Leptospira
Leptospira (Ancient Greek: leptos, 'fine, thin' and Latin: spira, 'coil')[1] is a genus of spirochaete bacteria, including a small number of pathogenic and saprophytic species.[2] Leptospira was first observed in 1907 in kidney tissue slices of a leptospirosis victim who was described as having died of "yellow fever".[3]
| Leptospira | |
|---|---|
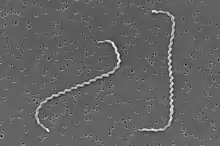 | |
| Scanning electron micrograph of Leptospira interrogans | |
| Scientific classification | |
| Domain: | Bacteria |
| Phylum: | Spirochaetota |
| Class: | Spirochaetia |
| Order: | Leptospirales |
| Family: | Leptospiraceae |
| Genus: | Leptospira Noguchi 1917 non Swainson 1840 non Boucot, Johnson & Staton 1964 |
| Type species | |
| Leptospira interrogans (Stimson 1907) Wenyon 1926 | |
| Species | |
See text | |
Taxonomy
Leptospira, together with the genera Leptonema and Turneria, is a member of the family Leptospiraceae. The genus Leptospira is divided into 20 species based on DNA hybridization studies.[4][5]
Pathogenic Leptospira
- Leptospira alstonii Smythe et al. 2013 ["Leptospira alstoni" Haake et al. 1993]
- Leptospira interrogans (Stimson 1907) Wenyon 1926 emend. Faine and Stallman 1982 ["Spirochaeta interrogans" Stimson 1907; "Spirochaeta nodosa" Hubener & Reiter 1916; "Spirochaeta icterohaemorrhagiae" Inada et al. 1916; "Spirochaeta icterogenes" Uhlenhuth & Fromme 1916; "Leptospira icteroides" Noguchi 1919]
- Leptospira kirschneri Ramadass et al. 1992
- Leptospira noguchii Yasuda et al. 1987
- Leptospira alexanderi Brenner et al. 1999
- Leptospira weilii Yasuda et al. 1987
- Leptospira borgpetersenii Yasuda et al. 1987
- Leptospira santarosai Yasuda et al. 1987
- Leptospira kmetyi Slack et al. 2009[6]
- Leptospira mayottensis Bourhy et al. 2014
Intermediates or opportunistic Leptospira
- Leptospira inadai Yasuda et al. 1987
- Leptospira fainei Perolat et al. 1998
- Leptospira broomii Levett et al. 2006[7]
- Leptospira licerasiae Matthias et al. 2009[8]
- Leptospira wolffii Slack et al. 2008[9]
Non-pathogenic Leptospira
- Leptospira biflexa (Wolbach and Binger 1914) Noguchi 1918 emend. Faine and Stallman 1982 ["Spirochaeta biflexa" Wolbach & Binger 1914]
- Leptospira idonii Saito et al. 2013
- Leptospira meyeri Yasuda et al. 1987
- Leptospira wolbachii Yasuda et al. 1987
- Leptospira vanthielii Smythe et al. 2013
- Leptospira terpstrae Smythe et al. 2013
- Leptospira yanagawae Smythe et al. 2013
Members of Leptospira are also grouped into serovars according to their antigenic relatedness. There are currently over 200 recognized serovars. A few serovars are found in more than one species of Leptospira.
At its 2002 meeting, the Committee on the Taxonomy of Leptospira of the International Union of Microbiological Societies approved the following nomenclature for serovars of Leptospira. Genus and species names are italicized as usual, with the serovar name not italicized and with an upper case first letter.
