Prostate cancer screening
Prostate cancer screening is the screening process used to detect undiagnosed prostate cancer in men without signs or symptoms.[1][2] When abnormal prostate tissue or cancer is found early, it may be easier to treat and cure, but it is unclear if early detection reduces mortality rates.[2]
| Prostate cancer screening | |
|---|---|
| Purpose | Detect prostate cancer (when no symptoms are present) |
Screening precedes a diagnosis and subsequent treatment. The digital rectal examination (DRE) is one screening tool, during which the prostate is manually assessed through the wall of the rectum. The second screening tool is the measurement of prostate-specific antigen (PSA) in the blood. The evidence remains insufficient to determine whether screening with PSA or DRE reduces mortality from prostate cancer.[1] A 2013 Cochrane review concluded PSA screening results in "no statistically significant difference in prostate cancer-specific mortality...".[3] The American studies were determined to have a high bias. European studies included in this review were of low bias and one reported "a significant reduction in prostate cancer-specific mortality." PSA screening with DRE was not assessed in this review. DRE was not assessed separately.[3]
Most recent guidelines have recommended that the decision whether or not to screen should be based on shared decision-making,[4] so that men are informed of the risks and benefits of screening.[5][6] In 2012, the American Society of Clinical Oncology recommends screening be discouraged for those who are expected to live less than ten years, while for those with a longer life expectancy a decision should be made by the person in question. In general, they conclude that based on recent research, "it is uncertain whether the benefits associated with PSA testing for prostate cancer screening are worth the harms associated with screening and subsequent unnecessary treatment."[7] However, by March 2022, experts who reviewed the latest evidence for the European Commission's Scientific Advice Mechanism judged that the balance of harms and benefits had changed due to technological improvements, and for the first time recommended organised prostate cancer screening at the population level, using blood tests; and they specifically advised against offering opportunistic tests to asymptomatic individuals outside of such screening programmes.[8]
Prostate biopsies are used to diagnose prostate cancer but are not done on asymptomatic men and therefore are not used for screening.[9][10] Infection after prostate biopsy occurs in about 1%, while death occurs as a result of biopsy in 0.2%.[11][12] Prostate biopsy guided by magnetic resonance imaging has improved the diagnostic accuracy of the procedure.[13][14]
Prostate-specific antigen
Prostate-specific antigen (PSA) is secreted by the epithelial cells of the prostate gland and can be detected in a sample of blood.[15] PSA is present in small quantities in the serum of men with healthy prostates, but is often elevated in the presence of prostate cancer or other prostate disorders.[16] PSA is not a unique indicator of prostate cancer, but may also detect prostatitis or benign prostatic hyperplasia.[17]
A 2018 United States Preventive Services Task Force (USPSTF) draft adjusted the prior opposition to PSA screening.[18] It suggests shared decision-making regarding screening in healthy males 55 to 69 years of age.[18] The final recommendation for that age group states screening should only be done in those who wish it.[19] In those 70 and over, screening remains not recommended.[19]
Screening with PSA has been associated with a number of harms including over-diagnosis, increased prostate biopsy with associated harms, increased anxiety, and unneeded treatment.[20] The evidence surrounding prostate cancer screening indicates that it may cause little to no difference in mortality.[3]
On the other hand, up to 25% of men diagnosed in their 70s or even 80s die of prostate cancer, if they have high-grade (i.e., aggressive) prostate cancer.[21] Conversely, some argue against PSA testing for men who are too young, because too many men would have to be screened to find one cancer, and too many men would have treatment for cancer that would not progress. Low-risk prostate cancer does not always require immediate treatment, but may be amenable to active surveillance.[22] A PSA test cannot 'prove' the existence of prostate cancer by itself; varying levels of the antigen can be due to other causes.[23]
Digital rectal examination
During a digital rectal examination (DRE), a healthcare provider slides a gloved finger into the rectum and presses on the prostate, to check its size and to detect any lumps on the accessible side. If the examination suggests anomalies, a PSA test is performed. If an elevated PSA level is found, a follow-up test is then performed.[2]
A 2018 review recommended against primary care screening for prostate cancer with DRE due to the lack of evidence of the effectiveness of the practice.[24]
The USPSTF recommends against digital rectal examination as a screening tool due to lack of evidence of benefits.[25] Although DRE has long been used to diagnose prostate cancer, no controlled studies have shown a reduction in the morbidity or mortality of prostate cancer when detected by DRE at any age.[26][27]
The American Urological Association in 2018 stated that for men aged 55 to 69, they could find no evidence to support the continued use of DRE as a first-line screening test; however, in men referred for an elevated PSA, DRE may be a useful secondary test.[28]
Follow-up tests
Biopsy
Prostate biopsies are considered the gold standard in detecting prostate cancer.[9][10] Infection is a possible risk.[11] MRI guided techniques have improved the diagnostic accuracy of the procedure.[13][14] Biopsies can be done through the rectum or penis.
