- Novacek MJ, Wyss AR (September 1986). "Higher-Level Relationships of the Recent Eutherian Orders: Morphological Evidence". Cladistics. 2 (4): 257–287. doi:10.1111/j.1096-0031.1986.tb00463.x. PMID 34949071. S2CID 85140444.
- "The Female - Swine Reproduction". livestocktrail.illinois.edu. Retrieved 2017-03-07.
- Harper D. "Cervix". Etymology Online. Retrieved 19 March 2014.
- Harper D. "Horn". Etymology Online. Retrieved 19 March 2014.
- Galen IJ, ed. (2011). Galen: On Diseases and Symptoms. Translated by Johnston I. Cambridge University Press. p. 247. ISBN 978-1-139-46084-2.
Cited texts
- "Intrapartum care: Care of healthy women and their babies during childbirth". NICE. September 2007. Archived from the original on 2014-04-26.
External links
 Media related to Cervix uteri at Wikimedia Commons
Media related to Cervix uteri at Wikimedia Commons

