Organophosphate poisoning
Organophosphate poisoning is poisoning due to organophosphates (OPs).[4] Organophosphates are used as insecticides, medications, and nerve agents.[4] Symptoms include increased saliva and tear production, diarrhea, vomiting, small pupils, sweating, muscle tremors, and confusion.[2] While onset of symptoms is often within minutes to hours, some symptoms can take weeks to appear.[5][1] Symptoms can last for days to weeks.[2]
| Organophosphate poisoning | |
|---|---|
| Other names | Organophosphate toxicity, organophosphate overdose, organophosphate intoxication |
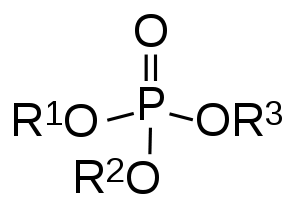 | |
| Phosphoric acid | |
| Specialty | Emergency medicine |
| Symptoms | Increased saliva, diarrhea, small pupils, muscle tremors, confusion |
| Usual onset | Within 12 hours[1] |
| Causes | Organophosphates[2] |
| Diagnostic method | Based on symptoms and confirmed by butyrylcholinesterase[2] |
| Differential diagnosis | Carbamate poisoning[2] |
| Prevention | decreasing access[2] |
| Medication | Atropine, oximes, diazepam[2] |
| Frequency | Nearly 3 million per year[3] |
| Deaths | >200,000 per year[2] |
Organophosphate poisoning occurs most commonly as a suicide attempt in farming areas of the developing world and less commonly by accident.[2] Exposure can be from drinking, breathing in the vapors, or skin exposure.[4] The underlying mechanism involves the inhibition of acetylcholinesterase (AChE), leading to the buildup of acetylcholine (ACh) in the body.[2] Diagnosis is typically based on the symptoms and can be confirmed by measuring butyrylcholinesterase activity in the blood.[2] Carbamate poisoning can present similarly.[2]
Prevention efforts include banning very toxic types of organophosphates.[2] Among those who work with pesticides the use of protective clothing and showering before going home is also useful.[6] In those who have organophosphate poisoning the primary treatments are atropine, oximes such as pralidoxime, and diazepam.[2][4] General measures such as oxygen and intravenous fluids are also recommended.[2] Attempts to decontaminate the stomach, with activated charcoal or other means, have not been shown to be useful.[2] While there is a theoretical risk of health care workers taking care of a poisoned person becoming poisoned themselves, the degree of risk appears to be very small.[2]
OPs are one of the most common causes of poisoning worldwide.[2] There are nearly 3 million poisonings per year resulting in two hundred thousand deaths.[2][3] Around 15% of people who are poisoned die as a result.[2] Organophosphate poisoning has been reported at least since 1962.[7]
Signs and symptoms
The symptoms of organophosphate poisoning include muscle weakness, fatigue, muscle cramps, fasciculation, and paralysis. Other symptoms include hypertension, and hypoglycemia.
Overstimulation of nicotinic acetylcholine receptors in the central nervous system, due to accumulation of ACh, results in anxiety, headache, convulsions, ataxia, depression of respiration and circulation, tremor, general weakness, and potentially coma. When there is expression of muscarinic overstimulation due to excess acetylcholine at muscarinic acetylcholine receptors symptoms of visual disturbances, tightness in chest, wheezing due to bronchoconstriction, increased bronchial secretions, increased salivation, lacrimation, sweating, peristalsis, and urination can occur.[8][9]
The effects of organophosphate poisoning on muscarinic receptors are recalled using the mnemonic SLUDGEM (salivation, lacrimation, urination, defecation, gastrointestinal motility, emesis, miosis)[10] An additional mnemonic is MUDDLES: miosis, urination, diarrhea, diaphoresis, lacrimation, excitation, and salivation.[11]
The onset and severity of symptoms, whether acute or chronic, depends upon the specific chemical, the route of exposure (skin, lungs, or GI tract), the dose, and the individuals ability to degrade the compound, which the PON1 enzyme level will affect.
