Nigericin
 | |
| Names | |
|---|---|
| Preferred IUPAC name
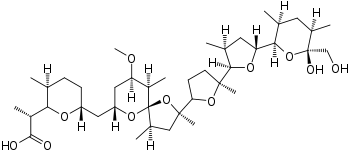
(2R)-2-[(2R,3S,6R)-6-{[(2S,4R,5R,7R,9R,10R)-2-{(2S,2′R,3′S,5R,5′R)-5′-[(2S,3S,5R,6R)-6-Hydroxy-6-(hydroxymethyl)-3,5-dimethyloxan-2-yl]-2,3′-dimethyl[2,2′-bioxolan]-5-yl}-9-methoxy-2,4,10-trimethyl-1,6-dixoaspiro[4.5]decan-7-yl]methyl}-3-methyloxan-2-yl]propanoic acid | |
| Other names
Polyetherin A, Azalomycin M, Helixin C, Helix C | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.212.814 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C40H68O11 |
| Molar mass | 724.96132 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Nigericin is an antibiotic derived from Streptomyces hygroscopicus. Its isolation was described in the 1950s,[1] and in 1968 the structure could be elucidated by X-ray crystallography.[2] The structure and properties of nigericin are similar to the antibiotic monensin. Commercially it is obtained as a byproduct, or contaminant, at the fermentation of Geldanamycin. It is also called Polyetherin A, Azalomycin M, Helixin C, Antibiotic K178, Antibiotic X-464.
Nigericin acts as an H+, K+, Pb2+ ionophore. Most commonly it is an antiporter of H+ and K+.
In the past nigericin was used as an antibiotic active against gram positive bacteria. It inhibits the Golgi functions in Eukaryotic cells. Its ability to induce K+ efflux also makes it a potent activator of the NLRP3 inflammasome[3][4]
References
- ↑ Graven SN, Estrada-O S, Lardy HA (1966). "Alkali metal cation release and respiratory inhibition induced by nigericin in rat liver mitochondria". Proc. Natl. Acad. Sci. U.S.A. 56 (2): 654–8. Bibcode:1966PNAS...56..654G. doi:10.1073/pnas.56.2.654. PMC 224422. PMID 5229984.
- ↑ Steinrauf LK, Pinkerton M, Chamberlin JW (1968). "The structure of nigericin". Biochem. Biophys. Res. Commun. 33 (1): 29–31. doi:10.1016/0006-291X(68)90249-0. PMID 5696503.
- ↑ Mariathasan, S; Weiss, DS; Newton, K; McBride, J; O'Rourke, K; Roose-Girma, M; Lee, WP; Weinrauch, Y; Monack, DM; Dixit, VM (9 March 2006). "Cryopyrin activates the inflammasome in response to toxins and ATP". Nature. 440 (7081): 228–32. Bibcode:2006Natur.440..228M. doi:10.1038/nature04515. PMID 16407890.
- ↑ Muñoz-Planillo, R; Kuffa, P; Martínez-Colón, G; Smith, BL; Rajendiran, TM; Núñez, G (27 June 2013). "K⁺ efflux is the common trigger of NLRP3 inflammasome activation by bacterial toxins and particulate matter". Immunity. 38 (6): 1142–53. doi:10.1016/j.immuni.2013.05.016. PMC 3730833. PMID 23809161.