Transition metal
In chemistry, the term transition metal (or transition element) has three possible definitions:
- The IUPAC definition[1] defines a transition metal as "an element whose atom has a partially filled d sub-shell, or which can give rise to cations with an incomplete d sub-shell".
- Many scientists describe a "transition metal" as any element in the d-block of the periodic table, which includes groups 3 to 12 on the periodic table.[2][3] In actual practice, the f-block lanthanide and actinide series are also considered transition metals and are called "inner transition metals".
- Cotton and Wilkinson[4] expand the brief IUPAC definition (see above) by specifying which elements are included. As well as the elements of groups 4 to 11, they add scandium and yttrium in group 3, which have a partially filled d sub-shell in the metallic state. Lanthanum and actinium, which they consider group 3 elements, are however classified as lanthanides and actinides respectively.
| Part of a series on the |
| Periodic table |
|---|
|
Sets of elements |
|
English chemist Charles Rugeley Bury (1890–1968) first used the word transition in this context in 1921, when he referred to a transition series of elements during the change of an inner layer of electrons (for example n = 3 in the 4th row of the periodic table) from a stable group of 8 to one of 18, or from 18 to 32.[5][6][7] These elements are now known as the d-block.

Classification
In the d-block, the atoms of the elements have between zero and ten d electrons.
| Group | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
|---|---|---|---|---|---|---|---|---|---|---|
| Period 4 | 21Sc | 22Ti | 23V | 24Cr | 25Mn | 26Fe | 27Co | 28Ni | 29Cu | 30Zn |
| 5 | 39Y | 40Zr | 41Nb | 42Mo | 43Tc | 44Ru | 45Rh | 46Pd | 47Ag | 48Cd |
| 6 | 71Lu | 72Hf | 73Ta | 74W | 75Re | 76Os | 77Ir | 78Pt | 79Au | 80Hg |
| 7 | 103Lr | 104Rf | 105Db | 106Sg | 107Bh | 108Hs | 109Mt | 110Ds | 111Rg | 112Cn |
The elements of groups 4–11 are generally recognized as transition metals, justified by their typical chemistry, i.e. a large range of complex ions in various oxidation states, coloured complexes, and catalytic properties either as the element or as ions (or both). Sc and Y in group 3 are also generally recognized as transition metals. However, the elements La–Lu and Ac–Lr and group 12 attract different definitions from different authors.
- Many chemistry textbooks and printed periodic tables classify La and Ac as group 3 elements and transition metals, since their atomic ground-state configurations are s2d1 like Sc and Y. The elements Ce–Lu are considered as the "lanthanide" series (or "lanthanoid" according to IUPAC) and Th–Lr as the "actinide" series.[8][9] The two series together are classified as f-block elements, or (in older sources) as "inner transition elements". However, this results in a split of the d-block into two quite uneven portions.[10]
- Some inorganic chemistry textbooks include La with the lanthanides and Ac with the actinides.[4][11][12] This classification is based on similarities in chemical behaviour (though this similarity mostly only exists among the lanthanides) and defines 15 elements in each of the two series, even though they correspond to the filling of an f sub-shell, which can only contain 14 electrons.[13]
- A third classification defines the f-block elements as La–Yb and Ac–No, while placing Lu and Lr in group 3.[14] This is based on the Aufbau principle (or Madelung rule) for filling electron sub-shells, in which 4f is filled before 5d (and 5f before 6d), so that the f sub-shell is actually full at Yb (and No), while Lu has an [ ]s2f14d1 configuration. (Lr is an exception where the d-electron is replaced by a p-electron, but the energy difference is small enough that in a chemical environment it often displays d-occupancy anyway.) La and Ac are, in this view, simply considered exceptions to the Aufbau principle with electron configuration [ ]s2d1 (not [ ]s2f1 as the Aufbau principle predicts).[15] Excited states for the free atom and ion can become the ground state in chemical environments, which justifies this interpretation; La and Ac have vacant low-lying f sub-shells which are filled in Lu and Lr, so excitation to f orbitals is possible in La and Ac but not in Lu or Lr. This justifies the idea that La and Ac simply have irregular configurations (similar to Th as s2d2), and that they are the real beginning of the f-block.[16]
