Mangafodipir
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Pregnancy category |
|
| Routes of administration | Intravenous infusion |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | NA |
| Protein binding | 27% (manganese) Negligible (DPDP) |
| Elimination half-life | 20 minutes (manganese) 50 minutes (DPDP) |
| Excretion | Renal and fecal (manganese) Renal (DPDP) |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C22H28MnN4O14P2 |
| Molar mass | 689.366 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
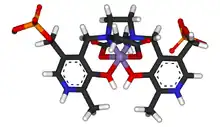
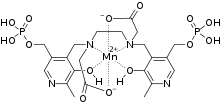
Mangafodipir (sold under the brand name Teslascan as mangafodipir trisodium) is a contrast agent delivered intravenously to enhance contrast in magnetic resonance imaging (MRI) of the liver, and has potential to serve as an adjunct for various chemotherapeutic agents and during coronary intervention. It has two parts, a paramagnetic manganese(II) ion and the fodipir (dipyridoxyl diphosphate, DPDP) chelating agent. When freed from the organic ligand, the manganese shortens the longitudinal relaxation time (T1) in an MRI scan. Normal liver tissue absorbs the manganese more than abnormal or cancerous tissue, which makes the normal tissue appear brighter in MRIs. This enhanced contrast allows lesions to be more easily identified.
Mangafodipir was withdrawn from the US market in 2003[1] and the European market in 2012 for commercial reasons by the manufacturer.[2]
Reactive oxygen species (ROS) and reactive nitrogen species (RNS) participate in pathological tissue damage. Mitochondrial manganese superoxide dismutase (MnSOD) normally keeps ROS and RNS in check. During development of mangafodipir as an MRI contrast agent, it was discovered that it possessed MnSOD mimetic activity. Mangafodipir has been tested as a chemotherapy adjunct in cancer patients and as an adjunct to percutaneous coronary intervention in patients with myocardial infarctions, with promising results.[3]
Mangafodipir has shown promising results in human brain imaging without detectable toxicity[4][5] and usefulness in detecting lesions in multiple sclerosis.[6]
Whereas MRI contrast depends on release of Mn2+, the MnSOD mimetic activity depends on Mn2+ that remains bound to DPDP. Calmangafodipir [Ca4Mn(DPDP)5] (brand name PledOx) is stabilized with respect to Mn2+ and has improved therapeutic activity.[3] Calmangafodipir is being explored as a chemotherapy adjunct in cancer patients.
References
- ↑ "January 2005: Additions and Deletions to the Drug Product List". U.S Food and Drug Administration.
- ↑ "Teslascan (mangafodipir): Withdrawal of the marketing authorisation in the European Union" (PDF). European Medicines Agency.
- 1 2 Karlsson JO, Ignarro LJ, Lundström I, Jynge P, Almén T (April 2015). "Calmangafodipir [Ca4Mn(DPDP)5], mangafodipir (MnDPDP) and MnPLED with special reference to their SOD mimetic and therapeutic properties". Drug Discovery Today. 20 (4): 411–21. doi:10.1016/j.drudis.2014.11.008. PMID 25463039.
- ↑ Sudarshana, D. M.; Nair, G.; Dwyer, J. T.; Dewey, B.; Steele, S. U.; Suto, D. J.; Wu, T.; Berkowitz, B. A.; Koretsky, A. P.; Cortese ICM; Reich, D. S. (2019). "Manganese-Enhanced MRI of the Brain in Healthy Volunteers". AJNR. American Journal of Neuroradiology. 40 (8): 1309–1316. doi:10.3174/ajnr.A6152. PMC 6754109. PMID 31371354.
- ↑ Clinical trial number NCT01326715 for "Manganese-Enhanced Magnetic Resonance Imaging in Healthy Volunteers and People With Multiple Sclerosis" at ClinicalTrials.gov.
- ↑ Suto, D. J.; Nair, G.; Sudarshana, D. M.; Steele, S. U.; Dwyer, J.; Beck, E. S.; Ohayon, J.; McFarland, H.; Koretsky, A. P.; Cortese ICM; Reich, D. S. (2020). "Manganese-Enhanced MRI in Patients with Multiple Sclerosis". AJNR. American Journal of Neuroradiology. 41 (9): 1569–1576. doi:10.3174/ajnr.A6665. PMC 7583091. PMID 32763897.