Vanadium(III) oxide
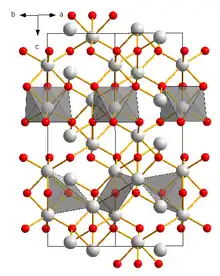
Vanadium(III) oxide is the inorganic compound with the formula V2O3. It is a black solid prepared by reduction of V2O5 with hydrogen or carbon monoxide.[3][4] It is a basic oxide dissolving in acids to give solutions of vanadium (III) complexes.[4] V2O3 has the corundum structure.[4] It is antiferromagnetic with a critical temperature of 160 K. [5] At this temperature there is an abrupt change in conductivity from metallic to insulating.[5] This also distorts the crystal structure to a monoclinic space group: C2/c.[1]
 | |
.jpg.webp) | |
| Names | |
|---|---|
| Other names
Vanadium sesquioxide, Vanadic oxide | |
| Identifiers | |
3D model (JSmol) |
|
| ECHA InfoCard | 100.013.847 |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| V2O3 | |
| Molar mass | 149.881 g/mol |
| Appearance | Black powder |
| Density | 4.87 g/cm3 |
| Melting point | 1,940 °C (3,520 °F; 2,210 K) |
| Solubility in other solvents | Insoluble |
| +1976.0·10−6 cm3/mol | |
| Structure[1] | |
| Corundum, hR30 | |
| R3c (No. 167) | |
a = 547 pm α = 53.74°, β = 90°, γ = 90° | |
| Thermochemistry | |
Std molar entropy (S⦵298) |
98.07 J/mol·K [2] |
Std enthalpy of formation (ΔfH⦵298) |
-1218.800 kJ/mol [2] |
Gibbs free energy (ΔfG⦵) |
-1139.052 kJ/mol [2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Upon exposure to air it gradually converts into indigo-blue V2O4.[5]
In nature it occurs as the rare mineral karelianite.[6]
References
- Shvets, Petr; Dikaya, Olga; Maksimova, Ksenia; Goikhman, Alexander (2019-05-15). "A review of Raman spectroscopy of vanadium oxides". Journal of Raman Spectroscopy. Wiley. 50 (8): 1226–1244. Bibcode:2019JRSp...50.1226S. doi:10.1002/jrs.5616. ISSN 0377-0486. S2CID 182370875.
- R. Robie, B. Hemingway, and J. Fisher, “Thermodynamic Properties of Minerals and Related Substances at 298.15K and 1bar Pressure and at Higher Temperatures,” US Geol. Surv., vol. 1452, 1978.
- Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 1. p. 1267.
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- E.M. Page, S.A.Wass (1994),Vanadium:Inorganic and Coordination chemistry, Encyclopedia of Inorganic Chemistry, John Wiley & Sons, ISBN 0-471-93620-0
- "Karelianite". www.mindat.org.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.