As the third form is the only form that allows simultaneous (1) preservation of the sequence of increasing atomic numbers, (2) a 14-element-wide f-block, and (3) avoidance of the split in the d-block, it has been suggested by a 2021 IUPAC preliminary report as the preferred form.[13] Such a modification, treating Lu as a transition element rather than as an inner transition element, was first suggested by Soviet physicists Lev Landau and Evgeny Lifshitz in 1948.[17] Following this, it was then suggested by many other physicists and chemists, and was generally the classification adopted by those who considered the issue,[18] but textbooks generally lagged in adopting it.[19]
Zinc, cadmium, and mercury are sometimes excluded from the transition metals,[5] as they have the electronic configuration [ ]d10s2, with no incomplete d shell.[20] In the oxidation state +2, the ions have the electronic configuration [ ]…d10. Although these elements can exist in other oxidation states, including the +1 oxidation state, as in the diatomic ion Hg2+
2, they still have a complete d shell in these oxidation states. The group 12 elements Zn, Cd and Hg may therefore, under certain criteria, be classed as post-transition metals in this case. However, it is often convenient to include these elements in a discussion of the transition elements. For example, when discussing the crystal field stabilization energy of first-row transition elements, it is convenient to also include the elements calcium and zinc, as both Ca2+
and Zn2+
have a value of zero, against which the value for other transition metal ions may be compared. Another example occurs in the Irving–Williams series of stability constants of complexes.
The recent (though disputed and so far not reproduced independently) synthesis of mercury(IV) fluoride (HgF
4) has been taken by some to reinforce the view that the group 12 elements should be considered transition metals,[21] but some authors still consider this compound to be exceptional.[22] Copernicium is expected to be able to use its d-electrons for chemistry as its 6d sub-shell is destabilised by strong relativistic effects due to its very high atomic number, and as such is expected to have transition-metal-like behaviour when it shows higher oxidation states than +2 (which are not definitely known for the lighter group 12 elements).
Although meitnerium, darmstadtium, and roentgenium are within the d-block and are expected to behave as transition metals analogous to their lighter congeners iridium, platinum, and gold, this has not yet been experimentally confirmed. Whether copernicium behaves more like mercury or has properties more similar to those of the noble gas radon is not clear.
Subclasses
Early transition metals are on the left side of the periodic table from group 3 to group 7. Late transition metals are on the right side of the d-block, from group 8 to 11 (and 12 if it is counted as transition metals).
Electronic configuration
The general electronic configuration of the d-block elements is (noble gas) (n − 1)d1–10ns0–2. Here "(noble gas)" is the configuration of the last noble gas preceding the atom in question, and n is the highest principal quantum number of an occupied orbital in that atom. For example Ti(Z = 22) is in period 4 so that n = 4, the first 18 electrons have the same configuration of Ar at the end of period 3, and the overall configuration is (Ar)3d24s2. The period 6 and 7 transition metals also add core (n − 2)f14 electrons, which are omitted from the tables below. The single exception is lawrencium, which has a 7p occupancy due to relativistic effects that become important at such high Z (though the p-orbitals can also contribute to chemical bonding in lighter transition elements).
The Madelung rule predicts that the inner d orbital is filled after the valence-shell s orbital. The typical electronic structure of transition metal atoms is then written as (noble gas) ns2(n − 1)dm. This rule is however only approximate – it only holds for some of the transition elements, and only then in the neutral ground states.
The d sub-shell is the next-to-last sub-shell and is denoted as sub-shell. The number of s electrons in the outermost s sub-shell is generally one or two except palladium (Pd), with no electron in that s sub shell in its ground state. The s sub-shell in the valence shell is represented as the ns sub-shell, e.g. 4s. In the periodic table, the transition metals are present in eight groups (4 to 11), with some authors including some elements in groups 3 or 12.
