Echinoderm
An echinoderm (/ɪˈkaɪnəˌdɜːrm, ˈɛkə-/)[2] is any member of the phylum Echinodermata (/ɪˌkaɪnoʊˈdɜːrmətə/). The adults are recognisable by their (usually five-point) radial symmetry, and include starfish, brittle stars, sea urchins, sand dollars, and sea cucumbers, as well as the sea lilies or "stone lilies".[3] Adult echinoderms are found on the sea bed at every ocean depth, from the intertidal zone to the abyssal zone. The phylum contains about 7,000 living species, making it the second-largest grouping of deuterostomes, after the chordates. Echinoderms are the largest entirely marine phylum. The first definitive echinoderms appeared near the start of the Cambrian.
| Echinoderms Temporal range: | |
|---|---|
 | |
| Extant and extinct echinoderms of six classes: Fromia indica (Asteroidea); Ophiocoma scolopendrina (Ophiuroidea); Stomopneustes variolaris (Echinoidea); Oxycomanthus bennetti (Crinoidea); Actinopyga echinites (Holothuroidea); Ctenocystoidea. | |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Subkingdom: | Eumetazoa |
| Clade: | ParaHoxozoa |
| Clade: | Bilateria |
| Clade: | Nephrozoa |
| Superphylum: | Deuterostomia |
| Clade: | Ambulacraria |
| Phylum: | Echinodermata Bruguière, 1791 [ex Klein, 1734] |
| Type genus | |
| Echinus Linnaeus, 1758 | |
| Subphyla and classes[1] | |
|
Homalozoa † Gill & Caster, 1960
†=Extinct | |
The echinoderms are important both ecologically and geologically. Ecologically, there are few other groupings so abundant in the biotic desert of the deep sea, as well as shallower oceans. Most echinoderms are able to reproduce asexually and regenerate tissue, organs, and limbs; in some cases, they can undergo complete regeneration from a single limb. Geologically, the value of echinoderms is in their ossified skeletons, which are major contributors to many limestone formations, and can provide valuable clues as to the geological environment. They were the most used species in regenerative research in the 19th and 20th centuries. Further, some scientists hold that the radiation of echinoderms was responsible for the Mesozoic Marine Revolution.
Taxonomy and evolution
The name echinoderm is from Ancient Greek ἐχῖνος (ekhînos) 'hedgehog', and δέρμα (dérma) 'skin'.[4] Echinoderms are bilaterians, meaning that their ancestors were mirror-symmetric. Among the bilaterians, they belong to the deuterostome division, meaning that the blastopore, the first opening to form during embryo development, becomes the anus instead of the mouth.[5][6] The characteristics of adult echinoderms are the possession of a water vascular system with external tube feet and a calcareous endoskeleton consisting of ossicles connected by a mesh of collagen fibres.[7]
Phylogeny
Historically, taxonomists believed that the Ophiuroidea were sister to the Asteroidea, or that they were sister to the (Holothuroidea + Echinoidea).[8] However, a 2014 analysis of 219 genes from all classes of echinoderms revised the phylogenetic tree.[9] An independent analysis in 2015 of RNA transcriptomes from 23 species across all classes of echinoderms gave the same tree.[8]
- External phylogeny
The context of the echinoderms within the Bilateria is:[10]
| Bilateria |
| ||||||||||||||||||||||||||||||
- Internal phylogeny
| Echino‑ |
| ||||||||||||||||||||||||
| dermata |
Diversity
There are about 7,000 extant species of echinoderm as well as about 13,000 extinct species.[7][11] All echinoderms are marine, but they are found in habitats ranging from shallow intertidal areas to abyssal depths. Two main subdivisions are traditionally recognised: the more familiar motile Eleutherozoa, which encompasses the Asteroidea (starfish, with some 1,745 species), Ophiuroidea (brittle stars, with around 2,300 species), Echinoidea (sea urchins and sand dollars, with some 900 species) and Holothuroidea (sea cucumbers, with about 1,430 species); and the Pelmatozoa, some of which are sessile while others are motile. These consist of the Crinoidea (feather stars and sea lilies, with around 580 species) and the extinct blastoids and Paracrinoids.[12][13]
- The subphyla of echinoderms
 A brittle star, Ophionereis reticulata
A brittle star, Ophionereis reticulata A sea cucumber, Stichopus chloronotus, from Malaysia
A sea cucumber, Stichopus chloronotus, from Malaysia Starfish of varied colours
Starfish of varied colours A sea urchin, Strongylocentrotus purpuratus
A sea urchin, Strongylocentrotus purpuratus Crinoid on a coral reef
Crinoid on a coral reef
Fossil history
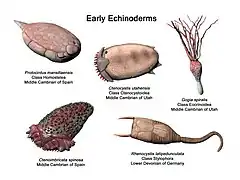
The oldest candidate echinoderm fossil is Arkarua from the Precambrian of Australia. These fossils are disc-like, with radial ridges on the rim and a five-pointed central depression marked with radial lines. However, the fossils have no stereom or internal structure indicating a water vascular system, so they cannot be conclusively identified.[14]
The first universally accepted echinoderms appear in the Lower Cambrian period; asterozoans appeared in the Ordovician, while the crinoids were a dominant group in the Paleozoic. Echinoderms left behind an extensive fossil record.[15] It is hypothesised that the ancestor of all echinoderms was a simple, motile, bilaterally symmetrical animal with a mouth, gut and anus. This ancestral organism adopted an attached mode of life with suspension feeding, and developed radial symmetry. Even so, the larvae of all echinoderms are bilaterally symmetrical, and all develop radial symmetry at metamorphosis. Like their ancestor, the starfish and crinoids still attach themselves to the seabed while changing to their adult form.[16]
The first echinoderms were non-motile,[16] but evolved into animals able to move freely. These soon developed endoskeletal plates with stereom structure, and external ciliary grooves for feeding.[17] The Paleozoic echinoderms were globular, attached to the substrate and were orientated with their oral surfaces facing upwards. These early echinoderms had ambulacral grooves extending down the side of the body, fringed on either side by brachioles, like the pinnules of a modern crinoid. Eventually, except for the crinoids, all the classes of echinoderms reversed their orientation to become mouth-downward. Before this happened, the podia probably had a feeding function, as they do in the crinoids today. The locomotor function of the podia came later, when the re-orientation of the mouth brought the podia into contact with the substrate for the first time.[16]
- Further information: Dibrachicystis
 The Ordovician cystoid Echinosphaerites from northeastern Estonia
The Ordovician cystoid Echinosphaerites from northeastern Estonia Fossil crinoid crowns
Fossil crinoid crowns
Anatomy and physiology
Echinoderms evolved from animals with bilateral symmetry. Although adult echinoderms possess pentaradial symmetry, their larvae are ciliated, free-swimming organisms with bilateral symmetry. Later, during metamorphosis, the left side of the body grows at the expense of the right side, which is eventually absorbed. The left side then grows in a pentaradially symmetric fashion, in which the body is arranged in five parts around a central axis.[18] Within the Asterozoa, there can be a few exceptions from the rule. Most starfish in the genus Leptasterias have six arms, although five-armed individuals can occur. The Brisingida too contain some six-armed species. Amongst the brittle stars, six-armed species such as Ophiothela danae, Ophiactis savignyi, and Ophionotus hexactis exists, and Ophiacantha vivipara often has more than six.[19]
Echinoderms have secondary radial symmetry in portions of their body at some stage of life, most likely an adaptation to a sessile or slow-moving existence.[20] Many crinoids and some seastars are symmetrical in multiples of the basic five; starfish such as Labidiaster annulatus possess up to fifty arms, while the sea-lily Comaster schlegelii has two hundred.[21]