Reproductive effects
Certain reproductive effects in fertility, growth, and development for males and females have been linked specifically to OP pesticide exposure. Most of the research on reproductive effects has been conducted on farmers working with pesticides and insecticides in rural areas. For those males exposed to OP pesticides, poor semen and sperm quality have been seen, including reduced seminal volume and percentage motility, as well as a decrease in sperm count per ejaculate. In females menstrual cycle disturbances, longer pregnancies, spontaneous abortions, stillbirths, and some developmental effects in offspring have been linked to OP pesticide exposure. Prenatal exposure has been linked to impaired fetal growth and development. The effects of OP exposure on infants and children are at this time currently being researched to come to a conclusive finding.[12][13] Evidence of OP exposure in pregnant mothers are linked to several health effects in the fetus. Some of these effects include delayed mental development, Pervasive developmental disorder (PDD),[9] morphological abnormalities in the cerebral surface.[14]
Neurotoxic effects
Neurotoxic effects have also been linked to poisoning with OP pesticides causing four neurotoxic effects in humans: cholinergic syndrome, intermediate syndrome, organophosphate-induced delayed polyneuropathy (OPIDP), and chronic organophosphate-induced neuropsychiatric disorder (COPIND). These syndromes result after acute and chronic exposure to OP pesticides.
Cholinergic syndrome occurs in acute poisonings with OP pesticides and is directly related to levels of AChE activity. Symptoms include miosis, sweating, lacrimation, gastrointestinal symptoms, respiratory difficulties, shortness of breath, slowed heart rate, cyanosis, vomiting, diarrhea, trouble sleeping, as well as other symptoms. Along with these central effects can be seen and finally seizures, convulsions, coma, respiratory failure. If the person survives the first day of poisoning, personality changes can occur, in addition to aggressive behavior, psychotic episodes, memory and attention disturbances, and other delayed effects. When death occurs, it is most commonly due to respiratory failure due to paralysis of respiratory muscles and depression of central nervous system, which is responsible for respiration. For people affected by cholinergic syndrome, atropine sulfate combined with an oxime is used to combat the effects of the acute OP poisoning. Diazepam is sometimes also administered if convulsions or muscle fasciculations begin.[15]
The intermediate syndrome (IMS) appears in the interval between the end of the cholinergic crisis and the onset of OPIDP. Symptoms associated with IMS manifest between 24 and 96 hours after exposure. The exact etiology, incidence, and risk factors associated with IMS are not well understood, but IMS is recognized as a disorder of neuromuscular junctions. IMS occurs when a person has a prolonged and severe inhibition of AChE. It has been linked to specific OP pesticides such as parathion, methylparathion, and dichlorvos. Patients generally present with increasing weakness in the facial, neck flexor, and respiratory muscles.
OPIDP occurs in a small percentage of cases, roughly two weeks after exposure, where temporary paralysis occurs. This loss of function and ataxia of peripheral nerves and spinal cord is the phenomenon of OPIDP. Once the symptoms begin with shooting pains in both legs, the symptoms continue to worsen for 3–6 months. In the most severe cases quadriplegia has been observed. Treatment only affects sensory nerves, not motor neurons which may permanently lose function. The aging and phosphorylation of more than 70% of functional NTE in peripheral nerves is one of the processes involved in OPIDP.[11][15] Standard treatments for OP poisoning are ineffective for OPIDP.
COPIND occurs without cholinergic symptoms and is independent of AChE inhibition. COPIND appears with a delay and is long lasting. Symptoms associated with COPIND include cognitive deficit, mood changes, autonomic dysfunction, peripheral neuropathy, and extrapyramidal symptoms. The underlying mechanisms of COPIND have not been determined, but it is hypothesized that withdrawal of OP pesticides after chronic exposure or acute exposure could be a factor.[15]
Pregnancy
Evidence of exposure to OP pesticides during gestation and early postnatal period have been linked to neurodevelopmental effects in animals, specifically rats. Animals exposed in utero to chlorpyrifos exhibited decreased balance, poorer cliff avoidance, decreased locomotion, delays in maze performance, and increased gait abnormalities. Early gestation is believed to be a critical time period for the neurodevelopmental effects of pesticides. OPs affect the cholinergic system of fetuses, so exposure to chlorpyrifos during critical periods of brain development potentially could cause cellular, synaptic, and neurobehavioral abnormalities in animals.[16] In rats exposed to methylparathion, studies found reduced AChE activity in all brain regions and subtle alterations in behaviors such as locomotor activity and impaired cage emergence. Organophosphates as whole have been linked to decreases in the length of limbs, head circumference, and slower rates of postnatal weight gain in mice.[9]
Cancer
The International Agency for Research on Cancer (IARC) found that organophosphate exposure may increase cancer risk.[17] Tetrachlorvinphos and parathion were classified as "possibly carcinogenic", malathion, and diazinon.[17]
Cause
OP pesticide exposure occurs through inhalation, ingestion and dermal contact.[18] Because OP pesticides degrade quickly when exposed to air and light, they have been considered relatively safe to consumers.[19] However, OP residues may linger on fruits and vegetables.[20] Certain OP pesticides have been banned for use on some crops, For example, methyl parathion is banned from use on some crops and permitted on others.[21] It can also occur through deliberate poisoning using nerve agents such as sarin and tabun.