The elements in group 3 have an ns2(n − 1)d1 configuration, except for lawrencium (Lr): its 7s27p1 configuration exceptionally does not fill the 6d orbitals at all. The first transition series is present in the 4th period, and starts after Ca (Z = 20) of group-2 with the configuration [Ar]4s2, or scandium (Sc), the first element of group 3 with atomic number Z = 21 and configuration [Ar]4s23d1, depending on the definition used. As we move from left to right, electrons are added to the same d sub-shell till it is complete. The element of group 11 in the first transition series is copper (Cu) with an atypical configuration [Ar]4s13d10. Despite the filled d sub-shell in metallic copper it nevertheless forms a stable ion with an incomplete d sub-shell. Since the electrons added fill the orbitals, the properties of the d-block elements are quite different from those of s and p block elements in which the filling occurs either in s or in p-orbitals of the valence shell. The electronic configuration of the individual elements present in all the d-block series are given below:[23]
| Group | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
|---|---|---|---|---|---|---|---|---|---|---|
| Atomic number | 21 | 22 | 23 | 24 | 25 | 26 | 27 | 28 | 29 | 30 |
| Element | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn |
| Electron configuration |
3d14s2 | 3d24s2 | 3d34s2 | 3d54s1 | 3d54s2 | 3d64s2 | 3d74s2 | 3d84s2 | 3d104s1 | 3d104s2 |
| Atomic number | 39 | 40 | 41 | 42 | 43 | 44 | 45 | 46 | 47 | 48 |
|---|---|---|---|---|---|---|---|---|---|---|
| Element | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd |
| Electron configuration |
4d15s2 | 4d25s2 | 4d45s1 | 4d55s1 | 4d55s2 | 4d75s1 | 4d85s1 | 4d105s0 | 4d105s1 | 4d105s2 |
| Atomic number | 71 | 72 | 73 | 74 | 75 | 76 | 77 | 78 | 79 | 80 |
|---|---|---|---|---|---|---|---|---|---|---|
| Element | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg |
| Electron configuration |
5d16s2 | 5d26s2 | 5d36s2 | 5d46s2 | 5d56s2 | 5d66s2 | 5d76s2 | 5d96s1 | 5d106s1 | 5d106s2 |
| Atomic number | 103 | 104 | 105 | 106 | 107 | 108 | 109 | 110 | 111 | 112 |
|---|---|---|---|---|---|---|---|---|---|---|
| Element | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn |
| Electron configuration |
7s27p1 | 6d27s2 | 6d37s2 | 6d47s2 | 6d57s2 | 6d67s2 | 6d77s2 | 6d87s2 | 6d97s2 | 6d107s2 |
A careful look at the electronic configuration of the elements reveals that there are certain exceptions to the Madelung rule. For Cr as an example the rule predicts the configuration 3d44s2, but the observed atomic spectra show that the real ground state is 3d54s1. To explain such exceptions, it is necessary to consider the effects of increasing nuclear charge on the orbital energies, as well as the electron-electron interactions including both Coulomb repulsion and exchange energy.[23]
The orbitals that are involved in the transition metals are very significant because they influence such properties as magnetic character, variable oxidation states, formation of coloured compounds etc. The valence and orbitals have very little contribution in this regard since they hardly change in the moving from left to the right in a transition series. In transition metals, there is a greater horizontal similarities in the properties of the elements in a period in comparison to the periods in which the d-orbitals are not involved. This is because in a transition series, the valence shell electronic configuration of the elements do not change. However, there are some group similarities as well.
Characteristic properties
There are a number of properties shared by the transition elements that are not found in other elements, which results from the partially filled d shell. These include
- the formation of compounds whose colour is due to d–d electronic transitions
- the formation of compounds in many oxidation states, due to the relatively low energy gap between different possible oxidation states[24]
- the formation of many paramagnetic compounds due to the presence of unpaired d electrons. A few compounds of main-group elements are also paramagnetic (e.g. nitric oxide, oxygen)
Most transition metals can be bound to a variety of ligands, allowing for a wide variety of transition metal complexes.[25]
Coloured compounds

3)
2 (red); K
2Cr
2O
7 (orange); K
2CrO
4 (yellow); NiCl
2 (turquoise); CuSO
4 (blue); KMnO
4 (purple).
Colour in transition-series metal compounds is generally due to electronic transitions of two principal types.
- charge transfer transitions. An electron may jump from a predominantly ligand orbital to a predominantly metal orbital, giving rise to a ligand-to-metal charge-transfer (LMCT) transition. These can most easily occur when the metal is in a high oxidation state. For example, the colour of chromate, dichromate and permanganate ions is due to LMCT transitions. Another example is that mercuric iodide, HgI2, is red because of a LMCT transition.