Skin and skeleton
Echinoderms have a mesodermal skeleton in the dermis, composed of calcite-based plates known as ossicles. If solid, these would form a heavy skeleton, so they have a sponge-like porous structure known as stereom.[22][23] Ossicles may be fused together, as in the test of sea urchins, or may articulate to form flexible joints as in the arms of sea stars, brittle stars and crinoids. The ossicles may bear external projections in the form of spines, granules or warts and they are supported by a tough epidermis. Skeletal elements are sometimes deployed in specialized ways, such as the chewing organ called "Aristotle's lantern" in sea urchins, the supportive stalks of crinoids, and the structural "lime ring" of sea cucumbers.[18]
Although individual ossicles are robust and fossilize readily, complete skeletons of starfish, brittle stars and crinoids are rare in the fossil record. On the other hand, sea urchins are often well preserved in chalk beds or limestone. During fossilization, the cavities in the stereom are filled in with calcite that is continuous with the surrounding rock. On fracturing such rock, paleontologists can observe distinctive cleavage patterns and sometimes even the intricate internal and external structure of the test.[24]
The epidermis contains pigment cells that provide the often vivid colours of echinoderms, which include deep red, stripes of black and white, and intense purple.[25] These cells may be light-sensitive, causing many echinoderms to change appearance completely as night falls. The reaction can happen quickly: the sea urchin Centrostephanus longispinus changes colour in just fifty minutes when exposed to light.[26]
One characteristic of most echinoderms is a special kind of tissue known as catch connective tissue. This collagen-based material can change its mechanical properties under nervous control rather than by muscular means. This tissue enables a starfish to go from moving flexibly around the seabed to becoming rigid while prying open a bivalve mollusc or preventing itself from being extracted from a crevice. Similarly, sea urchins can lock their normally mobile spines upright as a defensive mechanism when attacked.[27][28]
The water vascular system
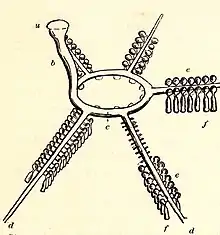
Echinoderms possess a unique water vascular system, a network of fluid-filled canals modified from the coelom (body cavity) that function in gas exchange, feeding, sensory reception and locomotion. This system varies between different classes of echinoderm but typically opens to the exterior through a sieve-like madreporite on the aboral (upper) surface of the animal. The madreporite is linked to a slender duct, the stone canal, which extends to a ring canal that encircles the mouth or oesophagus. The ring canal branches into a set of radial canals, which in asteroids extend along the arms, and in echinoids adjoin the test in the ambulacral areas. Short lateral canals branch off the radial canals, each one ending in an ampulla. Part of the ampulla can protrude through a pore (or a pair of pores in sea urchins) to the exterior, forming a podium or tube foot. The water vascular system assists with the distribution of nutrients throughout the animal's body; it is most visible in the tube feet which can be extended or contracted by the redistribution of fluid between the foot and the internal ampulla.[29][30]
The organisation of the water vascular system is somewhat different in ophiuroids, where the madreporite may be on the oral surface and the podia lack suckers.[31] In holothuroids, the system is reduced, often with few tube feet other than the specialised feeding tentacles, and the madreporite opens on to the coelom. Some holothuroids like the Apodida lack tube feet and canals along the body; others have longitudinal canals.[32] The arrangements in crinoids is similar to that in asteroids, but the tube feet lack suckers and are used in a back-and-forth wafting motion to pass food particles captured by the arms towards the central mouth. In the asteroids, the same motion is employed to move the animal across the ground.[33]
Other organs
Echinoderms possess a simple digestive system which varies according to the animal's diet. Starfish are mostly carnivorous and have a mouth, oesophagus, two-part stomach, intestine and rectum, with the anus located in the centre of the aboral body surface. With a few exceptions, the members of the order Paxillosida do not possess an anus.[34][35] In many species of starfish, the large cardiac stomach can be everted to digest food outside the body. Some other species are able to ingest whole food items such as molluscs.[36] Brittle stars, which have varying diets, have a blind gut with no intestine or anus; they expel food waste through their mouth.[37] Sea urchins are herbivores and use their specialised mouthparts to graze, tear and chew their food, mainly algae. They have an oesophagus, a large stomach and a rectum with the anus at the apex of the test.[38] Sea cucumbers are mostly detritivores, sorting through the sediment with modified tube feet around their mouth, the buccal tentacles. Sand and mud accompanies their food through their simple gut, which has a long coiled intestine and a large cloaca.[39] Crinoids are suspension feeders, passively catching plankton which drift into their outstretched arms. Boluses of mucus-trapped food are passed to the mouth, which is linked to the anus by a loop consisting of a short oesophagus and longer intestine.[40]
The coelomic cavities of echinoderms are complex. Aside from the water vascular system, echinoderms have a haemal coelom, a perivisceral coelom, a gonadal coelom and often also a perihaemal coelom.[41] During development, echinoderm coelom is divided into the metacoel, mesocoel and protocoel (also called somatocoel, hydrocoel and axocoel, respectively).[42] The water vascular system, haemal system and perihaemal system form the tubular coelomic system.[43] Echinoderms are unusual in having both a coelomic circulatory system (the water vascular system) and a haemal circulatory system, as most groups of animals have just one of the two.[44]
Haemal and perihaemal systems are derived from the original coelom, forming an open and reduced circulatory system. This usually consists of a central ring and five radial vessels. There is no true heart, and the blood often lacks any respiratory pigment. Gaseous exchange occurs via dermal branchiae or papulae in starfish, genital bursae in brittle stars, peristominal gills in sea urchins and cloacal trees in sea cucumbers. Exchange of gases also takes place through the tube feet. Echinoderms lack specialized excretory (waste disposal) organs and so nitrogenous waste, chiefly in the form of ammonia, diffuses out through the respiratory surfaces.[29]
The coelomic fluid contains the coelomocytes, or immune cells. There are several types of immune cells, which vary among classes and species. All classes possess a type of phagocytic amebocyte, which engulf invading particles and infected cells, aggregate or clot, and may be involved in cytotoxicity. These cells are usually large and granular, and are believed to be a main line of defense against potential pathogens.[45] Depending on the class, echinoderms may have spherule cells (for cytotoxicity, inflammation, and anti-bacterial activity), vibratile cells (for coelomic fluid movement and clotting), and crystal cells (which may serve for osmoregulation in sea cucumbers).[45][46] The coelomocytes secrete antimicrobial peptides against bacteria, and have a set of lectins and complement proteins as part of an innate immune system that is still being characterised.[47]
