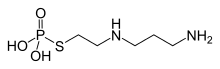
Amifostine
Amifostine (ethiofos) is a cytoprotective adjuvant used in cancer chemotherapy and radiotherapy involving DNA-binding chemotherapeutic agents. It is marketed by Clinigen Group under the trade name Ethyol.
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | complete |
| Elimination half-life | 8 minutes |
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.161.827 |
| Chemical and physical data | |
| Formula | C5H15N2O3PS |
| Molar mass | 214.22 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Indications
Amifostine is used therapeutically to reduce the incidence of neutropenia-related fever and infection induced by DNA-binding chemotherapeutic agents including alkylating agents (e.g. cyclophosphamide) and platinum-containing agents (e.g. cisplatin). It is also used to decrease the cumulative nephrotoxicity associated with platinum-containing agents. Amifostine is also indicated to reduce the incidence of xerostomia in patients undergoing radiotherapy for head and neck cancer.
Amifostine was originally indicated to reduce the cumulative renal toxicity from cisplatin in non-small cell lung cancer. However, while nephroprotection was observed, the probability that amifostine could protect tumors could not be excluded. Additional data have shown that amifostine-mediated tumor protection, in any clinical scenario, is unlikely.
Pharmacokinetics
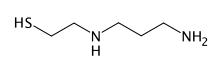
Amifostine is an organic thiophosphate prodrug which is hydrolysed in vivo by alkaline phosphatase to the active cytoprotective thiol metabolite, WR-1065. The selective protection of non-malignant tissues is believed to be due to higher alkaline phosphatase activity, higher pH, and vascular permeation of normal tissues.
Amifostine can be administered intravenously or subcutaneously after reconstitution with normal saline. Infusions lasting less than 15 minutes decrease the risk of adverse effects. The patient should be well-hydrated prior to administration.
Mechanism of action
Inside cells, amifostine detoxifies reactive metabolites of platinum and alkylating agents, as well as scavenges free radicals.[1][2] Other possible effects include accelerated DNA repair,[1] induction of cellular hypoxia,[1] inhibition of apoptosis,[2] alteration of gene expression[2] and modification of enzyme activity.[2] Amifostine is believed to radioprotect normal tissue via Warburg-type effects.[3]
Adverse effects
Common side effects of amifostine include hypocalcemia, diarrhea, nausea, vomiting, sneezing, somnolence, and hiccups. Serious side effects include: hypotension (found in 62% of patients), erythema multiforme, Stevens–Johnson syndrome and toxic epidermal necrolysis, immune hypersensitivity syndrome, erythroderma, anaphylaxis, and loss of consciousness (rare).
Contraindications
Contraindications to receiving amifostine include hypersensitivity to amifostine and aminothiol compounds like WR-1065. Ethyol contains mannitol.
References
- Kouvaris JR, Kouloulias VE, Vlahos LJ (June 2007). "Amifostine: the first selective-target and broad-spectrum radioprotector". Oncologist. 12 (6): 738–47. doi:10.1634/theoncologist.12-6-738. PMID 17602063.
- "Amifostine : BC Cancer Agency". British Columbia Cancer Agency. 2006-03-01. Archived from the original on 2015-03-14. Retrieved 2011-01-01.
- Koukourakis, Michael I.; Giatromanolaki, Alexandra; Zois, Christos E.; Kalamida, Dimitra; Pouliliou, Stamatia; Karagounis, Ilias V.; Yeh, Tzu-Lan; Abboud, Martine I.; Claridge, Timothy D. W. (2016-08-10). "Normal tissue radioprotection by amifostine via Warburg-type effects". Scientific Reports. 6: 30986. Bibcode:2016NatSR...630986K. doi:10.1038/srep30986. ISSN 2045-2322. PMC 4978965. PMID 27507219.