Examples
- Insecticides including malathion, parathion, diazinon, fenthion, dichlorvos, chlorpyrifos, ethion, trichlorfon
- Nerve agents including soman, sarin, tabun, VX
- Herbicides including tribufos [DEF], merphos are tricresyl phosphate–containing industrial chemicals.
Exposure to any of the above-listed organophosphates may occur through inhalation, skin absorption, and ingestion, most commonly of food that has been treated with an OP herbicide or insecticide. Exposure to these chemicals can occur at public buildings, schools, residential areas, and in agricultural areas. Chlorpyrifos and Malathion have been linked to reproductive effects, neurotoxicity, kidney/liver damage, and birth defects. Dichlorvos has also been linked to reproductive effects, neurotoxicity, and kidney/liver damage. It is also recognized to be a possible carcinogen.[22]
Pathophysiology
The health effects associated with organophosphate poisoning are a result of excess acetylcholine (ACh) present at different nerves and receptors in the body because acetylcholinesterase is blocked. Accumulation of ACh at motor nerves causes overstimulation of nicotinic expression at the neuromuscular junction. When there is an accumulation of ACh at autonomic ganglia synapses this causes overstimulation of muscarinic expression in the parasympathetic nervous system.
Organophosphates irreversibly and non-competitively inhibit acetylcholinesterase, causing poisoning by phosphorylating the serine hydroxyl residue on AChE, which inactivates AChE. AChE is critical for nerve function, so the inhibition of this enzyme, which causes acetylcholine accumulation, results in muscle overstimulation. This causes disturbances across the cholinergic synapses and can only be reactivated very slowly, if at all. Paraoxonase (PON1) is a key enzyme involved in OP toxicity and has been found to be critical in determining an organism's sensitivity to OP exposure.
PON1 can inactivate some OPs through hydrolysis. PON1 hydrolyzes the active metabolites in several OP insecticides such as chlorpyrifos oxon, and diazoxon, as well as, nerve agents such as soman, sarin, and VX. PON1 hydrolyzes the metabolites, not the parent compounds of insectides.[23] The presence of PON1 polymorphisms causes there to be different enzyme levels and catalytic efficiency of this esterase, which in turn suggests that different individuals may be more susceptible to the toxic effect of OP exposure. Higher levels of PON1 plasma hydrolytic activity provide a greater degree of protection against OP pesticides. Rats injected with purified PON1 from rabbit serum were more resistant to acute cholinergic activity than the control rats. PON1 knockouts in mice are found to be more sensitive to the toxicity of pesticides, like chlorpyrifos. Animal experiments indicate that while PON1 plays a significant role in regulating the toxicity of OPs its degree of protection given depends on the compound (i.e. Chlorpyrifos oxon or diazoxon). The catalytic efficiency with which PON1 can degrade toxic OPs determines the degree of protection that PON1 can provide for organism. The higher the concentration of PON1 the better the protection provided. PON1 activity is much lower in neonates, so neonates are more sensitive to OP exposure.[23] In 2006, reports up to a 13-fold variation was seen in PON1 levels in adults, as well as, specifically regarding sensitivity to diazoxon, a variation up to 26 and 14-fold was reported in a group of newborns and Latino mothers. This wide range in variability of enzyme levels determining a human's sensitivity to various OPs is being researched further.[24]
Diagnosis
A number of measurements exist to assess exposure and early biological effects for organophosphate poisoning. Measurements of OP metabolites in both the blood and urine can be used to determine if a person has been exposed to organophosphates. Specifically in the blood, metabolites of cholinesterases, such as butyrylcholinesterase (BuChE) activity in plasma, neuropathy target esterase (NTE) in lymphocytes, and of acetylcholinesterase (AChE) activity in red blood cells.[23] Due to both AChE and BuChE being the main targets of organophosphates, their measurement is widely used as an indication of an exposure to an OP. The main restriction on this type of diagnosis is that depending on the OP, the degree to which either AChE or BuChE are inhibited differs; therefore, measure of metabolites in blood and urine do not specify which OP agent is responsible for the poisoning.[23][25] However, for fast initial screening, determining AChE and BuChE activity in the blood are the most widely used procedures for confirming a diagnosis of OP poisoning.[25] The most widely used portable testing device is the Test-mate ChE field test,[26][27]: 13 which can be used to determine levels of Red Blood Cells (RBC), AChE and plasma (pseudo) cholinesterase (PChE) in the blood in about four minutes. This test has been shown to be just as effective as a regular laboratory test and because of this, the portable ChE field test is frequently used by people who work with pesticides on a daily basis.[28]