Echinoderms have a simple radial nervous system that consists of a modified nerve net of interconnected neurons with no central brain, although some do possess ganglia. Nerves radiate from central rings around the mouth into each arm or along the body wall; the branches of these nerves coordinate the movements of the organism and the synchronisation of the tube feet. Starfish have sensory cells in the epithelium and have simple eyespots and touch-sensitive tentacle-like tube feet at the tips of their arms. Sea urchins have no particular sense organs but do have statocysts that assist in gravitational orientation, and they too have sensory cells in their epidermis, particularly in the tube feet, spines and pedicellariae. Brittle stars, crinoids and sea cucumbers in general do not have sensory organs, but some burrowing sea cucumbers of the order Apodida have a single statocyst adjoining each radial nerve, and some have an eyespot at the base of each tentacle.[48]
The gonads at least periodically occupy much of the body cavities of sea urchins[49] and sea cucumbers, while the less voluminous crinoids, brittle stars and starfish have two gonads in each arm. While the ancestors of modern echinoderms are believed to have had one genital aperture, many organisms have multiple gonopores through which eggs or sperm may be released.[48]
Regeneration

Many echinoderms have great powers of regeneration. Many species routinely autotomize and regenerate arms and viscera. Sea cucumbers often discharge parts of their internal organs if they perceive themselves to be threatened, regenerating them over the course of several months. Sea urchins constantly replace spines lost through damage, while sea stars and sea lilies readily lose and regenerate their arms. In most cases, a single severed arm cannot grow into a new starfish in the absence of at least part of the disc.[50][51][52][53] However, in a few species a single arm can survive and develop into a complete individual, and arms are sometimes intentionally detached for the purpose of asexual reproduction.[51][52][53] During periods when they have lost their digestive tracts, sea cucumbers live off stored nutrients and absorb dissolved organic matter directly from the water.[54]
The regeneration of lost parts involves both epimorphosis and morphallaxis. In epimorphosis stem cells—either from a reserve pool or those produced by dedifferentiation—form a blastema and generate new tissues. Morphallactic regeneration involves the movement and remodelling of existing tissues to replace lost parts.[55] Direct transdifferentiation of one type of tissue to another during tissue replacement is also observed.[56]
Reproduction
Sexual reproduction
Echinoderms become sexually mature after approximately two to three years, depending on the species and the environmental conditions. Almost all species have separate male and female sexes, though some are hermaphroditic. The eggs and sperm cells are typically released into open water, where fertilisation takes place. The release of sperm and eggs is synchronised in some species, usually with regard to the lunar cycle. In other species, individuals may aggregate during the reproductive season, increasing the likelihood of successful fertilisation. Internal fertilisation has been observed in three species of sea star, three brittle stars and a deep water sea cucumber. Even at abyssal depths, where no light penetrates, echinoderms often synchronise their reproductive activity.[57]
Some echinoderms brood their eggs. This is especially common in cold water species where planktonic larvae might not be able to find sufficient food. These retained eggs are usually few in number and are supplied with large yolks to nourish the developing embryos. In starfish, the female may carry the eggs in special pouches, under her arms, under her arched body, or even in her cardiac stomach.[58] Many brittle stars are hermaphrodites; they often brood their eggs, usually in special chambers on their oral surfaces, but sometimes in the ovary or coelom.[59] In these starfish and brittle stars, development is usually direct to the adult form, without passing through a bilateral larval stage.[60] A few sea urchins and one species of sand dollar carry their eggs in cavities, or near their anus, holding them in place with their spines.[61] Some sea cucumbers use their buccal tentacles to transfer their eggs to their underside or back, where they are retained. In a very small number of species, the eggs are retained in the coelom where they develop viviparously, later emerging through ruptures in the body wall.[62] In some crinoids, the embryos develop in special breeding bags, where the eggs are held until sperm released by a male happens to find them.[63]
Asexual reproduction
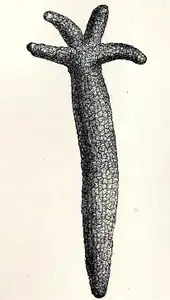
One species of seastar, Ophidiaster granifer, reproduces asexually by parthenogenesis.[64] In certain other asterozoans, adults reproduce asexually until they mature, then reproduce sexually. In most of these species, asexual reproduction is by transverse fission with the disc splitting in two. Both the lost disc area and the missing arms regrow, so an individual may have arms of varying lengths.[53][65] During the period of regrowth, they have a few tiny arms and one large arm, and are thus often known as "comets".[52][66]
Adult sea cucumbers reproduce asexually by transverse fission. Holothuria parvula uses this method frequently, splitting into two a little in front of the midpoint. The two halves each regenerate their missing organs over a period of several months, but the missing genital organs are often very slow to develop.[67]
The larvae of some echinoderms are capable of asexual reproduction. This has long been known to occur among starfish and brittle stars, but has more recently been observed in a sea cucumber, a sand dollar and a sea urchin.[68] This may be by autotomising parts that develop into secondary larvae, by budding, or by splitting transversely. Autotomised parts or buds may develop directly into fully formed larvae, or may pass through a gastrula or even a blastula stage. New larvae can develop from the preoral hood (a mound like structure above the mouth), the side body wall, the postero-lateral arms, or their rear ends.[68][69][70]
Cloning is costly to the larva both in resources and in development time. Larvae undergo this process when food is plentiful[71] or temperature conditions are optimal.[70] Cloning may occur to make use of the tissues that are normally lost during metamorphosis.[72] The larvae of some sand dollars clone themselves when they detect dissolved fish mucus, indicating the presence of predators.[70][72] Asexual reproduction produces many smaller larvae that escape better from planktivorous fish, implying that the mechanism may be an anti-predator adaptation.[73]
Larval development
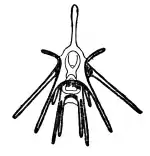
Development begins with a bilaterally symmetrical embryo, with a coeloblastula developing first. Gastrulation marks the opening of the "second mouth" that places echinoderms within the deuterostomes, and the mesoderm, which will host the skeleton, migrates inwards. The secondary body cavity, the coelom, forms by the partitioning of three body cavities. The larvae are often planktonic, but in some species the eggs are retained inside the female, while in some the female broods the larvae.[74][75]