Ultrasound
Transrectal ultrasonography (TRUS) has the advantage of being fast and minimally invasive, and better than MRI for the evaluation of superficial tumor.[2] It also gives details about the layers of the rectal wall, accurate and useful for staging primary rectal cancer. While MRI is better in visualization of locally advanced and stenosing cancers, for staging perirectal lymph nodes, both TRUS and MRI are capable. TRUS has a small field of view, but 3D TRUS can improve the diagnosis of anorectal diseases.[29]
Magnetic Resonance Imaging
MRI is used when screening suggests a malignancy.[30] This model potentially minimizes unnecessary prostate biopsies while maximizing biopsy yield.[31] Despite concerns about the cost of MRI scans, compared to the long-term cost burden of the PSA/TRUS biopsy-based standard of care, the imaging model has been found to be cost-effective. MRI imaging can be used for patients who have had a previous negative biopsy but their PSA continues to increase.[32] Consensus has not been determined as to which of the MRI-targeted biopsy techniques is more useful.[33]
Other imaging
68Ga-PSMA PET/CT imaging has become, in a relatively short period of time, the gold standard for restaging recurrent prostate cancer in clinical centers in which this imaging modality is available.[34] It is likely to become the standard imaging modality in the staging of intermediate-to-high risk primary prostate cancer.[34] The potential to guide therapy, and to facilitate more accurate prostatic biopsy is being explored.[34] In the theranostic paradigm, 68Ga-PSMA PET/CT imaging is critical for detecting prostate specific membrane antigen-avid disease which may then respond to targeted 177Lu-PSMA or 225Ac-PSMA therapies.[34] For local recurrence, 68Ga-PSMA PET/MR or PET/CT in combination with mpMR is most appropriate.[35] PSMA PET/CT may be potentially helpful for locating the cancer when combined with multiparametric MRI (mpMRI) for primary prostate care.[36] Prostate multiparametric MR imaging (mpMRI) is helpful in evaluating recurrence of primary prostate cancer following treatment.[37]
Other
A number of biomarkers for prostate cancer exist. These include:[38]
- The 4Kscore combines total, free and intact PSA together with human kallikrein 2.[39] It is used to try to determine the risk of a Gleason score greater than 6.[39]
- The Prostate Health Index (PHI) is a PSA-based blood test for early prostate cancer screening. It may be used to determine when a biopsy is needed.[39][30]
- Prostate cancer antigen 3 (PCA3) is a urine test that detects the overexpression of the PCA3 gene, an indicator of prostate cancer.[39][30][40]
- ConfirmMDx is performed on tissue taken during a prostate biopsy. The test identifies men with clinically significant prostate cancer who would benefit from further testing and treatment. It can also help men without significant prostate cancer avoid unnecessary repeat biopsies.[38]
Researchers at the Korea Institute of Science and Technology (KIST) developed a urinary multi marker sensor with the ability to measure trace amounts of biomarkers from naturally voided urine.[41] The correlation of clinical state with the sensing signals form urinary multi markers was analyzed by two machine learning algorithms, random forest and neural network. Both algorithms provided a monotonic increase in screening performance as the number of biomarkers was increased. With the best combination of biomarkers, the algorithms were able to screen prostate cancer patients with more than 99% accuracy.[41]
Guidelines
- In 2012 the United States Preventive Services Task Force (USPSTF) recommended against prostate cancer screening using PSA.[42] As of 2018 a draft for new recommendations suggests that screening be individualized for those between the ages of 55 to 69. It notes a small potential decrease in the risk of dying from prostate cancer, but harm from overtreatment.[43] In those over the age of 70, PSA based screening is still recommended against.[43]
- In 2022, the European Commission's Scientific Advice Mechanism found "strong scientific evidence" for the benefits of screening using blood tests, and recommended that population-level screening should be introduced.[44]
