Tellurium tetrachloride
Tellurium tetrachloride is the inorganic compound with the empirical formula TeCl4. The compound is volatile, subliming at 200 °C at 0.1 mmHg.[1] Molten TeCl4 is ionic, dissociating into TeCl3+ and Te2Cl102−.[1]
 | |
| Names | |
|---|---|
| IUPAC names
Tellurium(IV) chloride Tetratellurium hexadecachloride | |
| Other names
Tellurium chloride | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.030.038 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| TeCl4 | |
| Molar mass | 269.41 g/mol |
| Appearance | hygroscopic pale yellow solid (if fused, maroon liquid) |
| Density | 3.26 g/cm3, solid |
| Melting point | 224 °C (435 °F; 497 K) |
| Boiling point | 380 °C (716 °F; 653 K) |
| Structure | |
| Monoclinic, mS80 | |
| C12/c1, No. 15 | |
| Distorted octahedral (Te) | |
| Seesaw (gas phase) | |
| 2.59 D (gas phase) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Toxic, corrosive, respiratory irritant |
| Related compounds | |
Other anions |
Tellurium tetrafluoride Tellurium tetrabromide Tellurium tetraiodide |
Other cations |
Selenium tetrachloride Polonium tetrachloride |
Related compounds |
Tellurium dichloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
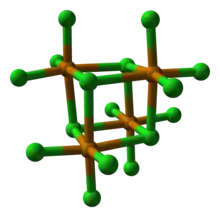
Structure
TeCl4 is monomeric in the gas phase, with a structure similar to that of SF4.[2] In the solid state, it is a tetrameric cubane-type cluster, consisting of a Te4Cl4 core and three terminal chloride ligands for each Te. Alternatively, this tetrameric structure can be considered as a Te4 tetrahedron with face-capping chlorines and three terminal chlorines per tellurium atom, giving each tellurium atom a distorted octahedral environment
Synthesis
TeCl4 is prepared by chlorination of tellurium powder:
- Te + 2 Cl2 → TeCl4
The reaction is initiated with heat. The product is isolated by distillation.[3]
Applications
TeCl4 is of occasional interest in organic synthesis.[4] It adds to alkenes to give Cl-C-C-TeCl3 derivatives, wherein the Te can be subsequently removed with sodium sulfide. Electron-rich arenes react to give aryl Te compounds. Thus, anisole gives TeCl2(C6H4OMe)2, which can be reduced to the diaryl telluride.
Safety considerations
As is the case for other tellurium compounds, TeCl4 is toxic. It also releases HCl upon hydrolysis.
References
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- Cotton, F. Albert; Wilkinson, Geoffrey; Murillo, Carlos A.; Bochmann, Manfred (1999), Advanced Inorganic Chemistry (6th ed.), New York: Wiley-Interscience, ISBN 0-471-19957-5
- Suttle, J. F.; Smith, C. R. F. (1950). Audrieth, Ludwig F. (ed.). Tellurium(IV) chloride. Inorganic Syntheses. Vol. 3. pp. 140–2. doi:10.1002/9780470132340. ISBN 978-0-470-13162-6.
{{cite book}}: CS1 maint: multiple names: authors list (link) - Petragnani, N.; Comasseto, J. V. (1991). "Tellurium Reagents in Organic Synthesis; Recent Advances. Part 1". Synthesis. 1991 (10): 793–817. doi:10.1055/s-1991-26577. and Petragnani, N.; Comasseto, J. V. (1991). "Tellurium Reagents in Organic Synthesis; Recent Advances. Part 2". Synthesis. 1991 (11): 897–919. doi:10.1055/s-1991-26605.