- Genus species serovar Serovar_name
For example:
- Leptospira interrogans serovar Australis
- Leptospira biflexa serovar Patoc
Phylogeny
The currently accepted taxonomy is based on the List of Prokaryotic names with Standing in Nomenclature (LPSN)[10] and National Center for Biotechnology Information (NCBI).[11]
| 16S rRNA based LTP_12_2021[12][13][14] | GTDB 07-RS207 by Genome Taxonomy Database[15][16][17] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
Morphology
Although over 200 serotypes of Leptospira have been described, all members of the genus have similar morphology. Leptospira are spiral-shaped bacteria that are 6-20 μm long and 0.1 μm in diameter with a wavelength of about 0.5 μm.[18] One or both ends of the spirochete are usually hooked. Because they are so thin, live Leptospira are best observed by darkfield microscopy.
The bacteria have a number of degrees of freedom; when ready to proliferate via binary fission, the bacterium noticeably bends in the place of the future split.
Cellular structure
Leptospira have a Gram-negative-like cell envelope consisting of a cytoplasmic and outer membrane. However, the peptidoglycan layer is associated with the cytoplasmic rather than the outer membrane, an arrangement that is unique to spirochetes. The two flagella of Leptospira extend from the cytoplasmic membrane at the ends of the bacterium into the periplasmic space and are necessary for the motility of Leptospira.[19]
The outer membrane contains a variety of lipoproteins and transmembrane outer membrane proteins.[20] As expected, the protein composition of the outer membrane differs when comparing Leptospira growing in artificial medium with Leptospira present in an infected animal.[21][22][23] Several leptospiral outer membrane proteins have been shown to attach to the host extracellular matrix and to factor H. These proteins may be important for adhesion of Leptospira to host tissues and in resisting complement, respectively.[24][25][26]
The outer membrane of Leptospira, like those of most other Gram-negative bacteria, contains lipopolysaccharide (LPS). Differences in the highly immunogenic LPS structure account for the numerous serovars of Leptospira.[18] Consequently, immunity is serovar specific; current leptospiral vaccines, which consist of one or several serovars of Leptospira endemic in the population to be immunized, protect only against the serovars contained in the vaccine preparation. Leptospiral LPS has low endotoxin activity.[18] An unusual feature of leptospiral LPS is that it activates host cells via TLR2 rather than TLR4.[27] The unique structure of the lipid A portion of the LPS molecule may account for this observation.[28] Finally, the LPS O antigen content of L. interrogans differs in an acutely infected versus a chronically infected animal.[29] The role of O antigen changes in the establishment or maintenance of acute or chronic infection, if any, is unknown.
Habitat
Leptospira, both pathogenic and saprophytic, can occupy diverse environments, habitats, and life cycles; these bacteria are found throughout the world, except in Antarctica. High humidity and neutral (6.9–7.4) pH are necessary for their survival in the environment, with stagnant water reservoirs—bogs, shallow lakes, ponds, puddles, etc.—being the natural habitat for the bacteria.
Nutrition
Leptospira are cultivated at 30 °C in Ellinghausen-McCullough-Johnson-Harris (EMJH) medium, which can be supplemented with 0.21% rabbit serum to enhance growth of fastidious strains.[30] Growth of pathogenic Leptospira in an artificial nutrient environment such as EMJH becomes noticeable in 4–7 days; growth of saprophytic strains occur within 2–3 days. The minimal growth temperature of pathogenic species is 13–15 °C. Because the minimal growth temperature of the saprophytes is 5–10 °C, the ability of Leptospira to grow at 13 °C can be used to distinguish saprophytic from pathogenic Leptospira species.[30] The optimal pH for growth of Leptospira is 7.2–7.6.
Leptospira are aerobes whose major carbon and energy source during in vitro growth is long-chain fatty acids, which are metabolized by beta-oxidation.[31][32] Fatty acids are provided in EMJH in the form of Tween.[30] Fatty acid molecules are bound by albumin in EMJH and are released slowly into the medium to prevent its toxic accumulation.