- The American Cancer Society "recommends that asymptomatic men who have at least a 10-year life expectancy have an opportunity to make an informed decision with their health care provider about screening for prostate cancer after they receive information about the uncertainties, risks, and potential benefits associated with prostate cancer screening. Prostate cancer screening should not occur without an informed decision-making process. Men at average risk should receive this information beginning at age 50 years. Men in higher risk groups should receive this information before age 50 years. Men should either receive this information directly from their health care providers or be referred to reliable and culturally appropriate sources."[45]
- Other guidelines and centers specializing in treating prostate cancer recommend obtaining a PSA in all men at age 45.[46] This is based on emerging data indicating that an increased baseline PSA can be used to detect future significant disease.[46]
- The American Urological Association in 2018 states that men under the age of 55 and over the age of 69 should not be routinely screened. The greatest benefit of screening appears to be in men ages 55 to 69 years. To reduce the harms of screening, a routine screening interval of two years or more may be preferred over annual screening in those men who have participated in shared decision-making and decided on screening. Screening is done by PSA (blood test).[28]
- As of 2018, the UK National Health Service did not offer general PSA screening, for similar reasons to those given above. Individuals over the age of 50 who request it can normally obtain testing covered by the NHS.[23]
- The Canadian Urological Association in 2017 suggested screening be offered as a possibility to those who are expected to live more than 10 years, with the final decision based on shared decision making.[39] They recommend a starting age for most people at 50, and age 45 among those at high risk.[39] The Canadian Task Force on Preventive Health Care in 2014 strongly recommended against screening in those under 55 and over 70 years of age.[12] They weakly recommended against screening among those 55–69.[12]
- Some men have germ-line mutations associated with prostate cancer development (e.g., BRCA1, BRCA2, HOXB13). Screening and its frequency is established after consulting with a geneticist.[39]
Controversy
Screening for prostate cancer continues to generate debate among clinicians and broader lay audiences.[47] Publications authored by governmental, non-governmental and medical organizations continue the debate and publish recommendations for screening.[3] One in six men will be diagnosed with prostate cancer during their lifetime but screening may result in the overdiagnosis and overtreatment of prostate cancer.[48][49] Though the death rates from prostate cancer continue to decline, 238,590 men were diagnosed with prostate cancer in the United States in 2013 while 29,720 died as a result. Death rates from prostate cancer have declined at a steady rate since 1992. Cancers of the prostate, lung and bronchus, and colorectum accounted for about 50% of all newly diagnosed cancers in American men in 2013, with prostate cancer constituting 28% of cases. Screening for prostate cancer varies by state and indicates differences in the use of screening for prostate cancer as well as variations between locales. Out all cases of prostate cancer, African American men have an incidence of 62%. African American men are less likely to receive standard therapy for prostate cancer. This discrepancy may indicate that if they were to receive higher quality cancer treatment their survival rates would be similar to whites.[48]
Prostate cancer is also extremely heterogeneous: most prostate cancers are indolent and would never progress to a clinically meaningful stage if left undiagnosed and untreated during a man's lifetime. On the other hand, a subset are potentially lethal, and screening can identify some of these within a window of opportunity for cure.[50] Thus, PSA screening is advocated by some as a means of detecting high-risk, potentially lethal prostate cancer, with the understanding that lower-risk disease, if discovered, often does not need treatment and may be amenable to active surveillance.[22][51]
Screening for prostate cancer is controversial because of cost and uncertain long-term benefits to patients.[52] Horan echos that sentiment in his book.[53] Private medical institutes, such as the Mayo Clinic, likewise acknowledge that "organizations vary in their recommendations about who should – and who shouldn't – get a PSA screening test. They conclude: "Ultimately, whether you should have a PSA test is something you'll have to decide after discussing it with your doctor, considering your risk factors and weighing your personal preferences."[54]