Treatment
Current antidotes for OP poisoning consist of a pretreatment with carbamates to protect AChE from inhibition by OP compounds and post-exposure treatments with anti-cholinergic drugs. Anti-cholinergic drugs work to counteract the effects of excess acetylcholine and reactivate AChE. Atropine can be used as an antidote in conjunction with pralidoxime or other pyridinium oximes (such as trimedoxime or obidoxime),[29][30] though the use of "-oximes" has been found to be of no benefit, or to be possibly harmful, in at least two meta-analyses.[31][32] Atropine is a muscarinic antagonist, and thus blocks the action of acetylcholine peripherally.[33] These antidotes are effective at preventing lethality from OP poisoning, but current treatment lack the ability to prevent post-exposure incapacitation, performance deficits, or permanent brain damage.[34] While the efficacy of atropine has been well-established, clinical experience with pralidoxime has led to widespread doubt about its efficacy in treatment of OP poisoning.[35]
Enzyme bioscavengers are being developed as a pretreatment to sequester highly toxic OPs before they can reach their physiological targets and prevent the toxic effects from occurring. Significant advances with cholinesterases (ChEs), specifically human serum BChE (HuBChE) have been made. HuBChe can offer a broad range of protection for nerve agents including soman, sarin, tabun, and VX. HuBChE also possess a very long retention time in the human circulation system and because it is from a human source it will not produce any antagonistic immunological responses. HuBChE is currently being assessed for inclusion into the protective regimen against OP nerve agent poisoning.[34] Currently there is potential for PON1 to be used to treat sarin exposure, but recombinant PON1 variants would need to first be generated to increase its catalytic efficiency.
Another potential treatment being researched is the Class III anti-arrhythmic agents. Hyperkalemia of the tissue is one of the symptoms associated with OP poisoning. While the cellular processes leading to cardiac toxicity are not well understood, the potassium current channels are believed to be involved. Class III anti-arrhythmic agents block the potassium membrane currents in cardiac cells, which makes them a candidate for become a therapeutic of OP poisoning.[36]
There is insufficient evidence to support using plasma alkalinisation to treat a person with organophosphate poisoning.[37]
Epidemiology
Organophosphate pesticides are one of the top causes of poisoning worldwide, with an annual incidence of poisonings among agricultural workers varying from 3-10% per country.[38]
History
Ginger Jake
A striking example of OPIDN occurred during the 1930s Prohibition Era when thousands of men in the American South and Midwest developed arm and leg weakness and pain after drinking a "medicinal" alcohol substitute. The drink, called "Ginger Jake," contained an adulterated Jamaican ginger extract containing tri-ortho-cresyl phosphate (TOCP) which resulted in partially reversible neurologic damage. The damage resulted in the limping "Jake Leg" or "Jake Walk" which were terms frequently used in the blues music of the period. Europe and Morocco both experienced outbreaks of TOCP poisoning from contaminated abortifacients and cooking oil, respectively.[39][40]
Gulf War syndrome
Research has linked the neurological abnormalities found in Persian Gulf War veterans who have chronic multisymptom illnesses to exposure to wartime combinations of organophosphate chemical nerve agents. Before, it was believed that veterans had a psychologically based disorder or depression, most likely post-traumatic stress disorder (PTSD). Many veterans were given pyridostigmine bromide (PB) pills to protect against nerve gas agents such as sarin and soman. During the war veterans were exposed to combinations of organophosphate pesticides and nerve agents, which produced symptoms associated with chronic organophosphate-induced delayed polyneuropathy (OPIDP) syndrome. Similar symptoms found in the veterans were the same symptoms reported for individuals in occupational settings who were acutely poisoned by organophosphates, such as chlorpyrifos. Studies found veterans experienced deficits in intellectual and academic abilities, simple motor skills, memory impairment, and impaired emotional function. These symptoms indicate brain damage, not a psychologically based disorder.[41][42][43]
Society and culture
United States