Like most bacteria, Leptospira require iron for growth.[33] L. interrogans and L. biflexa have the ability to acquire iron in different forms.[34] A TonB-dependent receptor required for utilization of the ferrous form of the iron has been identified in L. biflexa, and an ortholog of the receptor is encoded in the genome of L. interrogans. L. interrogans can also obtain iron from heme, which is bound to most of the iron in the human body. The HbpA hemin-binding protein, which may be involved in the uptake of hemin, has been identified on the surface of L. interrogans[35] Although other pathogenic species of Leptospira and L. biflexa lack HbpA, yet another hemin-binding protein, LipL41, may account for their ability to use hemin as a source of iron.[35] Although they do not secrete siderophores, L. biflexa and L. interrogans may be capable of obtaining iron from siderophores secreted by other microorganisms.[34]
Genome
The genome of pathogenic Leptospira consists of two chromosomes. The size of the genomes of L. interrogans serovars Copenhageni and Lai is approximately 4.6 Mb.[36][37] However, the genome of L. borgpetersenii serovar Hardjo is only 3.9 Mb in size with a large number of pseudogenes, gene fragments, and insertion sequences relative to the genomes of L. interrogans.[38] L. interrogans and L. borgpetersenii share 2708 genes from which 656 are pathogenic specific genes. The guanine plus cytosine (GC) content is between 35% and 41%.[39] L. borgpetersenii serovar Hardjo is usually transmitted by direct exposure to infected tissues, whereas L. interrogans is often acquired from water or soil contaminated by the urine of carrier animals harboring Leptospira in their kidneys. The high number of defective genes and insertion sequences in L. borgpetersenii Hardjo together with the poor survival outside of the host and difference in transmission patterns compared to L. interrogans suggest that L. borgpetersenii is undergoing insertion-sequence mediated genomic decay, with ongoing loss of genes necessary for survival outside of the host animal.[38]
Genotyping
Genome sequence determination several strains of Leptospira lead to the development of multilocus VNTR (Variable Number of Tandem Repeats) typing and multilocus sequence typing (MLST) for species level identification of pathogenic Leptospira species.[40] Both methods hold the potential to replace the highly ambiguous serotyping method currently in vogue for leptospiral strain identification.[40]
References
- "leptospirosis". American Heritage Dictionary of the English Language: Fourth Edition. Bartleby.com. 2000. Archived from the original on 2007-11-15. Retrieved 2007-05-13.
- Ryan KJ; Ray CG, eds. (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. ISBN 978-0-8385-8529-0.
- Stimson AM (1907). "Note on an organism found in yellow-fever tissue". Public Health Reports. 22 (18): 541. doi:10.2307/4559008. JSTOR 4559008.
- Brenner DJ, Kaufmann AF, Sulzer KR, Steigerwalt AG, Rogers FC, Weyant RS (1999). "Further determination of DNA relatedness between serogroups and serovars in the family Leptospiraceae with a proposal for Leptospira alexanderi sp. nov. and four new Leptospira genomospecies". Int. J. Syst. Bacteriol. 49 (2): 839–58. doi:10.1099/00207713-49-2-839. PMID 10319510.
- Bharti AR, Nally JE, Ricaldi JN, Matthias MA, Diaz MM, Lovett MA, Levett PN, Gilman RH, Willig MR, Gotuzzo E, Vinetz JM (2003). "Leptospirosis: a zoonotic disease of global importance". The Lancet Infectious Diseases. 3 (12): 757–71. doi:10.1016/S1473-3099(03)00830-2. PMID 14652202.
- Slack AT, Khairani-Bejo S, Symonds ML, et al. (April 2009). "Leptospira kmetyi sp. nov., isolated from an environmental source in Malaysia". Int. J. Syst. Evol. Microbiol. 59 (Pt 4): 705–8. doi:10.1099/ijs.0.002766-0. PMID 19329592.
- Levett PN, Morey RE, Galloway RL, Steigerwalt AG (2006). "Leptospira broomii sp. nov., isolated from humans with leptospirosis". Int. J. Syst. Evol. Microbiol. 56 (Pt 3): 671–3. doi:10.1099/ijs.0.63783-0. PMID 16514048.