A 2009 study in Europe resulted in only a small decline in death rates and concluded that 48 men would need to be treated to save one life. But of the 47 men who were treated, most would be unable to ever again function sexually and would require more frequent trips to the bathroom.[53] Aggressive marketing of screening tests by drug companies has also generated controversy as has the advocacy of testing by the American Urological Association.[53]
One commentator observed in 2011: “[I]t is prudent only to use a single PSA determination as a baseline, with biopsy and cancer treatment reserved for those with significant PSA changes over time, or for those with clinical manifestations mandating immediate therapy..... absolute levels of PSA are rarely meaningful; it is the relative change in PSA levels over time that provides insight, but not definitive proof of a cancerous condition necessitating therapy.“[55]
History
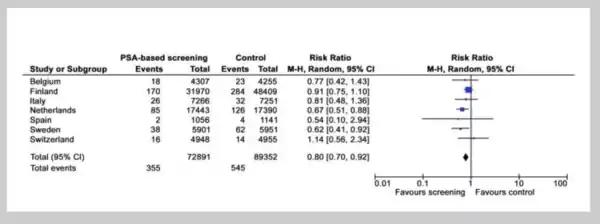
Screening of PSA began in the 1990s. In the European Randomized Study of Screening for Prostate Cancer (ERSPC) initiated in the early 1990s, the researchers concluded that PSA-based screening did reduce the rate of death from prostate cancer but created a high risk of overdiagnosis, i.e., 1410 men would need to be screened and 48 additional cases of prostate cancer would need to be treated to prevent just one death from prostate cancer within 9 years.[56]
A study published in the European Journal of Cancer (October 2009) documented that prostate cancer screening reduced prostate cancer mortality by 37 percent. By utilizing a control group of men from Northern Ireland, where PSA screening is infrequent, the research showed this substantial reduction in prostate cancer deaths when compared to men who were PSA tested as part of the ERSPC study.[57]
A study published in the New England Journal of Medicine in 2009 found that over a 7 to 10-year period, "screening did not reduce the death rate in men 55 and over."[53] Former screening proponents, including some from Stanford University, have come out against routine testing. In February 2010, the American Cancer Society urged "more caution in using the test." And the American College of Preventive Medicine concluded that "there was insufficient evidence to recommend routine screening."[53]
A further study, the NHS Comparison Arm for ProtecT (CAP), as part of the Prostate testing for cancer and Treatment (ProtecT) study, randomized GP practices with 460,000 men aged 50–69 at centers in 9 cities in Britain from 2001–2005 to usual care or prostate cancer screening with PSA (biopsy if PSA ≥ 3).[58] The "Comparison Arm" has yet to report as of early 2018.[59]
See also
References
- PDQ Screening and Prevention Editorial Board (2018). "Prostate Cancer Screening (PDQ®): Health Professional Version". PDQ Cancer Information Summaries. Bethesda (MD): National Cancer Institute (US). PMID 26389383.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - "Prostate Cancer Screening-Professional Version". National Cancer Institute. 1980-01-01. Retrieved 2018-02-28.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - Ilic D, Neuberger MM, Djulbegovic M, Dahm P (January 2013). "Screening for prostate cancer". The Cochrane Database of Systematic Reviews. 2013 (1): CD004720. doi:10.1002/14651858.cd004720.pub3. PMC 8406915. PMID 23440794.
- "Prostate Cancer Guidelines". Emedicine. 2017. Retrieved 18 February 2018.
- Greene KL, Albertsen PC, Babaian RJ, Carter HB, Gann PH, Han M, Kuban DA, Sartor AO, Stanford JL, Zietman A, Carroll P (January 2013). "Prostate specific antigen best practice statement: 2009 update". The Journal of Urology. 189 (1 Suppl): S2–S11. doi:10.1016/j.juro.2012.11.014. PMID 23234625.
- Wolf AM, Wender RC, Etzioni RB, Thompson IM, D'Amico AV, Volk RJ, Brooks DD, Dash C, Guessous I, Andrews K, DeSantis C, Smith RA (Mar–Apr 2010). "American Cancer Society guideline for the early detection of prostate cancer: update 2010". CA: A Cancer Journal for Clinicians. 60 (2): 70–98. doi:10.3322/caac.20066. PMID 20200110. S2CID 21548482.
- Basch E, Oliver TK, Vickers A, Thompson I, Kantoff P, Parnes H, Loblaw DA, Roth B, Williams J, Nam RK (August 2012). "Screening for prostate cancer with prostate-specific antigen testing: American Society of Clinical Oncology Provisional Clinical Opinion". Journal of Clinical Oncology. 30 (24): 3020–5. doi:10.1200/JCO.2012.43.3441. PMC 3776923. PMID 22802323.
