NMI Notes

October 2017
http://cste.us6.list-manage.com/subscribe?u=a74794e707a0d58b86a809758&id=69f74f148f
Overall NMI Updates
Welcome to NMI Notes
NMI Notes provides monthly news updates about the National Notifiable Diseases Surveillance System (NNDSS) Modernization Initiative (NMI). It is a collaboration by the Centers for Disease Control and Prevention (CDC), Council of State and Territorial Epidemiologists (CSTE), and Association of Public Health Laboratories (APHL).
If a colleague forwarded this issue to you, we encourage you to subscribe at this link to ensure that you receive future issues of NMI Notes.
- Due to the recent hurricane response and recovery efforts affecting state and federal agencies and our partners, CDC has postponed the September NMI eSHARE topic to the October session on 10/17/17 at 3:00 PM ET.
- The October 17 eSHARE will feature information from CDC and tips from states who have completed the arboviral v1.3 case notification implementation and onboarding process.
- Access past eSHARE presentations and details about future events at the NMI eSHARE website at http://www.cdc.gov/nmi/eshare.html.
Message Validation, Processing, and Provisioning System (MVPS) Updates
- The MVPS team has begun testing MVPS Upgrade (MVPSu) functionality for generic v2 and hepatitis, which will be the first implementation of the MVPSu processing changes. These changes, expected in late October, will implement the new algorithm to define a unique case.
- The MVPS development team continues to make enhancements to MVPS Classic, including implementing an automated check script that runs each time data are loaded to the provisioning database. The script proactively identifies when a provisioning script is interrupted and does not automatically re-start and notifies the operations and maintenance team.
- The NMI team encourages all non-pilot jurisdictions to complete the Secure Access Management Services (SAMS) proofing process and confirm their access with the MVPS support team. More information about SAMS access and training may be found on the NMI website at http://www.cdc.gov/nmi/ta-sams.html.
HL7 Case Notification Messages – IN PRODUCTION

Arboviral v1.3
-
-
- States now in production for arboviral v1.3 are Delaware, Florida, Idaho, New York, Oregon, South Dakota, Tennessee, Texas, and Wisconsin.
- Arkansas is engaged in onboarding.
-
Generic v2
-
-
- CDC updated the generic v2 message mapping guide (MMG) and test scenarios with wording corrections and information about the new algorithm to define a unique case. These documents were posted on 9/5/17 to the NNDSS HL7 Case Notification Resource Center at https://wwwn.cdc.gov/nndss/case-notification/ and will be implemented in October 2017.
- CDC also updated the MMG Related Documentation page of the HL7 Case Notification Resource Center at https://wwwn.cdc.gov/nndss/case-notification/related-documentation.html with new versions of the following documents:
- “PHIN Messaging Specification for Case Notification v3.0” contains several updates related to field lengths.
- “Generic Data Elements that Define a Unique Case” includes a new algorithm for unique cases in MVPS (to be implemented in October 2017).
- “FAQs for MMG Implementation” also was updated with information related to the new algorithm for unique cases.
- States now in production for generic v2 are Florida, Kansas, Michigan, Minnesota, New York, South Carolina, Oregon, and Wisconsin.
- Alaska, California, Illinois, Mississippi, and New Hampshire are engaged in onboarding.
-
Hepatitis
-
- States now in production for hepatitis are Florida, Idaho, Michigan, Minnesota, and Oregon.
- California is engaged in onboarding.
HL7 Case Notification Messages – IN DEVELOPMENT
Babesiosis Message Mapping Guide (MMG) (Stage 1—Draft)
- CDC revised the babesiosis MMG external review comments in preparation for publication.
- The APHL NMI technical assistance team is reviewing the draft babesiosis MMG.
Congenital Syphilis (CS) and STD MMGs (Stage II—Pilot Test-ready)
- NMI technical assistance is working with pilot states to implement the STD and CS messages.
Foodborne and Diarrheal Disease (FDD) MMG (Stage I—Draft):
- CDC and CSTE have set a tentative date of 10/25/17 at 3:00 PM ET to host the FDD MMG webinar. More details will be provided as they are available.
Malaria MMG (Stage I—Draft)
- The CDC program is reviewing an updated draft malaria MMG.
- APHL also is reviewing the draft MMG.
Measles MMG (Stage I—Draft)
- No new updates at this time.
Mumps and Pertussis MMGs (Stage II—Pilot Test-ready)
- CDC has identified potential pilot sites for the mumps and pertussis MMGs and will contact them in the near future.
Rubella and Congenital Rubella Syndrome (CRS) MMGs (Stage I—Draft)
- CDC is making updates to the draft CRS MMG based on feedback from internal preclearance.
- CDC has submitted the draft rubella MMG for CDC internal preclearance.
Trichinellosis (Requirements Analysis)
- No new updates at this time.
Varicella MMG (Stage I—Draft)
- CDC updated the varicella test messages per APHL feedback.
- The CDC program is reviewing pilot test-ready versions of the MMG, test scenarios, test messages, and annotated surveillance worksheet for signoff.
NMI Resources