- Matthias MA, Ricaldi JN, Cespedes M, Diaz MM, Galloway RL, Saito M, Steigerwalt AG, Patra KP, Ore CV, Gotuzzo E, Gilman RH, Levett PN, Vinetz JM (2008). Picardeau M (ed.). "Human Leptospirosis Caused by a New, Antigenically Unique Leptospira Associated with a Rattus Species Reservoir in the Peruvian Amazon". PLOS Negl Trop Dis. 2 (4): e213. doi:10.1371/journal.pntd.0000213. PMC 2271056. PMID 18382606.
- Slack AT, Kalambaheti T, Symonds ML, Dohnt MF, Galloway RL, Steigerwalt AG, Chaicumpa W, Bunyaraksyotin G, Craig S, Harrower BJ, Smythe LD (October 2008). "Leptospira wolffii sp. nov., isolated from a human with suspected leptospirosis in Thailand". International Journal of Systematic and Evolutionary Microbiology. 58 (Pt 10): 2305–8. doi:10.1099/ijs.0.64947-0. PMID 18842846.
- J.P. Euzéby. "Leptospira". List of Prokaryotic names with Standing in Nomenclature (LPSN). Retrieved 2021-03-20.
- Sayers; et al. "Leptospira". National Center for Biotechnology Information (NCBI) taxonomy database. Retrieved 2021-03-20.
- "The LTP". Retrieved 23 February 2021.
- "LTP_all tree in newick format". Retrieved 23 February 2021.
- "LTP_12_2021 Release Notes" (PDF). Retrieved 23 February 2021.
- "GTDB release 07-RS207". Genome Taxonomy Database. Retrieved 20 June 2022.
- "ar53_r207.sp_label". Genome Taxonomy Database. Retrieved 20 June 2022.
- "Taxon History". Genome Taxonomy Database. Retrieved 20 June 2022.
- Levett PN (2001). "Leptospirosis". Clin. Microbiol. Rev. 14 (2): 296–326. doi:10.1128/CMR.14.2.296-326.2001. PMC 88975. PMID 11292640.
- Picardeau M, Brenot A, Saint Girons I (2001). "First evidence for gene replacement in Leptospira spp. Inactivation of L. biflexa flaB results in non-motile mutants deficient in endoflagella". Mol. Microbiol. 40 (1): 189–99. doi:10.1046/j.1365-2958.2001.02374.x. PMID 11298286.
- Cullen PA, Cordwell SJ, Bulach DM, Haake DA, Adler B (2002). "Global Analysis of Outer Membrane Proteins from Leptospira interrogans Serovar Lai". Infect. Immun. 70 (5): 2311–8. doi:10.1128/IAI.70.5.2311-2318.2002. PMC 127947. PMID 11953365.
- Haake DA, Martinich C, Summers TA, Shang ES, Pruetz JD, McCoy AM, Mazel MK, Bolin CA (1998). "Characterization of Leptospiral Outer Membrane Lipoprotein LipL36: Downregulation Associated with Late-Log-Phase Growth and Mammalian Infection". Infect. Immun. 66 (4): 1579–87. doi:10.1128/IAI.66.4.1579-1587.1998. PMC 108091. PMID 9529084.
- Palaniappan RU, Chang YF, Jusuf SS, Artiushin S, Timoney JF, McDonough SP, Barr SC, Divers TJ, Simpson KW, McDonough PL, Mohammed HO (2002). "Cloning and Molecular Characterization of an Immunogenic LigA Protein of Leptospira interrogans". Infect. Immun. 70 (11): 5924–30. doi:10.1128/IAI.70.11.5924-5930.2002. PMC 130282. PMID 12379666.
- Nally JE, Whitelegge JP, Bassilian S, Blanco DR, Lovett MA (2007). "Characterization of the Outer Membrane Proteome of Leptospira interrogans Expressed during Acute Lethal Infection". Infect. Immun. 75 (2): 766–73. doi:10.1128/IAI.00741-06. PMC 1828474. PMID 17101664.