- SAPEA, Scientific Advice for Policy by European Academies. (2 March 2022). "Improving cancer screening in the European Union". Retrieved 10 March 2022.
{{cite web}}:|last=has generic name (help)CS1 maint: url-status (link) - Borghesi M, Ahmed H, Nam R, Schaeffer E, Schiavina R, Taneja S, Weidner W, Loeb S (March 2017). "Complications After Systematic, Random, and Image-guided Prostate Biopsy". European Urology. 71 (3): 353–365. doi:10.1016/j.eururo.2016.08.004. hdl:11585/559951. PMID 27543165.
- Loeb S, Vellekoop A, Ahmed HU, Catto J, Emberton M, Nam R, Rosario DJ, Scattoni V, Lotan Y (December 2013). "Systematic review of complications of prostate biopsy". European Urology. 64 (6): 876–92. doi:10.1016/j.eururo.2013.05.049. PMID 23787356.
- Bennett HY, Roberts MJ, Doi SA, Gardiner RA (June 2016). "The global burden of major infectious complications following prostate biopsy". Epidemiology and Infection. 144 (8): 1784–91. doi:10.1017/S0950268815002885. PMC 9150640. PMID 26645476. S2CID 82280.
- Bell, N; Connor Gorber, S; Shane, A; Joffres, M; Singh, H; Dickinson, J; Shaw, E; Dunfield, L; Tonelli, M; Canadian Task Force on Preventive Health, Care. (4 November 2014). "Recommendations on screening for prostate cancer with the prostate-specific antigen test". Canadian Medical Association Journal. 186 (16): 1225–34. doi:10.1503/cmaj.140703. PMC 4216256. PMID 25349003.
- Schoots IG, Roobol MJ, Nieboer D, Bangma CH, Steyerberg EW, Hunink MG (September 2015). "Magnetic resonance imaging-targeted biopsy may enhance the diagnostic accuracy of significant prostate cancer detection compared to standard transrectal ultrasound-guided biopsy: a systematic review and meta-analysis". European Urology. 68 (3): 438–50. doi:10.1016/j.eururo.2014.11.037. PMID 25480312.
- Wegelin O, van Melick HH, Hooft L, Bosch JL, Reitsma HB, Barentsz JO, Somford DM (April 2017). "Comparing Three Different Techniques for Magnetic Resonance Imaging-targeted Prostate Biopsies: A Systematic Review of In-bore versus Magnetic Resonance Imaging-transrectal Ultrasound fusion versus Cognitive Registration. Is There a Preferred Technique?". European Urology. 71 (4): 517–531. doi:10.1016/j.eururo.2016.07.041. PMID 27568655.
- Hellstrom WJG, ed. (1999). "Chapter 8: What is the prostate and what is its function?". American Society of Andrology Handbook. San Francisco: American Society of Andrology. ISBN 978-1-891276-02-6. Archived from the original on 2010-01-12. Retrieved 2018-02-28.
- Catalona WJ, Richie JP, Ahmann FR, Hudson MA, Scardino PT, Flanigan RC, deKernion JB, Ratliff TL, Kavoussi LR, Dalkin BL (May 1994). "Comparison of digital rectal examination and serum prostate specific antigen in the early detection of prostate cancer: results of a multicenter clinical trial of 6,630 men". The Journal of Urology. 151 (5): 1283–90. doi:10.1016/S0022-5347(17)35233-3. PMID 7512659.
- Velonas VM, Woo HH, dos Remedios CG, Assinder SJ (May 2013). "Current status of biomarkers for prostate cancer". International Journal of Molecular Sciences. 14 (6): 11034–60. doi:10.3390/ijms140611034. PMC 3709717. PMID 23708103.
- Catalona WJ (March 2018). "Prostate Cancer Screening". The Medical Clinics of North America. 102 (2): 199–214. doi:10.1016/j.mcna.2017.11.001. PMC 5935113. PMID 29406053.