A metal-to-ligand charge transfer (MLCT) transition will be most likely when the metal is in a low oxidation state and the ligand is easily reduced.
In general charge transfer transitions result in more intense colours than d-d transitions.
- d-d transitions. An electron jumps from one d-orbital to another. In complexes of the transition metals the d orbitals do not all have the same energy. The pattern of splitting of the d orbitals can be calculated using crystal field theory. The extent of the splitting depends on the particular metal, its oxidation state and the nature of the ligands. The actual energy levels are shown on Tanabe–Sugano diagrams.
In centrosymmetric complexes, such as octahedral complexes, d-d transitions are forbidden by the Laporte rule and only occur because of vibronic coupling in which a molecular vibration occurs together with a d-d transition. Tetrahedral complexes have somewhat more intense colour because mixing d and p orbitals is possible when there is no centre of symmetry, so transitions are not pure d-d transitions. The molar absorptivity (ε) of bands caused by d-d transitions are relatively low, roughly in the range 5-500 M−1cm−1 (where M = mol dm−3).[26] Some d-d transitions are spin forbidden. An example occurs in octahedral, high-spin complexes of manganese(II),
which has a d5 configuration in which all five electron has parallel spins; the colour of such complexes is much weaker than in complexes with spin-allowed transitions. Many compounds of manganese(II) appear almost colourless. The spectrum of [Mn(H
2O)
6]2+
shows a maximum molar absorptivity of about 0.04 M−1cm−1 in the visible spectrum.
Oxidation states
A characteristic of transition metals is that they exhibit two or more oxidation states, usually differing by one. For example, compounds of vanadium are known in all oxidation states between −1, such as [V(CO)
6]−
, and +5, such as VO3−
4.
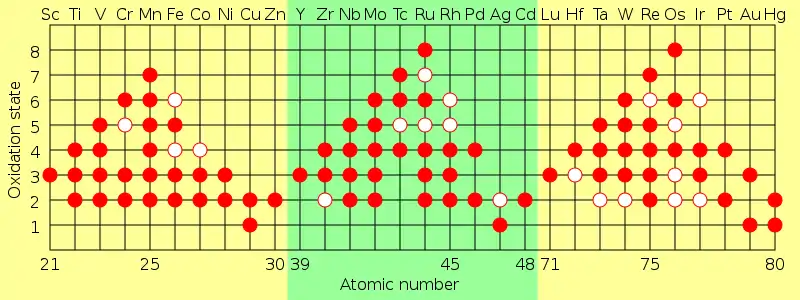
Main-group elements in groups 13 to 18 also exhibit multiple oxidation states. The "common" oxidation states of these elements typically differ by two instead of one. For example, compounds of gallium in oxidation states +1 and +3 exist in which there is a single gallium atom. Compounds of Ga(II) would have an unpaired electron and would behave as a free radical and generally be destroyed rapidly, but some stable radicals of Ga(II) are known.[27] Gallium also has a formal oxidation state of +2 in dimeric compounds, such as [Ga
2Cl
6]2−
, which contain a Ga-Ga bond formed from the unpaired electron on each Ga atom.[28] Thus the main difference in oxidation states, between transition elements and other elements is that oxidation states are known in which there is a single atom of the element and one or more unpaired electrons.
The maximum oxidation state in the first row transition metals is equal to the number of valence electrons from titanium (+4) up to manganese (+7), but decreases in the later elements. In the second row, the maximum occurs with ruthenium (+8), and in the third row, the maximum occurs with iridium (+9). In compounds such as [MnO
4]−
and OsO
4, the elements achieve a stable configuration by covalent bonding.
The lowest oxidation states are exhibited in metal carbonyl complexes such as Cr(CO)
6 (oxidation state zero) and [Fe(CO)
4]2−
(oxidation state −2) in which the 18-electron rule is obeyed. These complexes are also covalent.
Ionic compounds are mostly formed with oxidation states +2 and +3. In aqueous solution, the ions are hydrated by (usually) six water molecules arranged octahedrally.