- Verma A, Hellwage J, Artiushin S, Zipfel PF, Kraiczy P, Timoney JF, Stevenson B (2006). "LfhA, a Novel Factor H-Binding Protein of Leptospira interrogans". Infect. Immun. 74 (5): 2659–66. doi:10.1128/IAI.74.5.2659-2666.2006. PMC 1459737. PMID 16622202.
- Barbosa AS, Abreu PA, Neves FO, Atzingen MV, Watanabe MM, Vieira ML, Morais ZM, Vasconcellos SA, Nascimento AL (2006). "A Newly Identified Leptospiral Adhesin Mediates Attachment to Laminin". Infect. Immun. 74 (11): 6356–64. doi:10.1128/IAI.00460-06. PMC 1695492. PMID 16954400.
- Choy HA, Kelley MM, Chen TL, Møller AK, Matsunaga J, Haake DA (2007). "Physiological Osmotic Induction of Leptospira interrogans Adhesion: LigA and LigB Bind Extracellular Matrix Proteins and Fibrinogen". Infect. Immun. 75 (5): 2441–50. doi:10.1128/IAI.01635-06. PMC 1865782. PMID 17296754.
- Werts C, Tapping RI, Mathison JC, Chuang TH, Kravchenko V, Saint Girons I, Haake DA, Godowski PJ, Hayashi F, Ozinsky A, Underhill DM, Kirschning CJ, Wagner H, Aderem A, Tobias PS, Ulevitch RJ (2001). "Leptospiral lipopolysaccharide activates cells through a TLR2-dependent mechanism". Nat. Immunol. 2 (4): 346–52. doi:10.1038/86354. PMID 11276206. S2CID 9658033.
- Que-Gewirth NL, Ribeiro AA, Kalb SR, Cotter RJ, Bulach DM, Adler B, Girons IS, Werts C, Raetz CR (2004). "A Methylated Phosphate Group and Four Amide-linked Acyl Chains in Leptospira interrogans Lipid A. The Membrane Anchor of an Unusual Lipopolysaccharide that Activates TLR2". J. Biol. Chem. 279 (24): 25420–9. doi:10.1074/jbc.M400598200. PMC 2556802. PMID 15044492.
- Nally JE, Chow E, Fishbein MC, Blanco DR, Lovett MA (2005). "Changes in Lipopolysaccharide O Antigen Distinguish Acute versus Chronic Leptospira interrogans Infections". Infect. Immun. 73 (6): 3251–60. doi:10.1128/IAI.73.6.3251-3260.2005. PMC 1111870. PMID 15908349.
- Johnson RC, Harris VG (1967). "Differentiation of Pathogenic and Saprophytic Leptospires I. Growth at Low Temperatures". J. Bacteriol. 94 (1): 27–31. doi:10.1128/jb.94.1.27-31.1967. PMC 251866. PMID 6027998.
- Johnson RC, Gary ND (1963). "NUTRITION OF LEPTOSPIRA POMONA II. : Fatty Acid Requirements". J. Bacteriol. 85 (5): 976–82. doi:10.1128/jb.85.5.976-982.1963. PMC 278270. PMID 14044026.
- Henneberry RC, Cox CD (1970). "Beta-oxidation of fatty acids by Leptospira". Can. J. Microbiol. 16 (1): 41–5. doi:10.1139/m70-007. PMID 5415967.
- Faine S (1959). "Iron as a growth requirement for pathogenic Leptospira". J. Gen. Microbiol. 20 (2): 246–51. doi:10.1099/00221287-20-2-246. PMID 13654718.
- Louvel H, Bommezzadri S, Zidane N, Boursaux-Eude C, Creno S, Magnier A, Rouy Z, Médigue C, Saint Girons I, Bouchier C, Picardeau M (2006). "Comparative and Functional Genomic Analyses of Iron Transport and Regulation in Leptospira spp". J. Bacteriol. 188 (22): 7893–904. doi:10.1128/JB.00711-06. PMC 1636298. PMID 16980464.