Under a 1988 amendment to the Federal Insecticide, Fungicide and Rodenticide Act (FIFRA), the U.S. Environmental Protection Agency (EPA) regulates organophosphate pesticides[44] Its focus was initially on registering pesticides for use on food crops. No pesticide can be sold in the United States before the EPA has reviewed the manufacturer's application for registration and determined that the use of the product will not present an unreasonable risk to the public or the environment.[45] In 1996, with the passage of the Food Quality Protection Act, Congress required the EPA to reassess all existing pesticide tolerances with specific consideration for children.[46] This resulted in a 10-year review process of the health and environmental effects of all pesticides, beginning with the organophosphates. As part of that process, in 1999 the EPA announced a ban the use of organophosphate pesticide methyl parathion and significant restrictions on the use of another OP, azinphos methyl, in what they called "kid's food".[47] The review process was concluded in 2006 and eliminated or modified thousands of other uses of pesticides.[48] Other legislative action has been taken to protect children from the risks of organophosphates.
Many non-governmental and research groups, as well as the EPA's Office of Inspector General, have expressed concerns that the review did not take into account possible neurotoxic effects on developing fetuses and children, an area of developing research. OIG report. A group of leading EPA scientists sent a letter to the chief administrator, Stephen Johnson, decrying the lack of developmental neurotoxicity data in the review process. EPA Letter EHP article New studies have shown toxicity to developing organisms during certain "critical periods" at doses much lower than those previously suspected to cause harm.[49]
Even the restrictions which did successfully pass have been controversial. For example, in 1999 the EPA restricted the use of chlorpyrifos in households (under the commercial name Dursban). However, the EPA did not limit its use in agriculture. Chlorpyrifos remains one of the most widely used pesticides.[50] This may soon change. On February 8, 2013, the EPA requested comment on a preliminary evaluation of the potential risks to children and other bystanders from volatilization of chlorpyrifos from treated crops[51]
Vulnerable groups
Some populations are more vulnerable to pesticide poisoning. In the United States, farmworkers can be exposed via direct spray, drift, spills, direct contact with treated crops or soil, or defective or missing protective equipment.[52] Migrant workers may be at an especially high risk of chronic exposure as over the course of a growing season, they may work at multiple farms, thus increasing their exposure to pesticides.[52] Farmworkers in more permanent positions may receive more safety training and/or more "consistent reinforcement of safety behaviors than seasonal farmworkers or day laborers."[6] For migrant farmworkers, language barriers and/or education level could be a barrier to understanding posted warning signs, labels and safety warnings located on the pesticides, or understanding any safety training that is provided.[6]
Other factors that may lead to greater exposure for the migrant farmworker population include: limited or no access to safety equipment, little to no control over pesticide use, cultural factors, and fear of job loss if they report potential hazards.[53][52][6] Studies have also shown that there are some key beliefs by farmworkers that may exacerbate pesticide exposure, including the belief that "pesticides must be felt, seen, tasted, or smelled to be present; the skin blocks absorption and body openings facilitate it; exposure occurs only when a pesticide is wet;…and acute, not low-level chronic exposure is the primary danger."[6]
This, coupled with the difficulty or uncertainty of recognizing and/or diagnosing chronic pesticide poisoning by the medical community,[54] makes it difficult for exposed workers to receive an effective remedy.[52] Migrant workers may also be hesitant to seek-out medical care due to lack of health insurance, language barriers, immigration status, cost, cultural factors, lack of transportation, fear of job loss, and lack of awareness of workers' compensation benefits.[53]
Sergei and Yulia Skripal
In March 2018, Sergei Skripal and his daughter were poisoned in Salisbury, England, with an organophosphate poison known as a Novichok agent.[55] Both fell unconscious while sitting on a park bench. A first responder to the scene also became contaminated and had symptoms of organophosphate poisoning. All three survived after hospital treatment. Despite continually denying responsibility for the attack, Russia is suspected to be behind the poisonings.