- NMI Technical Assistance and Training Resource Center: https://www.cdc.gov/nmi/ta-trc/index.html
- NNDSS HL7 Case Notification Resource Center: https://wwwn.cdc.gov/nndss/case-notification/
- NMI Notes Monthly News Update: https://www.cdc.gov/nmi/news.html
- NMI eSHARE Monthly Webinar: http://www.cdc.gov/nmi/eshare.html
- NMI Newsroom: https://www.cdc.gov/nmi/newsroom.html
- NMI Internet Site (includes NMI fact sheets and FAQs): http://www.cdc.gov/nmi/
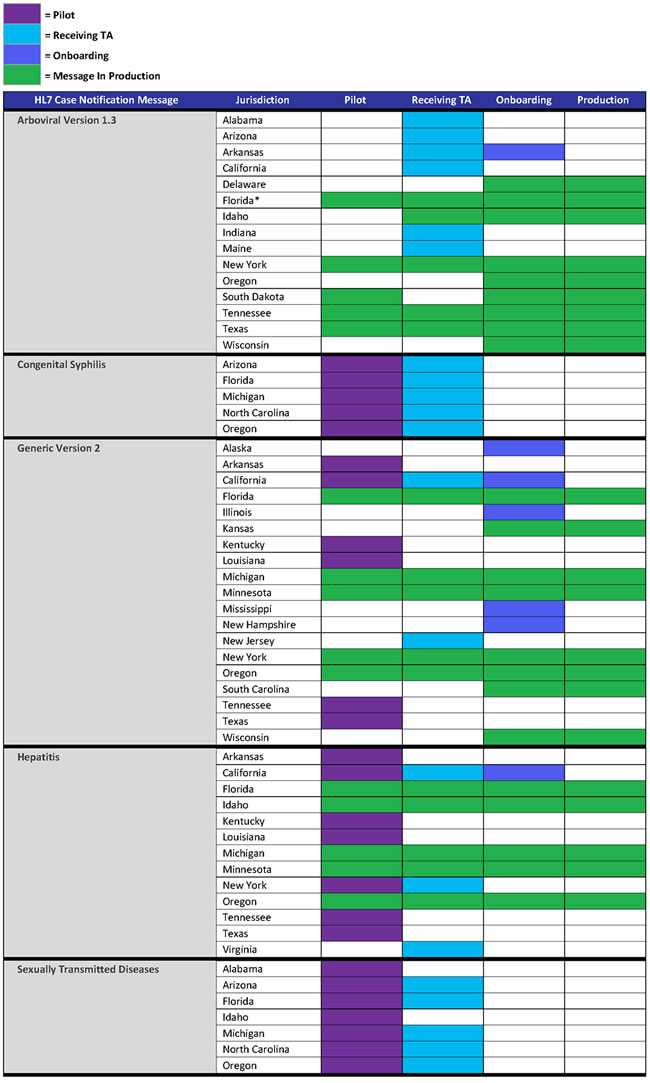
* Florida has implemented Arboviral 1.3 for Zika and is continuing to work on implementing the remaining arboviral conditions.
NMI Status Report: This chart shows the status of state public health departments who are piloting, receiving technical assistance, onboarding, or in production for finalized HL7 case notification messages developed as part of the NNDSS Modernization Initiative.
Upcoming Events
- NMI eSHARE; 10/17/17, 3:00PM ET; http://www.cdc.gov/nmi/eshare.html
Ready to request technical assistance or onboarding?
Please contact edx@cdc.gov for more information.
Have questions or feedback on NMI Notes?
Email edx@cdc.gov.
- Page last reviewed: September 29, 2017
- Page last updated: September 29, 2017
- Content source:



 ShareCompartir
ShareCompartir