- Asuthkar S, Velineni S, Stadlmann J, Altmann F, Sritharan M (2007). "Expression and Characterization of an Iron-Regulated Hemin-Binding Protein, HbpA, from Leptospira interrogans Serovar Lai". Infect. Immun. 75 (9): 4582–91. doi:10.1128/IAI.00324-07. PMC 1951163. PMID 17576761.
- Ren SX, Fu G, Jiang XG, Zeng R, Miao YG, Xu H, Zhang YX, Xiong H, Lu G, Lu LF, Jiang HQ, Jia J, Tu YF, Jiang JX, Gu WY, Zhang YQ, Cai Z, Sheng HH, Yin HF, Zhang Y, Zhu GF, Wan M, Huang HL, Qian Z, Wang SY, Ma W, Yao ZJ, Shen Y, Qiang BQ, Xia QC, Guo XK, Danchin A, Saint Girons I, Somerville RL, Wen YM, Shi MH, Chen Z, Xu JG, Zhao GP (2003). "Unique physiological and pathogenic features of Leptospira interrogans revealed by whole-genome sequencing". Nature. 422 (6934): 888–93. Bibcode:2003Natur.422..888R. doi:10.1038/nature01597. PMID 12712204.
- Nascimento AL, Ko AI, Martins EA, Monteiro-Vitorello CB, Ho PL, Haake DA, Verjovski-Almeida S, Hartskeerl RA, Marques MV, Oliveira MC, Menck CF, Leite LC, Carrer H, Coutinho LL, Degrave WM, Dellagostin OA, El-Dorry H, Ferro ES, Ferro MI, Furlan LR, Gamberini M, Giglioti EA, Góes-Neto A, Goldman GH, Goldman MH, Harakava R, Jerônimo SM, Junqueira-de-Azevedo IL, Kimura ET, Kuramae EE, Lemos EG, Lemos MV, Marino CL, Nunes LR, de Oliveira RC, Pereira GG, Reis MS, Schriefer A, Siqueira WJ, Sommer P, Tsai SM, Simpson AJ, Ferro JA, Camargo LE, Kitajima JP, Setubal JC, Van Sluys MA (2004). "Comparative Genomics of Two Leptospira interrogans Serovars Reveals Novel Insights into Physiology and Pathogenesis". J. Bacteriol. 186 (7): 2164–72. doi:10.1128/JB.186.7.2164-2172.2004. PMC 374407. PMID 15028702.
- Bulach DM, Zuerner RL, Wilson P, Seemann T, McGrath A, Cullen PA, Davis J, Johnson M, Kuczek E, Alt DP, Peterson-Burch B, Coppel RL, Rood JI, Davies JK, Adler B (2006). "Genome reduction in Leptospira borgpetersenii reflects limited transmission potential". Proc. Natl. Acad. Sci. U.S.A. 103 (39): 14560–5. Bibcode:2006PNAS..10314560B. doi:10.1073/pnas.0603979103. PMC 1599999. PMID 16973745.
- Ko AI, Goarant C, Picardeau M (October 2009). "Leptospira: the dawn of the molecular genetics era for an emerging zoonotic pathogen". Nat. Rev. Microbiol. 7 (10): 736–47. doi:10.1038/nrmicro2208. PMC 3384523. PMID 19756012.
- Cerqueira GM, Picardeau M (September 2009). "A century of Leptospira strain typing". Infect. Genet. Evol. 9 (5): 760–8. doi:10.1016/j.meegid.2009.06.009. PMID 19540362.
External links
![]() Data related to Leptospira at Wikispecies
Data related to Leptospira at Wikispecies
- Leptospira page at Kenyon College MicrobeWiki.
- Pasteur Institute — Leptospira Molecular Genetics Server
- "Leptospira". NCBI Taxonomy Browser. 171.