Alexei Navalny
On 20 August 2020, Russian politician Alexei Navalny developed life-threatening acute poisoning symptoms on a flight. He was later transferred to Berlin, where poisoning by a cholinesterase inhibitor was diagnosed and confirmed by multiple tests in independent laboratories.[56]
References
- Stoller JK, Michota FA, Mandell BF (2009). The Cleveland Clinic Foundation Intensive Review of Internal Medicine. Lippincott Williams & Wilkins. p. 108. ISBN 9780781790796. Archived from the original on 2017-09-10.
- Eddleston M, Buckley NA, Eyer P, Dawson AH (February 2008). "Management of acute organophosphorus pesticide poisoning". Lancet. 371 (9612): 597–607. doi:10.1016/S0140-6736(07)61202-1. PMC 2493390. PMID 17706760.
- Berg S, Bittner EA (2013). The MGH Review of Critical Care Medicine. Lippincott Williams & Wilkins. p. 298. ISBN 9781451173680. Archived from the original on 2017-09-10.
- King AM, Aaron CK (February 2015). "Organophosphate and carbamate poisoning". Emergency Medicine Clinics of North America. 33 (1): 133–51. doi:10.1016/j.emc.2014.09.010. PMID 25455666.
- Peter JV, Sudarsan TI, Moran JL (November 2014). "Clinical features of organophosphate poisoning: A review of different classification systems and approaches". Indian Journal of Critical Care Medicine. 18 (11): 735–45. doi:10.4103/0972-5229.144017. PMC 4238091. PMID 25425841.
- Quandt SA, Hernández-Valero MA, Grzywacz JG, Hovey JD, Gonzales M, Arcury TA (June 2006). "Workplace, household, and personal predictors of pesticide exposure for farmworkers". Environmental Health Perspectives. 114 (6): 943–52. doi:10.1289/ehp.8529. PMC 1480506. PMID 16759999.
- Neurological Practice: An Indian Perspective. Elsevier India. 2005. p. 479. ISBN 9788181475497. Archived from the original on 2017-09-10.
- Leibson T, Lifshitz M (2008). "Organophosphate and Carbamate Poisoning: Review of the Current Literature and Summary of Clinical and Laboratory Experience in Southern Israel". J Toxicol. 10 (11): 767–7704. PMID 19070283.
- Eskenazi B, Bradman A, Castorina R (1999). "Exposures of Children to Organophosphate Pesticides and Their Potential Adverse Health Effects". Environmental Health Perspectives. 107: 409–419. doi:10.1289/ehp.99107s3409. PMC 1566222. PMID 10346990.
- Toxicity, Organophosphate and Carbamate at eMedicine
- Moore C (2009). Children and Pollution: Why Scientists Disagree. Oxford University Press. pp. 109–112. ISBN 978-0-19-538666-0.
- Woodruff T, Janssen S, Guillete L, Giudice L (2010). Environmental Impacts on Reproductive Health and Fertility. Cambridge University Press. pp. 109. ISBN 978-0-521-51952-6.
- Peiris-John R, Wickremasinghe R (2008). "Impact of low-level exposure to organophosphates on human reproduction and survival". Royal Society of Tropical Medicine and Hygiene. 102 (3): 239–245. doi:10.1016/j.trstmh.2007.11.012. PMID 18242652.
- Rauh V, Arunajadai S, Horton M, et al. (2011). "Seven-year neurodevelopmental scores and prenatal exposure to chlorpyrifos, a common agricultural pesticide". Environmental Health Perspectives. 119 (8): 1189–1195. doi:10.1289/ehp.1003160. PMC 3237355. PMID 21507777.
- Jokanovic M, Kosanovic M (2010). "Neurotoxic effects in patients poisoned with organophosphate pesticides". Environmental Toxicology and Pharmacology. 29 (3): 195–201. doi:10.1016/j.etap.2010.01.006. PMID 21787602.
- Eskenazi B, Harley K, Bradman A, Weltzien E, Jewell N, Barr D, Furlong C, Holland N (2004). "Association of in Utero Organophosphate Pesticide Exposure and Fetal Growth and Length of Gestation in an Agricultural Population". Environmental Health Perspectives. 112 (10): 116–1124. doi:10.1289/ehp.6789. PMC 1247387. PMID 15238287.