The larvae pass through several stages, which have specific names derived from the taxonomic names of the adults or from their appearance. For example, a sea urchin has an 'echinopluteus' larva while a brittle star has an 'ophiopluteus' larva. A starfish has a 'bipinnaria' larva, which develops into a multi-armed 'brachiolaria' larva. A sea cucumber's larva is an 'auricularia' while a crinoid's is a 'vitellaria'. All these larvae are bilaterally symmetrical and have bands of cilia with which they swim; some, usually known as 'pluteus' larvae, have arms. When fully developed they settle on the seabed to undergo metamorphosis, and the larval arms and gut degenerate. The left hand side of the larva develops into the oral surface of the juvenile, while the right side becomes the aboral surface. At this stage the pentaradial symmetry develops.[74][76]
A plankton-eating larva, living and feeding in the water column, is considered to be the ancestral larval type for echinoderms, but in extant echinoderms, some 68% of species develop using a yolk-feeding larva.[12] The provision of a yolk-sac means that smaller numbers of eggs are produced, the larvae have a shorter development period and a smaller dispersal potential, but a greater chance of survival.[12]
Distribution and habitat
Echinoderms are globally distributed in almost all depths, latitudes and environments in the ocean. Adults are mainly benthic, living on the seabed, whereas larvae are often pelagic, living as plankton in the open ocean. Some holothuroid adults such as Pelagothuria are however pelagic.[77] Some crinoids are pseudo-planktonic, attaching themselves to floating logs and debris, although this behaviour was exercised most extensively in the Paleozoic, before competition from organisms such as barnacles restricted the extent of the behaviour.[78]
Mode of life
Locomotion

Echinoderms primarily use their tube feet to move about, though some sea urchins also use their spines. The tube feet typically have a tip shaped like a suction pad in which a vacuum can be created by contraction of muscles. This combines with some stickiness from the secretion of mucus to provide adhesion. The tube feet contract and relax in waves which move along the adherent surface, and the animal moves slowly along.[79]
Brittle stars are the most agile of the echinoderms. Any one of the arms can form the axis of symmetry, pointing either forwards or back. The animal then moves in a co-ordinated way, propelled by the other four arms. During locomotion, the propelling arms can made either snake-like or rowing movements.[80] Starfish move using their tube feet, keeping their arms almost still, including in genera like Pycnopodia where the arms are flexible. The oral surface is covered with thousands of tube feet which move out of time with each other, but not in a metachronal rhythm; in some way, however, the tube feet are coordinated, as the animal glides steadily along.[81] Some burrowing starfish have points rather than suckers on their tube feet and they are able to "glide" across the seabed at a faster rate.[82]
Sea urchins use their tube feet to move around in a similar way to starfish. Some also use their articulated spines to push or lever themselves along or lift their oral surfaces off the substrate. If a sea urchin is overturned, it can extend its tube feet in one ambulacral area far enough to bring them within reach of the substrate and then successively attach feet from the adjoining area until it is righted. Some species bore into rock, usually by grinding away at the surface with their mouthparts.[83]
.jpg.webp)
Sea cucumbers are generally sluggish animals. Many can move on the surface of the sea bed or burrow through sand or mud using peristaltic movements; some have short tube feet on their under surface with which they can creep along in the manner of a starfish. Some species drag themselves along using their buccal tentacles, while others manage to swim with peristaltic movements or rhythmic flexing. Many live in cracks, hollows and burrows and hardly move at all. Some deep water species are pelagic and can float in the water with webbed papillae forming sails or fins.[84]
The majority of crinoids are motile, but sea lilies are sessile and attached to hard substrates by stalks. Movement in most sea lilies is limited to bending their stems can bend and rolling and unrolling their arms; a few species can relocate themselves on the seabed by crawling. The sea feathers are unattached and usually live in crevices, under corals or inside sponges with their arms the only visible part. Some sea feathers emerge at night and perch themselves on nearby eminences to better exploit food-bearing currents. Many species can "walk" across the seabed, raising their body with the help of their arms, or swim using their arms. Most species of sea feather, however, are largely sedentary, seldom moving far from their chosen place of concealment.[85]
Feeding
The modes of feeding vary greatly between the different echinoderm taxa. Crinoids and some brittle stars tend to be passive filter-feeders,[86][87] enmeshing suspended particles from passing water. Most sea urchins are grazers;[88] sea cucumbers are deposit feeders;[89] and the majority of starfish are active hunters.[90]
Crinoids catch food particles using the tube feet on their outspread pinnules, move them into the ambulacral grooves, wrap them in mucus, and convey them to the mouth using the cilia lining the grooves.[86] The exact dietary requirements of crinoids have been little researched, but in the laboratory they can be fed with diatoms.[91]
Basket stars are suspension feeders, raising their branched arms to collect zooplankton, while other brittle stars use several methods of feeding. Some are suspension feeders, securing food particles with mucus strands, spines or tube feet on their raised arms. Others are scavengers and detritus feeders. Others again are voracious carnivores and able to lasso their waterborne prey with a sudden encirclement by their flexible arms. The limbs then bend under the disc to transfer the food to the jaws and mouth.[87]
Many sea urchins feed on algae, often scraping off the thin layer of algae covering the surfaces of rocks with their specialised mouthparts known as Aristotle's lantern. Other species devour smaller organisms, which they may catch with their tube feet. They may also feed on dead fish and other animal matter.[88] Sand dollars may perform suspension feeding and feed on phytoplankton, detritus, algal pieces and the bacterial layer surrounding grains of sand.[92]
Sea cucumbers are often mobile deposit or suspension feeders, using their buccal podia to actively capture food and then stuffing the particles individually into their buccal cavities. Others ingest large quantities of sediment, absorb the organic matter and pass the indigestible mineral particles through their guts. In this way they disturb and process large volumes of substrate, often leaving characteristic ridges of sediment on the sea bed. Some sea cucumbers live infaunally in burrows, anterior-end down and anus on the surface, swallowing sediment and passing it through their gut. Other burrowers live anterior-end up and wait for detritus to fall into the entrances of the burrows or rake in debris from the surface nearby with their buccal podia.[89]
Nearly all starfish are detritus feeders or carnivores, though a few are suspension feeders. Small fish landing on the upper surface may be captured by pedicilaria and dead animal matter may be scavenged but the main prey items are living invertebrates, mostly bivalve molluscs. To feed on one of these, the starfish moves over it, attaches its tube feet and exerts pressure on the valves by arching its back. When a small gap between the valves is formed, the starfish inserts part of its stomach into the prey, excretes digestive enzymes and slowly liquefies the soft body parts. As the adductor muscle of the bivalve relaxes, more stomach is inserted and when digestion is complete, the stomach is returned to its usual position in the starfish with its now liquefied bivalve meal inside it. Other starfish evert the stomach to feed on sponges, sea anemones, corals, detritus and algal films.[90]