- Grossman, David C.; Curry, Susan J.; Owens, Douglas K.; Bibbins-Domingo, Kirsten; Caughey, Aaron B.; Davidson, Karina W.; Doubeni, Chyke A.; Ebell, Mark; Epling, John W.; Kemper, Alex R.; Krist, Alex H.; Kubik, Martha; Landefeld, C. Seth; Mangione, Carol M.; Silverstein, Michael; Simon, Melissa A.; Siu, Albert L.; Tseng, Chien-Wen (8 May 2018). "Screening for Prostate Cancer". JAMA. 319 (18): 1901–1913. doi:10.1001/jama.2018.3710. PMID 29801017.
Clinicians should not screen men who do not express a preference for screening.
- Stewart, Rosalyn W.; Lizama, Sergio; Peairs, Kimberly; Sateia, Heather F.; Choi, Youngjee (February 2017). "Screening for prostate cancer". Seminars in Oncology. 44 (1): 47–56. doi:10.1053/j.seminoncol.2017.02.001. PMID 28395763.
- Lu-Yao GL, Albertsen PC, Moore DF, Shih W, Lin Y, DiPaola RS, Barry MJ, Zietman A, et al. (September 2009). "Outcomes of localized prostate cancer following conservative management". JAMA. 302 (11): 1202–9. doi:10.1001/jama.2009.1348. PMC 2822438. PMID 19755699.
- Cooperberg MR, Carroll PR, Klotz L (September 2011). "Active surveillance for prostate cancer: progress and promise". Journal of Clinical Oncology. 29 (27): 3669–76. doi:10.1200/JCO.2011.34.9738. PMID 21825257. S2CID 24885033.
- "Prostate cancer - PSA testing - NHS Choices". NHS Choices. 3 January 2015.
- Naji, Leen; Randhawa, Harkanwal; Sohani, Zahra; Dennis, Brittany; Lautenbach, Deanna; Kavanagh, Owen; Bawor, Monica; Banfield, Laura; Profetto, Jason (12 March 2018). "Digital Rectal Examination for Prostate Cancer Screening in Primary Care: A Systematic Review and Meta-Analysis". The Annals of Family Medicine. 16 (2): 149–154. doi:10.1370/afm.2205. PMC 5847354. PMID 29531107.
- Tseng, Chien-Wen; Siu, Albert L.; Simon, Melissa A.; Silverstein, Michael; Mangione, Carol M.; Landefeld, C. Seth; Kubik, Martha; Krist, Alex H.; Kemper, Alex R. (2018-05-08). "Screening for Prostate Cancer: US Preventive Services Task Force Recommendation Statement". JAMA. 319 (18): 1901–1913. doi:10.1001/jama.2018.3710. ISSN 0098-7484. PMID 29801017.
- "UpToDate". www.uptodate.com. Retrieved 2019-02-07.
- Campbell's Urology 8th Edition. 2002. pp. Epstein JI. Pathology of prostatic neoplasia.
- "Prostate Cancer: Early Detection Guideline - American Urological Association". www.auanet.org. Retrieved 2019-10-10.
- Kim MJ (January 2015). "Transrectal ultrasonography of anorectal diseases: advantages and disadvantages". Ultrasonography. 34 (1): 19–31. doi:10.14366/usg.14051. PMC 4282231. PMID 25492891.
- Sarkar S, Das S (2016-03-02). "A Review of Imaging Methods for Prostate Cancer Detection". Biomedical Engineering and Computational Biology. 7 (Suppl 1): 1–15. doi:10.4137/BECB.S34255. PMC 4777886. PMID 26966397.
- Bjurlin MA, Taneja SS (1 May 2015). "Standards for prostate biopsy". Current Opinion in Urology. Curr Opin Urol. 24 (2): 155–161. doi:10.1097/MOU.0000000000000031. PMC 4142196. PMID 24451092.
- Turkbey, Baris; Choyke, Peter L. (2018). "Future Perspectives and Challenges of Prostate MR Imaging". Radiologic Clinics of North America. 56 (2): 327–337. doi:10.1016/j.rcl.2017.10.013. ISSN 0033-8389. PMC 5808592. PMID 29420986.
- Giganti, Francesco; Moore, Caroline M. (2017). "A critical comparison of techniques for MRI-targeted biopsy of the prostate". Translational Andrology and Urology. 6 (3): 432–443. doi:10.21037/tau.2017.03.77. ISSN 2223-4683. PMC 5503959. PMID 28725585.