- "IARC Monographs Volume 112: evaluation of five organophosphate insecticides and herbicides" (PDF). World Health Organization. Archived (PDF) from the original on 2017-04-17.
- "Cholinesterase Inhibition". Archived from the original on 2013-04-02.
- "Pesticide Application and Safety Training for Applicators of Public Health Pesticides". Archived from the original on 2010-08-29. Retrieved 2013-03-25.
- "Because some foods carry organophosphate residues".
- "Methyl Parathion Risk Management Decision". Archived from the original on 2013-04-01. Retrieved 2013-03-25.
- "Health Effects of 30 Commonly Used Lawn Pesticides" (PDF). Archived (PDF) from the original on 2011-11-12.
- Costa L, Cole T, Vitalone A, Furlong C (2005). "Measurement of paraoxonase (PON1) status as a potential biomarker of susceptibility to organophosphate toxicity". Clinica Chimica Acta. 352 (1–2): 37–47. doi:10.1016/j.cccn.2004.09.019. PMID 15653099.
- "Organophosphorous Cumulative Risk Assessment 2006 Update" (PDF). US EPA Office of Pesticide Programs. Archived (PDF) from the original on 2011-06-04. Retrieved 2011-12-10.
- Worek F, Koller M, Thiermann H, Szinicz L (2005). "Diagnostic aspects of organophosphate poisoning". J Toxicol. 214 (3): 182–9. doi:10.1016/j.tox.2005.06.012. PMID 16051411.
- EQM Research. Test-mate Official Site Archived 2014-12-17 at the Wayback Machine Page accessed 8 Feb 2015.
- Paul L. Knechtges. May 2008 USACEHR Technical Report 0801: An Evaluation of Blood Cholinesterase Testing Methods for Military Health Surveillance Archived 2015-02-09 at the Wayback Machine
- K.D. Katz, D.E. Brooks, "Organophosphate Toxicity Workup" Archived 2015-02-09 at the Wayback Machine, "MedScape", 27 Jan. 2015
- Jokanović M, Prostran M (2009). "Pyridinium oximes as cholinesterase reactivators. Structure-activity relationship and efficacy in the treatment of poisoning with organophosphorus compounds". Curr. Med. Chem. 16 (17): 2177–88. doi:10.2174/092986709788612729. PMID 19519385. Archived from the original on 2017-09-10. Retrieved 2020-12-23.
- Balali-Mood M, Shariat M (1998). "Treatment of organophosphate poisoning. Experience of nerve agents and acute pesticide poisoning on the effects of oximes". Journal of Physiology. 92 (5–6): 375–378. doi:10.1016/s0928-4257(99)80008-4. PMID 9789840. S2CID 24915367.
- Rahimi R, Nikfar S, Abdollahi M (March 2006). "Increased morbidity and mortality in acute human organophosphate-poisoned patients treated by oximes: a meta-analysis of clinical trials". Hum Exp Toxicol. 25 (3): 157–62. doi:10.1191/0960327106ht602oa. PMID 16634335. S2CID 33212211.
- Peter JV, Moran JL, Graham P (February 2006). "Oxime therapy and outcomes in human organophosphate poisoning: an evaluation using meta-analytic techniques". Crit. Care Med. 34 (2): 502–10. doi:10.1097/01.CCM.0000198325.46538.AD. PMID 16424734. S2CID 22309335.
- Walker C (2001). Organic Pollutants: An Ecotoxicological Perspective. Taylor & Francis. pp. 186–193. ISBN 978-0-7484-0962-4.
- Doctor B, Saxena A (2005). "Bioscavengers for the protection of humans against organophosphate toxicity". Chemico-Biological Interactions. 157–158: 167–171. doi:10.1016/j.cbi.2005.10.024. PMID 16293236.
- Banerjee I, Tripathi SK, Roy AS (2014). "Efficacy of pralidoxime in organophosphorus poisoning: Revisiting the controversy in Indian setting". J Postgrad Med. 60 (1): 27–30. doi:10.4103/0022-3859.128803. PMID 24625936.
- Zoltani C, Baskin S (2002). "Organophosphate Caused Cardia Toxicity: Action Potential Dynamics in Atrial Tissue". Army Research Laboratory: 1–15.