Antipredator defence
.jpg.webp)
Despite their low nutrition value and the abundance of indigestible calcite, echinoderms are preyed upon by many organisms, including bony fish, sharks, eider ducks, gulls, crabs, gastropod molluscs, other echinoderms, sea otters, Arctic foxes and humans. Larger starfish prey on smaller ones; the great quantity of eggs and larva that they produce form part of the zooplankton, consumed by many marine creatures. Crinoids, on the other hand, are relatively free from predation.[93]
Antipredator defences include the presence of spines, toxins (inherent or delivered through the tube feet), and the discharge of sticky entangling threads by sea cucumbers. Although most echinoderm spines are blunt, those of the crown-of-thorns starfish are long and sharp and can cause a painful puncture wound as the epithelium covering them contains a toxin.[94] Because of their catch connective tissue, which can change rapidly from a flaccid to a rigid state, echinoderms are very difficult to dislodge from crevices. Some sea cucumbers have a cluster of cuvierian tubules which can be ejected as long sticky threads from their anus to entangle and permanently disable an attacker. Sea cucumbers occasionally defend themselves by rupturing their body wall and discharging the gut and internal organs.[95] Starfish and brittle stars may undergo autotomy when attacked, detaching an arm; this may distract the predator for long enough for the animal to escape. Some starfish species can swim away from danger.[96]
Ecology

Echinoderms are numerous invertebrates whose adults play an important role in benthic ecosystems, while the larvae are a major component of the plankton. Among the ecological roles of adults are the grazing of sea urchins, the sediment processing of heart urchins, and the suspension and deposit feeding of crinoids and sea cucumbers.[12][77] Some sea urchins can bore into solid rock, destabilising rock faces and releasing nutrients into the ocean. Coral reefs are also bored into in this way, but the rate of accretion of carbonate material is often greater than the erosion produced by the sea urchin.[97] Echinoderms sequester about 0.1 gigatonnes of carbon dioxide per year as calcium carbonate, making them important contributors in the global carbon cycle.[98]
Echinoderms sometimes have large population swings which can transform ecosystems. In 1983, for example, the mass mortality of the tropical sea urchin Diadema antillarum in the Caribbean caused a change from a coral-dominated reef system to an alga-dominated one.[99] Sea urchins are among the main herbivores on reefs and there is usually a fine balance between the urchins and the kelp and other algae on which they graze. A diminution of the numbers of predators (otters, lobsters and fish) can result in an increase in urchin numbers, causing overgrazing of kelp forests, resulting in an alga-denuded "urchin barren".[100] On the Great Barrier Reef, an unexplained increase in the numbers of crown-of-thorns starfish (Acanthaster planci), which graze on living coral tissue, has greatly increased coral mortality and reduced coral reef biodiversity.[101]
Use by humans
As food and medicine


In 2019, 129,052 tonnes of echinoderms were harvested. The majority of these were sea cucumbers (59,262 tonnes) and sea urchins (66,341 tonnes).[102] These are used mainly for food, but also in traditional Chinese medicine.[103] Sea cucumbers are considered a delicacy in some countries of southeast Asia; as such, they are in imminent danger of being over-harvested.[104] Popular species include the pineapple roller Thelenota ananas (susuhan) and the red sea cucumber Holothuria edulis. These and other species are colloquially known as bêche de mer or trepang in China and Indonesia. The sea cucumbers are boiled for twenty minutes and then dried both naturally and later over a fire which gives them a smoky tang. In China they are used as a basis for gelatinous soups and stews.[105] Both male and female gonads of sea urchins are consumed, particularly in Japan and France. The taste is described as soft and melting, like a mixture of seafood and fruit.[106][107] Sea urchin breeding trials have been undertaken to try to compensate for overexploitation.[108]
In research
Because of their robust larval growth, sea urchins are widely used in research, particularly as model organisms in developmental biology and ecotoxicology.[109][110][111][112] Strongylocentrotus purpuratus and Arbacia punctulata are used for this purpose in embryological studies.[113] The large size and the transparency of the eggs enables the observation of sperm cells in the process of fertilising ova.[109] The arm regeneration potential of brittle stars is being studied in connection with understanding and treating neurodegenerative diseases in humans.[114] Genomic data relevant to echinoderm model organisms are collected in Echinobase.[115]
Other uses
The calcareous tests or shells of echinoderms are used as a source of lime by farmers in areas where limestone is unavailable and some are used in the manufacture of fish meal.[116] Four thousand tons of the animals are used annually for these purposes. This trade is often carried out in conjunction with shellfish farmers, for whom the starfish pose a major threat by eating their cultured stock. Other uses for the starfish they recover include the manufacture of animal feed, composting and the preparation of dried specimens for the arts and craft trade.[114]
References
- Stöhr, Sabine (2014). "Echinodermata". WoRMS. World Register of Marine Species. Retrieved 23 February 2014.
- "echinoderm". Dictionary.com Unabridged (Online). n.d.
- Hall, Danielle (3 February 2022). "Echinoderms | Smithsonian Ocean". ocean.si.edu. Retrieved 31 March 2023.
- "echinoderm". Online Etymology Dictionary.
- Dorit, Walker & Barnes 1991, pp. 777–779.
- Fox, Richard. "Asterias forbesi". Invertebrate Anatomy OnLine. Lander University. Retrieved 19 May 2012.
- Wray, Gregory A. (1999). "Echinodermata: Spiny-skinned animals: sea urchins, starfish, and their allies". Tree of Life. Archived from the original on 23 May 2015. Retrieved 19 October 2012.
- Escriva, Hector; Reich, Adrian; Dunn, Casey; Akasaka, Koji; Wessel, Gary (2015). "Phylogenomic Analyses of Echinodermata Support the Sister Groups of Asterozoa and Echinozoa". PLOS ONE. 10 (3): e0119627. Bibcode:2015PLoSO..1019627R. doi:10.1371/journal.pone.0119627. PMC 4368666. PMID 25794146.
- Telford, M. J.; Lowe, C. J.; Cameron, C. B.; Ortega-Martinez, O.; Aronowicz, J.; Oliveri, P.; Copley, R. R. (2014). "Phylogenomic analysis of echinoderm class relationships supports Asterozoa". Proceedings of the Royal Society B: Biological Sciences. 281 (1786): 20140479. doi:10.1098/rspb.2014.0479. PMC 4046411. PMID 24850925.
- Edgecombe, Gregory D.; Giribet, Gonzalo; Dunn, Casey W.; Hejnol, Andreas; Kristensen, Reinhardt M.; Neves, Ricardo C.; Rouse, Greg W.; Worsaae, Katrine; Sørensen, Martin V. (June 2011). "Higher-level metazoan relationships: recent progress and remaining questions". Organisms, Diversity & Evolution. 11 (2): 151–172. doi:10.1007/s13127-011-0044-4. S2CID 32169826.
- "Animal Diversity Web - Echinodermata". University of Michigan Museum of Zoology. Retrieved 26 August 2012.
- Uthicke, Sven; Schaffelke, Britta; Byrne, Maria (1 January 2009). "A boom–bust phylum? Ecological and evolutionary consequences of density variations in echinoderms". Ecological Monographs. 79: 3–24. doi:10.1890/07-2136.1.
- Arnone, Maria Ina; Byrne, Maria; Martinez, Pedro (2015). "Echinodermata". Evolutionary Developmental Biology of Invertebrates 6. Vienna: Springer Vienna. pp. 1–58. doi:10.1007/978-3-7091-1856-6_1. ISBN 978-3-7091-1855-9.
- Smith, Dave (28 September 2005). "Vendian Animals: Arkarua". Retrieved 14 March 2013.