- Lenzo NP, Meyrick D, Turner JH (February 2018). "Review of Gallium-68 PSMA PET/CT Imaging in the Management of Prostate Cancer". Diagnostics. 8 (1): 16. doi:10.3390/diagnostics8010016. PMC 5871999. PMID 29439481.
 This article incorporates text by Nat P. Lenzo, Danielle Meyrick, and J. Harvey Turner available under the CC BY 4.0 license.
This article incorporates text by Nat P. Lenzo, Danielle Meyrick, and J. Harvey Turner available under the CC BY 4.0 license. - Virgolini I, Decristoforo C, Haug A, Fanti S, Uprimny C (March 2018). "Current status of theranostics in prostate cancer". European Journal of Nuclear Medicine and Molecular Imaging. 45 (3): 471–495. doi:10.1007/s00259-017-3882-2. PMC 5787224. PMID 29282518.
 This article incorporates text by Irene Virgolini, Clemens Decristoforo, Alexander Haug, Stefano Fanti, and Christian Uprimny available under the CC BY 4.0 license.
This article incorporates text by Irene Virgolini, Clemens Decristoforo, Alexander Haug, Stefano Fanti, and Christian Uprimny available under the CC BY 4.0 license. - Bouchelouche, Kirsten; Choyke, Peter L. (2018). "Advances in prostate-specific membrane antigen PET of prostate cancer". Current Opinion in Oncology. 30 (3): 189–196. doi:10.1097/CCO.0000000000000439. ISSN 1040-8746. PMC 6003670. PMID 29465429.
- Gaur, Sonia; Turkbey, Baris (2018). "Prostate MR Imaging for Posttreatment Evaluation and Recurrence". Radiologic Clinics of North America. 56 (2): 263–275. doi:10.1016/j.rcl.2017.10.008. ISSN 0033-8389. PMC 5844499. PMID 29420981.
- Kretschmer, Alexander; Tilki, Derya (2017). "Biomarkers in prostate cancer – Current clinical utility and future perspectives". Critical Reviews in Oncology/Hematology. 120: 180–193. doi:10.1016/j.critrevonc.2017.11.007. ISSN 1040-8428. PMID 29198331.
- Rendon RA, Mason RJ, Marzouk K, Finelli A, Saad F, So A, Violette P, Breau RH (October 2017). "Canadian Urological Association recommendations on prostate cancer screening and early diagnosis". Canadian Urological Association Journal. 11 (10): 298–309. doi:10.5489/cuaj.4888. PMC 5659858. PMID 29381452.
- Progensa PCA3 Assay [package insert]. 502083-IFU-PI, Rev. 001. San Diego, CA: Hologic, Inc.; 2015.
- Kim, Hojun; Park, Sungwook; Jeong, In Gab; Song, Sang Hoon; Jeong, Youngdo; Kim, Choung-Soo; Lee, Kwan Hyi (9 December 2020). "Noninvasive Precision Screening of Prostate Cancer by Urinary Multimarker Sensor and Artificial Intelligence Analysis". ACS Nano. 15 (3): 4054–4065. doi:10.1021/acsnano.0c06946. PMID 33296173. S2CID 228089390.
- "Final Update Summary: Prostate Cancer: Screening - US Preventive Services Task Force". www.uspreventiveservicestaskforce.org. Retrieved 2 March 2018.
- "Home Page - USPSTF Draft Prostate Screening Recommendations". USPSTF Draft Prostate Screening Recommendations. Archived from the original on 2 March 2018. Retrieved 2 March 2018.
- SAPEA, Scientific Advice for Policy by European Academies. (2 March 2022). "Improving cancer screening in the European Union". Retrieved 10 March 2022.
{{cite web}}:|last=has generic name (help)CS1 maint: url-status (link) - Wolf AM, Wender RC, Etzioni RB, Thompson IM, D'Amico AV, Volk RJ, Brooks DD, Dash C, Guessous I, Andrews K, DeSantis C, Smith RA (2010). "American Cancer Society guideline for the early detection of prostate cancer: update 2010". CA: A Cancer Journal for Clinicians. 60 (2): 70–98. doi:10.3322/caac.20066. PMID 20200110. S2CID 21548482.