- Roberts D, Buckley NA (January 2005). "Alkalinisation for organophosphorus pesticide poisoning". The Cochrane Database of Systematic Reviews. 2010 (1): CD004897. doi:10.1002/14651858.CD004897.pub2. PMC 8855931. PMID 15674967.
- Freire C, Koifman S (2013). "Pesticides, depression and suicide: A systematic review of the epidemiological evidence". International Journal of Hygiene and Environmental Health. 216 (4): 445–460. doi:10.1016/j.ijheh.2012.12.003. PMID 23422404.
- Morgan JP, Tulloss TC (December 1976). "The Jake Walk Blues. A toxicologic tragedy mirrored in American popular music". Ann. Intern. Med. 85 (6): 804–8. doi:10.7326/0003-4819-85-6-804. PMID 793467.
- Segalla Spencer (2011). "The 1959 Moroccan Oil Poisoning and US Cold War Disaster Diplomacy". Journal of North African Studies. 17 (2): 315–336. doi:10.1080/13629387.2011.610118. S2CID 144007393.
- Horn J, Haley R, Kurt T (1997). "Neuropsychological Correlates of Gulf War Syndrome". Archives of Clinical Neuropsychology. 12 (6): 531–544. doi:10.1093/arclin/12.6.531. PMID 14590665.
- Haley R, Kurt T (Jan 1997). "Self-reported exposure to neurotoxic chemical combinations in the Gulf War: A cross-sectional epidemiologic study". Journal of the American Medical Association. 277 (3): 231–237. doi:10.1001/jama.277.3.231. PMID 9005273.
- Toomy R, Alpern R, Vasterling J, Backer D, Reda D, Lyons M, Henderson W, Kang H, Eisen S, Murphy F (2009). "Neuropsychological functioning of U.S. Gulf War veterans 10 years after the war". Journal of the International Neuropsychological Society. 15 (5): 717–29. doi:10.1017/S1355617709990294. PMID 19640317. S2CID 9689427.
- "Pesticide Registration (PR) Notice 1988-6" (PDF). Archived (PDF) from the original on 2012-09-25. Retrieved 2013-03-28.
- "Setting Tolerances for Pesticide Residues in Foods". Archived from the original on 2013-04-01. Retrieved 2013-03-28.
- "Food Quality Protection Act (FQPA) of 1996". Archived from the original on 2012-11-04. Retrieved 2013-03-28.
- "Children Are at Greater Risks from Pesticide Exposure". United States Environmental Protection Agency. January 2002. Archived from the original on 20 February 2015. Retrieved 18 February 2015.
- Janofsky, Michael. "E.P.A. Recommends Limits On Thousands of Pesticides" Archived 2016-03-04 at the Wayback Machine, The New York Times, August 4, 2006, accessed April 1, 2008.
- Slotkin TA, Levin ED, Seidler FJ (May 2006). "Comparative developmental neurotoxicity of organophosphate insecticides: effects on brain development are separable from systemic toxicity". Environmental Health Perspectives. 114 (5): 746–51. doi:10.1289/ehp.8828. PMC 1459930. PMID 16675431.
- "Reregistration Eligibility Decision for Chlorpyrifos" (PDF). 31 July 2006. Archived from the original (PDF) on 19 November 2012.
- "Pesticide News Story: Chlorpyrifos Preliminary Volatilization Assessment Suggests Bystander Risks of Concern; EPA Requests Comment to Address Uncertainties". 2013-02-08. Archived from the original on 2013-04-14. Retrieved 2013-03-28.
- Human Rights Council. Report of the Special Rapporteur on the right to food. 2017; General Assembly, 34th session, Agenda item 3. "Ods Home Page" (PDF). Archived (PDF) from the original on 2017-03-25. Retrieved 2017-03-24.
- Pesticide Safety. "Children's Vaccine Song". Archived from the original on 2017-03-25. Retrieved 2017-03-24.
- Jaga, K. & Dharmani, C. Ocular toxicity from pesticide exposure: A recent review. Environ. Health Prev. Med. (2006) 11: 102. doi:10.1265/ehpm.11.102
- Benjamin Kentish (12 April 2018). "Poison used on Sergei and Yulia Skripal in Salisbury attack was novichok nerve agent, confirms chemical weapons watchdog". The Independent.
- "Statement by Charité: Clinical findings indicate Alexei Navalny was poisoned". Charité Berlin. 2020-08-20.