- Waggoner, Ben (16 January 1995). "Echinodermata: Fossil Record". Introduction to the Echinodermata. Museum of Paleontology: University of California at Berkeley. Retrieved 14 March 2013.
- Dorit, Walker & Barnes 1991, pp. 792–793.
- UCMP Berkeley. "Echinodermata: Morphology". University of California Museum of Paleontology. Retrieved 21 March 2011.
- Ruppert, Fox & Barnes 2004, p. 873.
- Byrne, Marie; O'Hara, Tim; CSIRO (2017). Australian echinoderms: biology, ecology and evolution. Clayton South, Victoria. pp. 322–324. ISBN 978-1-4863-0763-0. OCLC 960042778.
{{cite book}}: CS1 maint: location missing publisher (link) - Holló, Gábor; Novák, Mihály (2012). "The manoeuvrability hypothesis to explain the maintenance of bilateral symmetry in animal evolution". Biology Direct. 7 (1): 22. doi:10.1186/1745-6150-7-22. PMC 3438024. PMID 22789130.
- Messing, Charles. "Crown and calyx". Charles Messing's Crinoid Pages. Archived from the original on 29 October 2013. Retrieved 29 July 2012.
- Behrens, Peter; Bäuerlein, Edmund (2007). Handbook of Biomineralization: Biomimetic and bioinspired chemistry'. Wiley-VCH. p. 393. ISBN 978-3-527-31805-6.
- Brusca, Moore & Shuster 2016, pp. 979–980.
- Davies, A. Morley (1925). An Introduction to Palaeontology. Thomas Murby. pp. 240–241.
- Perillo, Margherita; Oulhen, Nathalie; Foster, Stephany; Spurrell, Maxwell; Calestani, Cristina; Wessel, Gary (19 August 2020). "Regulation of dynamic pigment cell states at single-cell resolution". eLife. 9. doi:10.7554/elife.60388. PMC 7455242. PMID 32812865.
- Weber, W.; Dambach, M. (April 1974). "Light-sensitivity of isolated pigment cells of the sea urchin Centrostephanus longispinus". Cell and Tissue Research. 148 (3): 437–440. doi:10.1007/BF00224270. PMID 4831958. S2CID 1669387.
- Motokawa, Tatsuo (May 1984). "Connective tissue catch in echinoderms". Biological Reviews. 59 (2): 255–270. doi:10.1111/j.1469-185X.1984.tb00409.x. S2CID 86391676.
- Brusca, Moore & Shuster 2016, p. 980.
- Dorit, Walker & Barnes 1991, pp. 780–791.
- Brusca, Moore & Shuster 2016, pp. 980–982.
- Dorit, Walker & Barnes 1991, pp. 784–785.
- Brusca, Moore & Shuster 2016, p. 982.
- Dorit, Walker & Barnes 1991, pp. 790–793.
- Jangoux, Michel (1982). "11. Digestive Systems". In Jangoux, Michel; Lawrence, John M. (eds.). Echinoderm Nutrition. Rotterdam: A. A. Balkema. p. 244. ISBN 978-90-6191-080-0.
- Rigby, P. Robin; Iken, Katrin; Shirayama, Yoshihisa (2007). Sampling Biodiversity in Coastal Communities: NaGISA Protocols for Seagrass and Macroalgal Habitats. NUS Press. p. 44. ISBN 978-9971693688.
- Ruppert, Fox & Barnes 2004, p. 885.
- Ruppert, Fox & Barnes 2004, p. 891.
- Ruppert, Fox & Barnes 2004, pp. 902–904.
- Ruppert, Fox & Barnes 2004, p. 912.
- Ruppert, Fox & Barnes 2004, p. 920.
- Moore, Janet; Overhill, Raith (2006). An Introduction to the Invertebrates (2nd ed.). Cambridge University Press. p. 245. ISBN 978-0521674065.
- Hickman, C. Jr.; Roberts, L.; Larson, A. (2003). Animal Diversity. Granite Hill Publishers. p. 271. ISBN 978-0071195492. OCLC 1148032077.
- "Macrobenthos of the North Sea - Echinodermata > Introduction". etibioinformatics.nl.
- Nielsen, Claus (2012). Animal Evolution: Interrelationships of the Living Phyla (3rd ed.). Oxford University Press. p. 78. ISBN 978-0199606023.
- Ramirez-Gomez, Fransisco (27 September 2010). "Echinoderm Immunity". Invertebrate Survival Journal.
- Smith, Courtney (January 2010). "Echinoderm Immunity". Invertebrate Immunity. Advances in Experimental Medicine and Biology. Vol. 708. pp. 260–301. doi:10.1007/978-1-4419-8059-5_14. ISBN 978-1-4419-8058-8. PMID 21528703.
- Smith, L. Courtney; Ghosh, Julie; Buckley, Katherine M.; et al. (2010). "Echinoderm Immunity". Invertebrate Immunity. Advances in Experimental Medicine and Biology. Vol. 708. Boston, Massachusetts: Springer. pp. 260–301. doi:10.1007/978-1-4419-8059-5_14. ISBN 978-1-4419-8058-8. PMID 21528703.
- Ruppert, Fox & Barnes 2004, pp. 872–929.
- James, Philip; Siikavuopio, Sten; Johansson, Gunhild Seljehaug (2018). "A Guide to the Sea Urchin Reproductive Cycle and Staging Sea Urchin Gonad Samples" (PDF). Nofima. Archived (PDF) from the original on 9 October 2022. Retrieved 21 June 2022.
The GI [% of body weight made up by the gonad] of urchins in the wild can vary hugely and can be less than 1% or as high as 20%, whilst for cultured sea urchins GI values can be as high as 35%
- Edmondson, C.H. (1935). "Autotomy and regeneration of Hawaiian starfishes" (PDF). Bishop Museum Occasional Papers. 11 (8): 3–20. Archived (PDF) from the original on 9 October 2022.
- McAlary, Florence A. (1993). Population Structure and Reproduction of the Fissiparous Seastar, Linckia columbiae Gray, on Santa Catalina Island, California. 3rd California Islands Symposium. National Park Service. Retrieved 15 April 2012.
- See last paragraph in review above AnalysisHotchkiss, Frederick H. C. (1 June 2000). "On the Number of Rays in Starfish". American Zoologist. 40 (3): 340–354. doi:10.1093/icb/40.3.340.
- Fisher, W. K. (1 March 1925). "Asexual Reproduction in the Starfish, Sclerasterias" (PDF). Biological Bulletin. 48 (3): 171–175. doi:10.2307/1536659. JSTOR 1536659. Archived (PDF) from the original on 9 October 2022. Retrieved 15 July 2011.
- Dobson, W. E.; S. E. Stancyk; L. A. Clements; R. M. Showman (1 February 1991). "Nutrient Translocation during Early Disc Regeneration in the Brittlestar Microphiopholis gracillima (Stimpson) (Echinodermata: Ophiuroidea)". Biol Bull. 180 (1): 167–184. doi:10.2307/1542439. JSTOR 1542439. PMID 29303627. Retrieved 14 July 2011.