- Fleshner, Katherine; Carlsson, Sigrid V.; Roobol, Monique J. (2016). "The effect of the USPSTF PSA screening recommendation on prostate cancer incidence patterns in the USA". Nature Reviews Urology. 14 (1): 26–37. doi:10.1038/nrurol.2016.251. ISSN 1759-4812. PMC 5341610. PMID 27995937.
- Sadi, Marcus V. (2017). "PSA screening for prostate cancer". Revista da Associação Médica Brasileira. 63 (8): 722–725. doi:10.1590/1806-9282.63.08.722. ISSN 0104-4230. PMID 28977112.
- Siegel R, Naishadham D, Jemal A (January 2013). "Cancer statistics, 2013". CA: A Cancer Journal for Clinicians. 63 (1): 11–30. doi:10.3322/caac.21166. PMID 23335087. S2CID 24926725.
- Ashley VA, Joseph MB, Kamlesh KY, Shalini SY, Ashutosh KT, Joseph R (2017). "The Use of Biomarkers in Prostate Cancer Screening and Treatment". Reviews in Urology. 19 (4): 221–234. doi:10.3909/riu0772 (inactive 31 July 2022). PMC 5811879. PMID 29472826.
{{cite journal}}: CS1 maint: DOI inactive as of July 2022 (link) - Esserman L, Shieh Y, Thompson I (October 2009). "Rethinking screening for breast cancer and prostate cancer". JAMA. 302 (15): 1685–92. doi:10.1001/jama.2009.1498. PMID 19843904. S2CID 25598632.
- Carroll PR, Whitson JM, Cooperberg MR (February 2011). "Serum prostate-specific antigen for the early detection of prostate cancer: always, never, or only sometimes?". Journal of Clinical Oncology. 29 (4): 345–7. doi:10.1200/JCO.2010.32.5308. PMID 21189396. S2CID 20098979.
- Collins MM, Barry MJ (December 1996). "Controversies in prostate cancer screening. Analogies to the early lung cancer screening debate". JAMA. 276 (24): 1976–9. doi:10.1001/jama.276.24.1976. PMID 8971068.
- Anthony Horan (August 2009). The Big Scare: The Business of Prostate Cancer. STERLINGHOUSE. ISBN 978-1-58501-119-3. Retrieved 18 August 2013.
- "Prostate cancer screening: Should you get a PSA test?" MayoClinic.com, updated March 6, 2010
- Haythorn MR, Ablin RJ (August 2011). "Prostate-specific antigen testing across the spectrum of prostate cancer". Biomarkers in Medicine. 5 (4): 515–26. doi:10.2217/bmm.11.53. PMID 21861672.
- Schröder FH, Hugosson J, Roobol MJ, Tammela TL, Ciatto S, Nelen V, Kwiatkowski M, Lujan M, Lilja H, Zappa M, Denis LJ, Recker F, Berenguer A, Määttänen L, Bangma CH, Aus G, Villers A, Rebillard X, van der Kwast T, Blijenberg BG, Moss SM, de Koning HJ, Auvinen A (March 2009). "Screening and prostate-cancer mortality in a randomized European study". The New England Journal of Medicine. 360 (13): 1320–8. doi:10.1056/NEJMoa0810084. hdl:2027.42/137255. PMID 19297566. S2CID 9602977.
- van Leeuwen PJ, Connolly D, Gavin A, Roobol MJ, Black A, Bangma CH, Schröder FH (January 2010). "Prostate cancer mortality in screen and clinically detected prostate cancer: estimating the screening benefit". European Journal of Cancer. 46 (2): 377–83. doi:10.1016/j.ejca.2009.09.008. PMID 19804966. S2CID 207211780. Archived from the original on 2015-09-01. Retrieved 2019-02-02.
- ProtecT (July 25, 2007). "ProtecT Study (Prostate testing for cancer and Treatment)". University of Bristol Dept. of Social Medicine. Archived from the original on 2007-10-28. Retrieved 2007-11-19.
ISRCTN (March 6, 2006). "The CAP (Comparison Arm for ProtecT) study". isrctn.org. doi:10.1186/ISRCTN92187251.{{cite journal}}: Cite journal requires|journal=(help)
ISRCTN (November 19, 2007). "The ProtecT trial". isrctn.org. doi:10.1186/ISRCTN20141297.{{cite journal}}: Cite journal requires|journal=(help) - "Outline of the ProtecT study and its data and samples", on the study website at Bristol University.