Cytochrome b
Cytochrome b within both molecular and cell biology, is a protein found in the mitochondria of eukaryotic cells. It functions as part of the electron transport chain and is the main subunit of transmembrane cytochrome bc1 and b6f complexes.[1][2]
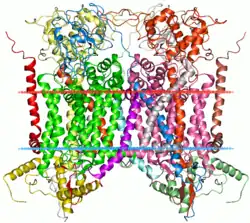 Mitochondrial cytochrome bc1 complex | |||||||||
| Identifiers | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Symbol | Cytochrom_B_N | ||||||||
| Pfam | PF00033 | ||||||||
| InterPro | IPR005797 | ||||||||
| PROSITE | PDOC00171 | ||||||||
| SCOP2 | 3bcc / SCOPe / SUPFAM | ||||||||
| TCDB | 3.D.3 | ||||||||
| OPM superfamily | 3 | ||||||||
| OPM protein | 3h1j | ||||||||
| CDD | cd00284 | ||||||||
| |||||||||
Function
In the mitochondrion of eukaryotes and in aerobic prokaryotes, cytochrome b is a component of respiratory chain complex III (EC 1.10.2.2) — also known as the bc1 complex or ubiquinol-cytochrome c reductase. In plant chloroplasts and cyanobacteria, there is an analogous protein, cytochrome b6, a component of the plastoquinone-plastocyanin reductase (EC 1.10.99.1), also known as the b6f complex. These complexes are involved in electron transport, the pumping of protons to create a proton-motive force (PMF). This proton gradient is used for the generation of ATP. These complexes play a vital role in cells.[3]
Structure
Cytochrome b/b6[4][5] is an integral membrane protein of approximately 400 amino acid residues that probably has 8 transmembrane segments. In plants and cyanobacteria, cytochrome b6 consists of two protein subunits encoded by the petB and petD genes. Cytochrome b/b6 non-covalently binds two heme groups, known as b562 and b566. Four conserved histidine residues are postulated to be the ligands of the iron atoms of these two heme groups.
Use in phylogenetics
Cytochrome b is commonly used as a region of mitochondrial DNA for determining phylogenetic relationships between organisms, due to its sequence variability. It is considered to be most useful in determining relationships within families and genera. Comparative studies involving cytochrome b have resulted in new classification schemes and have been used to assign newly described species to a genus as well as to deepen the understanding of evolutionary relationships.[6]
Clinical significance
Mutations in cytochrome b primarily result in exercise intolerance in human patients; though more rare, severe multi-system pathologies have also been reported.[7]
Single-point mutations in cytochrome b of Plasmodium falciparum and P. berghei are associated with resistance to the anti-malarial drug atovaquone.[8]
Human genes
Human genes encoding cytochrome b proteins include:
Fungicide target
Cyt b is targeted by the QoI class of fungicides, Fungicide Resistance Action Committee group 11. The cyt b mutations G143A and F129L provide resistance against the main body of group 11, although G143A does not work against metyltetraprole (11A).[9] G143A is significant in Botrytis cinerea in California strawberry production.[10]
References
- Howell N (August 1989). "Evolutionary conservation of protein regions in the proton motive cytochrome b and their possible roles in redox catalysis". J. Mol. Evol. 29 (2): 157–69. Bibcode:1989JMolE..29..157H. doi:10.1007/BF02100114. PMID 2509716. S2CID 7298013.
- Esposti MD, De Vries S, Crimi M, Ghelli A, Patarnello T, Meyer A (July 1993). "Mitochondrial cytochrome b: evolution and structure of the protein" (PDF). Biochim. Biophys. Acta. 1143 (3): 243–71. doi:10.1016/0005-2728(93)90197-N. PMID 8329437.
- Blankenship, Robert (2009). Molecular Mechanisms of Photosynthesis. Blackwell Publishing. pp. 124–132.
- Howell N (1989). "Evolutionary conservation of protein regions in the protonmotive cytochrome b and their possible roles in redox catalysis". J. Mol. Evol. 29 (2): 157–169. Bibcode:1989JMolE..29..157H. doi:10.1007/BF02100114. PMID 2509716. S2CID 7298013.
- Esposti MD, Crimi M, Ghelli A, Patarnello T, Meyer A, De Vries S (1993). "Mitochondrial cytochrome b: evolution and structure of the protein" (PDF). Biochim. Biophys. Acta. 1143 (3): 243–271. doi:10.1016/0005-2728(93)90197-N. PMID 8329437.
- Castresana, J. (2001). "Cytochrome b Phylogeny and the Taxonomy of Great Apes and Mammals". Molecular Biology and Evolution. 18 (4): 465–471. doi:10.1093/oxfordjournals.molbev.a003825. PMID 11264397.
- Blakely EL, Mitchell AL, Fisher N, Meunier B, Nijtmans LG, Schaefer AM, Jackson MJ, Turnbull DM, Taylor RW (July 2005). "A mitochondrial cytochrome b mutation causing severe respiratory chain enzyme deficiency in humans and yeast". FEBS J. 272 (14): 3583–92. doi:10.1111/j.1742-4658.2005.04779.x. PMID 16008558. S2CID 13938075.
- Siregar JE, Syafruddin D, Matsuoka H, Kita K, Marzuki S (June 2008). "Mutation underlying resistance of Plasmodium berghei to atovaquone in the quinone binding domain 2 (Qo(2)) of the cytochrome b gene". Parasitology International. 57 (2): 229–32. doi:10.1016/j.parint.2007.12.002. PMID 18248769.
- FRAC (Fungicide Resistance Action Committee) (March 2021). "FRAC Code List ©*2021: Fungal control agents sorted by cross resistance pattern and mode of action (including coding for FRAC Groups on product labels)" (PDF).
-
- Petrasch, Stefan; Knapp, Steven J.; van Kan, Jan A. L.; Blanco‐Ulate, Barbara (2019-04-04). "Grey mould of strawberry, a devastating disease caused by the ubiquitous necrotrophic fungal pathogen Botrytis cinerea". Molecular Plant Pathology. British Society for Plant Pathology (W-B). 20 (6): 877–892. doi:10.1111/mpp.12794. ISSN 1464-6722. PMC 6637890. PMID 30945788. S2CID 93002697.
- Cosseboom, Scott D.; Ivors, Kelly L.; Schnabel, Guido; Bryson, Patricia K.; Holmes, Gerald J. (2019). "Within-Season Shift in Fungicide Resistance Profiles of Botrytis cinerea in California Strawberry Fields". Plant Disease. American Phytopathological Society. 103 (1): 59–64. doi:10.1094/pdis-03-18-0406-re. ISSN 0191-2917. PMID 30422743. S2CID 205345358.
External links
- Cytochromes+b at the U.S. National Library of Medicine Medical Subject Headings (MeSH)