- Candia-Carnevali, M. D.; Thorndyke, M. C.; Matranga, V. (2009). "Regenerating Echinoderms: A Promise to Understand Stem Cells Potential". In Rinkevich, B.; Matranga, V. (eds.). Stem Cells in Marine Organisms. Dordrecht: Springer. pp. 165–186. doi:10.1007/978-90-481-2767-2_7. ISBN 978-90-481-2766-5.
- Mashanov, Vladimir S.; Igor Yu. Dolmatov; Thomas Heinzeller (1 December 2005). "Transdifferentiation in Holothurian Gut Regeneration". The Biological Bulletin. 209 (3): 184–193. doi:10.2307/3593108. JSTOR 3593108. PMID 16382166. S2CID 27847466.
- Young, Craig M.; Eckelbarger, Kevin J. (1994). Reproduction, Larval Biology, and Recruitment of the Deep-Sea Benthos. Columbia University Press. pp. 179–194. ISBN 0231080042.
- Ruppert, Fox & Barnes 2004, pp. 887–888.
- Ruppert, Fox & Barnes 2004, p. 895.
- Ruppert, Fox & Barnes 2004, p. 888.
- Ruppert, Fox & Barnes 2004, p. 908.
- Ruppert, Fox & Barnes 2004, p. 916.
- Ruppert, Fox & Barnes 2004, p. 922.
- Yamaguchi, M.; J. S. Lucas (1984). "Natural parthenogenesis, larval and juvenile development, and geographical distribution of the coral reef asteroid Ophidiaster granifer". Marine Biology. 83 (1): 33–42. doi:10.1007/BF00393083. S2CID 84475593.
- McGovern, Tamara M. (5 April 2002). "Patterns of sexual and asexual reproduction in the brittle star Ophiactis savignyi in the Florida Keys" (PDF). Marine Ecology Progress Series. 230: 119–126. Bibcode:2002MEPS..230..119M. doi:10.3354/meps230119. Archived (PDF) from the original on 9 October 2022.
- Monks, Sarah P. (1 April 1904). "Variability and Autotomy of Phataria". Proceedings of the Academy of Natural Sciences of Philadelphia. 56 (2): 596–600. JSTOR 4063000.
- Kille, Frank R. (1942). "Regeneration of the Reproductive System Following Binary Fission in the Sea-Cucumber, Holothuria parvula (Selenka)" (PDF). Biological Bulletin. 83 (1): 55–66. doi:10.2307/1538013. JSTOR 1538013. Archived (PDF) from the original on 9 October 2022.
- Eaves, Alexandra A.; Palmer, A. Richard (11 September 2003). "Reproduction: Widespread cloning in echinoderm larvae". Nature. 425 (6954): 146. Bibcode:2003Natur.425..146E. doi:10.1038/425146a. hdl:10402/era.17195. PMID 12968170. S2CID 4430104.
- Jaeckle, William B. (1 February 1994). "Multiple Modes of Asexual Reproduction by Tropical and Subtropical Sea Star Larvae: An Unusual Adaptation for Genet Dispersal and Survival" (PDF). Biological Bulletin. 186 (1): 62–71. doi:10.2307/1542036. JSTOR 1542036. PMID 29283296. Archived (PDF) from the original on 9 October 2022. Retrieved 13 July 2011.
- Vaughn, Dawn (October 2009). "Predator-Induced Larval Cloning in the Sand Dollar Dendraster excentricus: Might Mothers Matter?". Biological Bulletin. 217 (2): 103–114. doi:10.1086/BBLv217n2p103. PMID 19875816. S2CID 26030855.
- McDonald, Kathryn A.; Dawn Vaughn (1 August 2010). "Abrupt Change in Food Environment Induces Cloning in Plutei of Dendraster excentricus". Biological Bulletin. 219 (1): 38–49. doi:10.1086/BBLv219n1p38. PMID 20813988. S2CID 24422314. Retrieved 16 July 2011.
- Vaughn, Dawn; Richard R. Strathmann (14 March 2008). "Predators induce cloning in echinoderm larvae". Science. 319 (5869): 1503. Bibcode:2008Sci...319.1503V. doi:10.1126/science.1151995. JSTOR 40284699. PMID 18339931. S2CID 206509992. Retrieved 16 July 2011.
- Vaughn, Dawn (March 2010). "Why run and hide when you can divide? Evidence for larval cloning and reduced larval size as an adaptive inducible defense". Marine Biology. 157 (6): 1301–1312. doi:10.1007/s00227-010-1410-z. S2CID 84336247.
- Dorit, Walker & Barnes 1991, p. 778.
- Brusca, Moore & Shuster 2016, pp. 997–998.
- van Egmond, Wim (1 July 2000). "Gallery of Echinoderm Larvae". Microscopy UK. Retrieved 2 February 2013.
- Brusca, Moore & Shuster 2016, p. 968.
- Xiaofeng, Wang; Hagdorn, Hans; Chuanshang, Wang (September 2006). "Pseudoplanktonic lifestyle of the Triassic crinoid Traumatocrinus from Southwest China". Lethaia. 39 (3): 187–193. doi:10.1080/00241160600715321.
- Smith, J. E. (1937). "The structure and function of the tube feet in certain echinoderms" (PDF). Journal of the Marine Biological Association of the United Kingdom. 22 (1): 345–357. doi:10.1017/S0025315400012042. S2CID 55933156. Archived from the original (PDF) on 15 November 2013.
- Astley, H. C. (2012). "Getting around when you're round: Quantitative analysis of the locomotion of the blunt-spined brittle star, Ophiocoma echinata". Journal of Experimental Biology. 215 (11): 1923–1929. doi:10.1242/jeb.068460. PMID 22573771.
- Brusca, Moore & Shuster 2016, pp. 982–983.
- "Sand star - Luidia foliolata". Sea Stars of the Pacific Northwest. Archived from the original on 9 September 2012. Retrieved 26 September 2012.
- Ruppert, Fox & Barnes 2004, pp. 899–900.
- Ruppert, Fox & Barnes 2004, pp. 911–912.
- Messing, Charles. "The crinoid feeding mechanism". Charles Messing's Crinoid Pages. Archived from the original on 14 January 2013. Retrieved 26 July 2012.
- Barnes, Robert D. (1982). Invertebrate Zoology. Holt-Saunders International. pp. 997–1007. ISBN 0-03-056747-5.
- Ruppert, Fox & Barnes 2004, p. 893.
- Carefoot, Tom. "Learn about sea urchins: feeding, nutrition & growth". A Snail's Odyssey. Archived from the original on 22 March 2013. Retrieved 23 February 2013.
- Ruppert, Fox & Barnes 2004, p. 914.
- Ruppert, Fox & Barnes 2004, pp. 884–885.
- Carefoot, Tom. "Learn about feather stars: feeding & growth". A Snail's Odyssey. Archived from the original on 12 January 2013. Retrieved 23 February 2013.
