Mitoxantrone
 | |
 | |
| Names | |
|---|---|
| Trade names | Novantrone, Onkotrone, others |
| Other names | Mitozantrone |
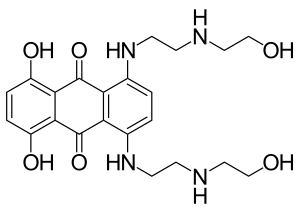
IUPAC name
| |
| Clinical data | |
| Drug class | Antineoplastic (anthracenedione)[1] |
| Main uses | Cancer, multiple sclerosis[1] |
| Side effects | Nausea, diarrhea, hair loss, infection, fever, swelling, shortness of breath[1] |
| Pregnancy category |
|
| Routes of use | Mainly intravenous |
| External links | |
| AHFS/Drugs.com | Monograph |
| US NLM | Mitoxantrone |
| MedlinePlus | a608019 |
| Legal | |
| Legal status |
|
| Pharmacokinetics | |
| Bioavailability | n/a |
| Protein binding | 78% |
| Metabolism | Liver (CYP2E1) |
| Elimination half-life | 75 hours |
| Excretion | Kidney |
| Chemical and physical data | |
| Formula | C22H28N4O6 |
| Molar mass | 444.488 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Mitoxantrone, also known as mitozantrone, is a medication used to treat cancer and multiple sclerosis.[1][2] Types of cancer it is used for include acute myeloid leukemia, prostate cancer, breast cancer, and non-Hodgkin's lymphoma.[1][2] It is given by gradual injection into a vein.[1]
Common side effects include nausea, diarrhea, hair loss, infection, fever, swelling, and shortness of breath.[1] Other side effects may include tumor lysis syndrome, neuropathy, bone marrow suppression, and heart damage.[1] Use in pregnancy may harm the baby.[1] It is an anthracenedione antineoplastic agent.[1]
Mitoxantrone was approved for medical use in the United States in 1987.[1] It is available as a generic medication.[2] In the United Kingdom it costs the NHS about £51 per 20 mg vial as of 2021.[2] This amount in the United States costs about 180 USD.[3]
Medical uses
Mitoxantrone is used to treat certain types of cancer, mostly acute myeloid leukemia. It improves the survival rate of children suffering from acute lymphoblastic leukemia relapse.[4]
The combination of mitoxantrone and prednisone is approved as a second-line treatment for metastatic hormone-refractory prostate cancer. Until recently this combination was the first line of treatment; however, a combination of docetaxel and prednisone improves survival rates and lengthens the disease-free period.[5]
Mitoxantrone is also used to treat multiple sclerosis (MS), most notably the subset of the disease known as secondary-progressive MS. In the absence of a cure, mitoxantrone is effective in slowing the progression of secondary-progressive MS and extending the time between relapses in both relapsing-remitting MS and progressive-relapsing MS.[6]
Side effects
Mitoxantrone, as with other drugs in its class, may cause adverse reactions of varying severity, including nausea, vomiting, hair loss, heart damage and immunosuppression, possibly with delayed onset. Cardiomyopathy is a particularly concerning effect as it is irreversible; thus regular monitoring with echocardiograms or MUGA scans is recommended for patients.
Because of the risk of cardiomyopathy, mitoxantrone carries a limit on the cumulative lifetime dose (based on body surface area) in MS patients.[7]
Mechanism of action
Mitoxantrone is a type II topoisomerase inhibitor; it disrupts DNA synthesis and DNA repair in both healthy cells and cancer cells by intercalation[9][10] between DNA bases. It is also classified as an antibiotic.[11]
See also
- Pixantrone, a mitoxantrone analogue under development
- Losoxantrone
References
- 1 2 3 4 5 6 7 8 9 10 11 "MitoXANTRONE Monograph for Professionals". Drugs.com. Archived from the original on 7 May 2021. Retrieved 18 November 2021.
- 1 2 3 4 BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 948. ISBN 978-0857114105.
- ↑ "Mitoxantrone Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 19 April 2021. Retrieved 18 November 2021.
- ↑ Parker C, Waters R, Leighton C, Hancock J, Sutton R, Moorman AV, Ancliff P, Morgan M, Masurekar A, Goulden N, Green N, Révész T, Darbyshire P, Love S, Saha V (2010). "Effect of mitoxantrone on outcome of children with first relapse of acute lymphoblastic leukaemia (ALL R3): an open-label randomised trial". Lancet. 376 (9757): 2009–2017. doi:10.1016/S0140-6736(10)62002-8. PMC 3010035. PMID 21131038.
- ↑ Katzung, Bertram G. (2006). "Cancer Chemotherapy". Basic and clinical pharmacology (10th ed.). New York: McGraw-Hill Medical Publishing Division. ISBN 0-07-145153-6. OCLC 157011367.
- ↑ Fox E (2006). "Management of worsening multiple sclerosis with mitoxantrone: a review". Clin Ther. 28 (4): 461–74. doi:10.1016/j.clinthera.2006.04.013. PMID 16750460.
- ↑ "Mitoxantrone Hydrochloride (marketed as Novantrone and generics) – Healthcare Professional Sheet text version". U.S. Food and Drug Administration. Archived from the original on 9 August 2014. Retrieved 19 September 2014.
- ↑ Wu, C. -C.; Li, Y. -C.; Wang, Y. -R.; Li, T. -K.; Chan, N. -L. (2013). "On the structural basis and design guidelines for type II topoisomerase-targeting anticancer drugs". Nucleic Acids Research. 41 (22): 10630–10640. doi:10.1093/nar/gkt828. PMC 3905874. PMID 24038465.
- ↑ Mazerski J, Martelli S, Borowski E (1998). "The geometry of intercalation complex of antitumor mitoxantrone and ametantrone with DNA: molecular dynamics simulations". Acta Biochim. Pol. 45 (1): 1–11. doi:10.18388/abp.1998_4280. PMID 9701490.
- ↑ Kapuscinski, J; Darzynkiewicz, Z (1985). "Interactions of antitumor agents Ametantrone and Mitoxantrone (Novatrone) with double-stranded DNA". Biochem Pharmacol. 34 (24): 4203–13. doi:10.1016/0006-2952(85)90275-8. PMID 4074383.
- ↑ "Mitoxantrone". Archived from the original on 2019-04-08. Retrieved 2021-08-02.
External links
| External sites: |
|
|---|---|
| Identifiers: |