Magnetism
Transition metal compounds are paramagnetic when they have one or more unpaired d electrons.[29] In octahedral complexes with between four and seven d electrons both high spin and low spin states are possible. Tetrahedral transition metal complexes such as [FeCl
4]2−
are high spin because the crystal field splitting is small so that the energy to be gained by virtue of the electrons being in lower energy orbitals is always less than the energy needed to pair up the spins. Some compounds are diamagnetic. These include octahedral, low-spin, d6 and square-planar d8 complexes. In these cases, crystal field splitting is such that all the electrons are paired up.
Ferromagnetism occurs when individual atoms are paramagnetic and the spin vectors are aligned parallel to each other in a crystalline material. Metallic iron and the alloy alnico are examples of ferromagnetic materials involving transition metals. Anti-ferromagnetism is another example of a magnetic property arising from a particular alignment of individual spins in the solid state.
Catalytic properties
The transition metals and their compounds are known for their homogeneous and heterogeneous catalytic activity. This activity is ascribed to their ability to adopt multiple oxidation states and to form complexes. Vanadium(V) oxide (in the contact process), finely divided iron (in the Haber process), and nickel (in catalytic hydrogenation) are some of the examples. Catalysts at a solid surface (nanomaterial-based catalysts) involve the formation of bonds between reactant molecules and atoms of the surface of the catalyst (first row transition metals utilize 3d and 4s electrons for bonding). This has the effect of increasing the concentration of the reactants at the catalyst surface and also weakening of the bonds in the reacting molecules (the activation energy is lowered). Also because the transition metal ions can change their oxidation states, they become more effective as catalysts.
An interesting type of catalysis occurs when the products of a reaction catalyse the reaction producing more catalyst (autocatalysis). One example is the reaction of oxalic acid with acidified potassium permanganate (or manganate (VII)).[30] Once a little Mn2+ has been produced, it can react with MnO4− forming Mn3+. This then reacts with C2O4− ions forming Mn2+ again.
Physical properties
As implied by the name, all transition metals are metals and thus conductors of electricity.
In general, transition metals possess a high density and high melting points and boiling points. These properties are due to metallic bonding by delocalized d electrons, leading to cohesion which increases with the number of shared electrons. However the group 12 metals have much lower melting and boiling points since their full d sub-shells prevent d–d bonding, which again tends to differentiate them from the accepted transition metals. Mercury has a melting point of −38.83 °C (−37.89 °F) and is a liquid at room temperature.
See also
- Inner transition element, a name given to any member of the f-block
- Main-group element, an element other than a transition metal
- Ligand field theory a development of crystal field theory taking covalency into account
- Crystal field theory a model that describes the breaking of degeneracies of electronic orbital states
- Post-transition metal, a metallic element to the right of the transition metals in the periodic table
References
- IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "transition element". doi:10.1351/goldbook.T06456
- Petrucci, Ralph H.; Harwood, William S.; Herring, F. Geoffrey (2002). General chemistry: principles and modern applications (8th ed.). Upper Saddle River, N.J: Prentice Hall. pp. 341–342. ISBN 978-0-13-014329-7. LCCN 2001032331. OCLC 46872308.
- Housecroft, C. E. and Sharpe, A. G. (2005) Inorganic Chemistry, 2nd ed, Pearson Prentice-Hall, pp. 20–21.
- Cotton, F. A. and Wilkinson, G. (1988) Inorganic Chemistry, 5th ed., Wiley, pp. 625–627. ISBN 978-0-471-84997-1.
- Jensen, William B. (2003). "The Place of Zinc, Cadmium, and Mercury in the Periodic Table" (PDF). Journal of Chemical Education. 80 (8): 952–961. Bibcode:2003JChEd..80..952J. doi:10.1021/ed080p952.
- Bury, C. R. (1921). "Langmuir's theory of the arrangement of electrons in atoms and molecules". J. Am. Chem. Soc. 43 (7): 1602–1609. doi:10.1021/ja01440a023.
- Bury, Charles Rugeley. Encyclopedia.com Complete dictionary of scientific biography (2008).
- Petrucci, Harwood & Herring 2002, pp. 49–50, 951.
- Miessler, G. L. and Tarr, D. A. (1999) Inorganic Chemistry, 2nd edn, Prentice-Hall, p. 16. ISBN 978-0-13-841891-5.
- Scerri, Eric (2009). "Which Elements Belong in Group 3?". Journal of Chemical Education. 86 (10): 1188. Bibcode:2009JChEd..86.1188S. doi:10.1021/ed086p1188.