- Carefoot, Tom. "Learn about sand dollars: feeding & growth". A Snail's Odyssey. Archived from the original on 19 October 2012. Retrieved 23 February 2013.
- Miller, John E. "Echinoderm: role in nature". Encyclopædia Britannica. Retrieved 23 June 2022.
- Dorit, Walker & Barnes 1991, p. 779.
- Dorit, Walker & Barnes 1991, pp. 789–790.
- Mladenov, Philip V.; Igdoura, Suleiman; Asotra, Satish; Burke, Robert D. (1 April 1989). "Purification and partial characterisation of an autotomy-promoting factor from the sea star Pycnopodia Helianthoides". The Biological Bulletin. 176 (2): 169–175. doi:10.2307/1541585. JSTOR 1541585.
- Herrera-Escalante, T.; López-Pérez, R. A.; Leyte-Morales, G. E. (2005). "Bioerosion caused by the sea urchin Diadema mexicanum (Echinodermata: Echinoidea) at Bahías de Huatulco, Western Mexico". Revista de Biología Tropical. 53 (3): 263–273. PMID 17469255.
- Lebrato, Mario; Iglesias-Rodríguez, Debora; Feely, Richard A.; et al. (2010). "Global contribution of echinoderms to the marine carbon cycle: CaCO3 budget and benthic compartments". Ecological Monographs. 80 (3): 441–467. doi:10.1890/09-0553.1.
- Osborne, Patrick L. (2000). Tropical Ecosystem and Ecological Concepts. Cambridge University Press. p. 464. ISBN 0-521-64523-9.
- Lawrence, J. M. (1975). "On the relationships between marine plants and sea urchins" (PDF). Oceanographic Marine Biological Annual Review. 13: 213–286. Archived from the original (PDF) on 5 February 2011.
- Birkeland, Charles; Lucas, John S. (1990). "Effects of A. planci on Coral-Reef Communities". Acanthaster planci: Major Management Problem of Coral Reefs. Florida: CRC Press. pp. 148–159. ISBN 0849365996.
- "B-76 Sea-urchins and other echinoderms Capture production by species, fishing areas and countries or areas" (PDF). FAO. pp. 463–465. Archived (PDF) from the original on 9 October 2022. Retrieved 23 June 2022.
- Pangestuti, Ratih; Arifin, Zainal (2018). "Medicinal and health benefit effects of functional sea cucumbers". Journal of Traditional and Complementary Medicine. 8 (3): 341–351. doi:10.1016/j.jtcme.2017.06.007. PMC 6035309. PMID 29992105.
- "Sea Cucumbers Threatened by Asian Trade". The New York Times. April 2009. Retrieved 1 April 2009.
- Wikisource:1911 Encyclopædia Britannica/Bêche-de-Mer
- Lawrence, John M. (2001). "The edible sea urchins". In John M. Lawrence (ed.). Edible Sea Urchins: Biology and Ecology. Developments in Aquaculture and Fisheries Science. Vol. 37. pp. 1–4. doi:10.1016/S0167-9309(01)80002-8. ISBN 978-0444503909.
- Davidson, Alan (2014) Oxford Companion to Food. Oxford University Press, 3rd edition. p. 730.
- Sartori D., Scuderi A.; Sansone G.; Gaion A. (February 2015). "Echinoculture: the rearing of Paracentrotus lividus in a recirculating aquaculture system—experiments of artificial diets for the maintenance of sexual maturation". Aquaculture International. 23: 111–125. doi:10.1007/s10499-014-9802-6. S2CID 18856183.
- "Insight from the Sea Urchin". Microscope Imaging Station. Exploratorium. Archived from the original on 5 February 2018. Retrieved 23 June 2022.
- Sartori, D.; Gaion, A. (2015). "Toxicity of polyunsaturated aldehydes of diatoms to Indo-Pacific bioindicator organism Echinometra mathaei". Drug and Chemical Toxicology. 39 (2): 124–8. doi:10.3109/01480545.2015.1041602. PMID 25945412. S2CID 207437380.
- Gaion, A.; Scuderi, A.; Pellegrini, D.; Sartori, D. (2013). "Arsenic Exposure Affects Embryo Development of Sea Urchin, Paracentrotus lividus (Lamarck, 1816)". Bulletin of Environmental Contamination and Toxicology. 39 (2): 124–8. doi:10.3109/01480545.2015.1041602. PMID 25945412. S2CID 207437380.
- Hart, M. W. (January–February 2002). "Life history evolution and comparative developmental biology of echinoderms". Evolution and Development. 4 (1): 62–71. doi:10.1046/j.1525-142x.2002.01052.x. PMID 11868659. S2CID 39175905.
- Longo, F. J.; Anderson, E. (June 1969). "Sperm differentiation in the sea urchins Arbacia punctulata and Strongylocentrotus purpuratus". Journal of Ultrastructure Research. 27 (5): 486–509. doi:10.1016/S0022-5320(69)80046-8. PMID 5816822.
- Barkhouse, C.; Niles, M.; Davidson, L. A. (2007). "A literature review of sea star control methods for bottom and off bottom shellfish cultures" (PDF). Canadian Industry Report of Fisheries and Aquatic Sciences. 279 (7): 14–15. Archived from the original (PDF) on 22 May 2013.
- Arshinoff, Bradley I; Cary, Gregory A; Karimi, Kamran; Foley, Saoirse; Agalakov, Sergei; Delgado, Francisco; Lotay, Vaneet S; Ku, Carolyn J; Pells, Troy J; Beatman, Thomas R; Kim, Eugene; Cameron, R Andrew; Vize, Peter D; Telmer, Cheryl A; Croce, Jenifer C; Ettensohn, Charles A; Hinman, Veronica F (7 January 2022). "Echinobase: leveraging an extant model organism database to build a knowledgebase supporting research on the genomics and biology of echinoderms". Nucleic Acids Research. 50 (D1): D970–D979. doi:10.1093/nar/gkab1005. PMC 8728261. PMID 34791383.
- "Sea stars". Wild Singapore. Retrieved 4 February 2013.
Cited texts
- Brusca, Richard C.; Moore, Wendy; Shuster, Stephen M. (2016). Invertebrates (3rd ed.). Sunderland, Massachusetts. ISBN 978-1-60535-375-3. OCLC 928750550.
{{cite book}}: CS1 maint: location missing publisher (link) - Dorit, R. L.; Walker, W. F.; Barnes, R. D. (1991). Zoology, International edition. Saunders College Publishing. ISBN 978-0-03-030504-7.
- Ruppert, Edward E.; Fox, Richard, S.; Barnes, Robert D. (2004). Invertebrate Zoology (7th ed.). Cengage Learning. ISBN 81-315-0104-3.
{{cite book}}: CS1 maint: multiple names: authors list (link)
External links
- The Echinoid Directory from the Natural History Museum
- Echinodermata from the Tree of Life Web Project
- Echinoderms of the North Sea Archived 13 April 2008 at the Wayback Machine
- Larval Echinodermata Fact Sheet
_001.png.webp)

.jpg.webp)