Relamorelin
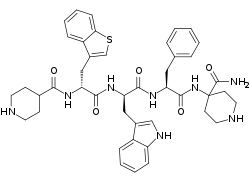
Relamorelin (INN, USAN) (developmental code names RM-131, BIM-28131, BIM-28163) is a synthetic peptide, centrally penetrant, selective agonist of the ghrelin/growth hormone secretagogue receptor (GHSR) which is under development by Allergan pharmaceuticals for the treatment of diabetic gastroparesis, chronic idiopathic constipation, and anorexia nervosa.[1][2][3] It is a pentapeptide and an analogue of ghrelin with improved potency and pharmacokinetics.[1][2] In humans, relamorelin produces increases in plasma growth hormone, prolactin, and cortisol levels,[2][4] and, like other GHSR agonists, increases appetite.[3] As of June 2015, relamorelin is in phase II clinical trials for diabetic gastroparesis and constipation.[5] The United States Food and Drug Administration (FDA) has granted Fast Track designation to relamorelin for diabetic gastroparesis.[6] The development of the drug is uncertain as the most recent mention of it was in 2019 SEC filing from the drug manufacturer lists the drug's expected launch year as 2024 but not in subsequent filings or press releases.[7][8]
 | |
| Clinical data | |
|---|---|
| Routes of administration | Subcutaneous |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C43H50N8O5S |
| Molar mass | 790.98 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
See also
References
- Haddley K (2014). "Relamorelin. Ghrelin receptor agonist, treatment of constipation, treatment of anorexia nervosa, treatment of diabetic gastroparesis". Drugs of the Future. 39 (11): 775. doi:10.1358/dof.2014.039.011.2231856. ISSN 0377-8282.
- Camilleri M, Acosta A (March 2015). "Emerging treatments in Neurogastroenterology: relamorelin: a novel gastrocolokinetic synthetic ghrelin agonist". Neurogastroenterology and Motility. 27 (3): 324–32. doi:10.1111/nmo.12490. PMC 4424792. PMID 25545036.
- Acosta A, Camilleri M, Kolar G, Iturrino J, Szarka LA, Boldingh A, et al. (December 2015). "Relamorelin Relieves Constipation and Accelerates Colonic Transit in a Phase 2, Placebo-Controlled, Randomized Trial". Clinical Gastroenterology and Hepatology. 13 (13): 2312–9.e1. doi:10.1016/j.cgh.2015.04.184. PMID 26001337.
- Shin A, Camilleri M, Busciglio I, Burton D, Stoner E, Noonan P, et al. (January 2013). "Randomized controlled phase Ib study of ghrelin agonist, RM-131, in type 2 diabetic women with delayed gastric emptying: pharmacokinetics and pharmacodynamics". Diabetes Care. 36 (1): 41–8. doi:10.2337/dc12-1128. PMC 3526234. PMID 22961573.
- Valentin N, Acosta A, Camilleri M (June 2015). "Early investigational therapeutics for gastrointestinal motility disorders: from animal studies to Phase II trials". Expert Opinion on Investigational Drugs. 24 (6): 769–79. doi:10.1517/13543784.2015.1025132. PMC 4578705. PMID 25971881.
- Rhythm Pharmaceuticals (2014). "Rhythm Presents Positive Phase 2 Study Results for Relamorelin for Chronic Constipation". PRNewsire. Retrieved June 10, 2015.
- "AbbVie Inc SEC filing 2019". AbbVie. November 7, 2019.
- "AbbVie Reports First-Quarter 2023 Financial Results".