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- Housecroft, C. E. and Sharpe, A. G. (2005) Inorganic Chemistry, 2nd ed., Pearson Prentice-Hall, p. 741.
- Scerri, Eric (18 January 2021). "Provisional Report on Discussions on Group 3 of the Periodic Table". Chemistry International. 43 (1): 31–34. doi:10.1515/ci-2021-0115. S2CID 231694898.
- William B. Jensen (1982). "The Positions of Lanthanum (Actinium) and Lutetium (Lawrencium) in the Periodic Table". J. Chem. Educ. 59 (8): 634–636. Bibcode:1982JChEd..59..634J. doi:10.1021/ed059p634.
- Scerri, E. R. (2011) A Very Short Introduction to the Periodic Table, Oxford University Press.
- Jensen, W. B. (2015). "Some Comments on the Position of Lawrencium in the Periodic Table" (PDF). Archived from the original (PDF) on 23 December 2015. Retrieved 20 September 2015.
- L. D. Landau, E. M. Lifshitz (1958). Quantum Mechanics: Non-Relativistic Theory. Vol. 3 (1st ed.). Pergamon Press. pp. 256–7.
- Jensen, William B. (2015). "The positions of lanthanum (actinium) and lutetium (lawrencium) in the periodic table: an update". Foundations of Chemistry. 17: 23–31. doi:10.1007/s10698-015-9216-1. S2CID 98624395. Retrieved 28 January 2021.
- "The constitution of group 3 of the periodic table". IUPAC. 2015. Archived from the original on 5 July 2016. Retrieved 30 July 2016.
- Cotton, F. Albert; Wilkinson, G.; Murillo, C. A. (1999). Advanced Inorganic Chemistry (6th ed.). New York: Wiley, ISBN 978-0-471-19957-1.
- Wang, Xuefang; Andrews, Lester; Riedel, Sebastian; Kaupp, Martin (2007). "Mercury Is a Transition Metal: The First Experimental Evidence for HgF4". Angew. Chem. Int. Ed. 46 (44): 8371–8375. doi:10.1002/anie.200703710. PMID 17899620.
- Jensen, William B. (2008). "Is Mercury Now a Transition Element?". J. Chem. Educ. 85 (9): 1182–1183. Bibcode:2008JChEd..85.1182J. doi:10.1021/ed085p1182.
- Miessler, G. L. and Tarr, D. A. (1999) Inorganic Chemistry, 2nd edn, Prentice-Hall, p. 38-39 ISBN 978-0-13-841891-5
- Matsumoto, Paul S (2005). "Trends in Ionization Energy of Transition-Metal Elements". Journal of Chemical Education. 82 (11): 1660. Bibcode:2005JChEd..82.1660M. doi:10.1021/ed082p1660.
- Hogan, C. Michael (2010). "Heavy metal" in Encyclopedia of Earth. National Council for Science and the Environment. E. Monosson and C. Cleveland (eds.) Washington DC.
- Orgel, L.E. (1966). An Introduction to Transition-Metal Chemistry, Ligand field theory (2nd. ed.). London: Methuen.
- Protchenko, Andrey V.; Dange, Deepak; Harmer, Jeffrey R.; Tang, Christina Y.; Schwarz, Andrew D.; Kelly, Michael J.; Phillips, Nicholas; Tirfoin, Remi; Birjkumar, Krishna Hassomal; Jones, Cameron; Kaltsoyannis, Nikolas; Mountford, Philip; Aldridge, Simon (16 February 2014). "Stable GaX2, InX2 and TlX2 radicals". Nature Chemistry. 6 (4): 315–319. Bibcode:2014NatCh...6..315P. doi:10.1038/nchem.1870. PMID 24651198.
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8. p. 240
- Figgis, B.N.; Lewis, J. (1960). Lewis, J.; Wilkins, R.G. (eds.). The Magnetochemistry of Complex Compounds. Modern Coordination Chemistry. New York: Wiley Interscience. pp. 400–454.
- Kovacs KA, Grof P, Burai L, Riedel M (2004). "Revising the Mechanism of the Permanganate/Oxalate Reaction". J. Phys. Chem. A. 108 (50): 11026–11031. Bibcode:2004JPCA..10811026K. doi:10.1021/jp047061u.