Polyene antimycotic
Polyene antimycotics, sometimes referred to as polyene antibiotics, are a class of antimicrobial polyene compounds that target fungi.[1] These polyene antimycotics are typically obtained from some species of Streptomyces bacteria. Previously, polyenes were thought to bind to ergosterol in the fungal cell membrane and thus weakening it and causing leakage of K+ and Na+ ions, which could contribute to fungal cell death. However, more detailed studies of polyene molecular properties have challenged this model suggesting that polyenes instead bind and extract ergosterol directly from the cellular membrane thus disrupting the many cellular functions ergosterols perform.[2][3] Amphotericin B, nystatin, and natamycin are examples of polyene antimycotics. They are a subgroup of macrolides.[4]
Structures
Their chemical structures feature a large ring of atoms (in essence, a cyclic ester ring) containing multiple conjugated carbon-carbon double bonds (hence polyene) on one side of the ring and multiple hydroxyl groups bonded to the other side of the ring. Their structures also often have a D-mycosamine (a type of amino-glycoside) group bonded to the molecule.[5] The series of conjugated double bonds typically absorbs strongly in the ultraviolet-visible region of the electromagnetic spectrum, often resulting in the polyene antibiotics having a yellow color.
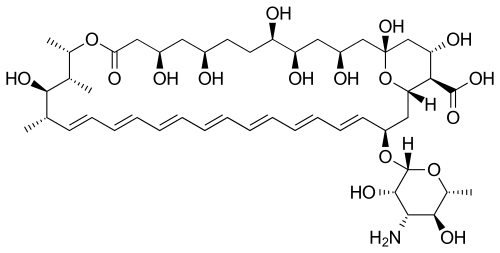

Biosynthesis
The natural route to synthesis includes polyketide synthase components.[6]
References
- "Antifungal Agents". Polyene Antifungal Drugs. NCBI Bookshelf. The University of Texas Medical Branch at Galveston. 1996. ISBN 9780963117212. Retrieved 29 January 2010.
- Thomas M, Anderson; Clay C, Mary; Cioffi G, Alexander; Diaz A, Katrina; Hisao S, Grant; Turrle D, Marcus; Nieuwkoop J, Andrew; Comellas, Gemma; Maryum, Nashrah; Wang, Shu; Uno E, Bruce; Wildeman L, Erin; Tamir, Gonene; Rienstra M, Chad; Burke D, Martub (Mar 30, 2014). "Amphotericin forms an extramembranous and fungicidal sterol sponge". Nature Chemical Biology. 10 (5): 400–406. doi:10.1038/nchembio.1496. PMC 3992202. PMID 24681535.
- Robbins, Nicole; Caplan, Tavia; Cowen, Leah E. (September 8, 2017). "Molecular Evolution of Antifungal Drug Resistance". Annual Review of Microbiology. 71: 753–775. doi:10.1146/annurev-micro-030117-020345. ISSN 1545-3251. PMID 28886681.
- Hamilton-Miller (1973). "Chemistry and Biology of the Polyene Macrolide Antibiotics". Bacteriological Reviews. American Society for Microbiology. 37 (2): 166–196. doi:10.1128/BR.37.3.166-196.1973. PMC 413810. PMID 4578757.
- Volpon, Laurent; Lancelin, Jean-Marc (2002). "Solution NMR structure of five representative glycosylated polyene macrolide antibiotics with a sterol-dependent antifungal activity". European Journal of Biochemistry. 269 (18): 4533–4541. doi:10.1046/j.1432-1033.2002.03147.x. PMID 12230565.
- Khan N, Rawlings B, Caffrey P (Jan 26, 2011). "A labile point in mutant amphotericin polyketide synthases". Biotechnol. Lett. 33 (6): 1121–6. doi:10.1007/s10529-011-0538-3. PMID 21267757. S2CID 10209476.