This article was co-authored by Bess Ruff, MA. Bess Ruff is a Geography PhD student at Florida State University. She received her MA in Environmental Science and Management from the University of California, Santa Barbara in 2016. She has conducted survey work for marine spatial planning projects in the Caribbean and provided research support as a graduate fellow for the Sustainable Fisheries Group.
There are 8 references cited in this article, which can be found at the bottom of the page.
This article has been viewed 67,964 times.
Phosphate buffered saline (PBS) is a salty mixture used in biological research. It is used to dilute a group of cells or to separate cells. This buffer maintains a steady pH so cells are not destroyed while being looked at and stored. You can make a 1X solution that can be used directly on cells or you can make a 10X stock that can be diluted down to 1X. If your lab uses a lot of PBS it might be a good idea to make a 10X stock since it will last longer.
Steps
Making a 1X Stock
-
1Gather your materials and ingredients. To make a 1X (the normal concentration used for experiments) solution of phosphate buffered saline (PBS), you will need a 1-L graduated cylinder, a balance, a 2-L beaker, a stir bar, a stir plate, a pH meter, an autoclave, and a glass bottle for storage. To make 1 L of PBS, you will need the following amounts of each ingredient:[1]
- Small amounts of dilute HCl and/or NaOH to pH the final solution
- 800 mL of distilled water
- 8 g of NaCl (sodium chloride)
- 0.2 g of KCl (potassium chloride)
- 1.44 g of Na2HPO4 (disodium phosphate)
- 0.24 g of KH2PO4 (monopotassium phosphate)
- The final concentrations of each compound should be 137 mm NaCl, 10 mM Na2HPO4, 1.8 mM KH2PO4, 2.7 mM KCl, and a pH 7.4.[2]
-
2Place the stir bar in the beaker and place the beaker on top of the stir plate. When making any solution, you want to keep the solution constantly moving. This will help everything dissolve faster. To avoid splashing once you add water, place the stir bar into the empty beaker and then put it on top of the stir plate.[3]
- A stir bar contains a magnet that allows the bar to spin and stir the solution when a container of liquid is placed on the stir plate, which contains a motor.[4]
- Wait until there is solution in the beaker before turning it on.
Advertisement -
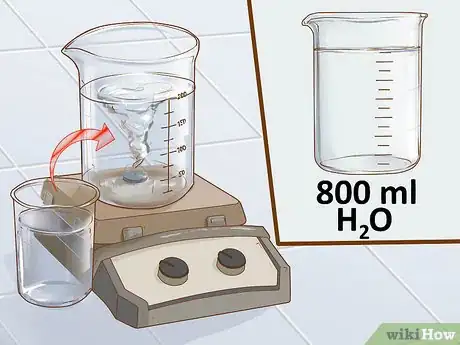
3Measure out 800 mL of distilled water into the 2-L beaker, then turn on the stir plate. When preparing a solution, start with less than the final volume of water. At the end, you will add the rest of the water to bring the total volume up to 1 L. Using the graduated cylinder, measure 800 mL of water and pour this directly into a clean beaker.[5]
- With the water in the beaker, turn on the stir plate to a medium speed. You should see a small whirlpool form in the water as the stir bar begins to spin.
-
4Weigh out the proper amounts of each chemical. Weigh out 8 g of NaCl, 0.2 g of KCl, 1.44 g of Na2HPO4, and 0.24 g of KH2PO4. Use the balance to weigh out each chemical one at a time in their own weigh boats.[6]
- If you add too much of a chemical, simply remove the excess until you have the right amount.
-
5Add all of the chemicals to the solution and dissolve. Pour each chemical into the water while it is stirring. There is no special order to add each one, so you can add everything all at once. Let everything continue to stir until it is fully dissolved.[7]
- You can let this sit while you do something else and check on it later. It will take at least 5-10 minutes to dissolve.
-
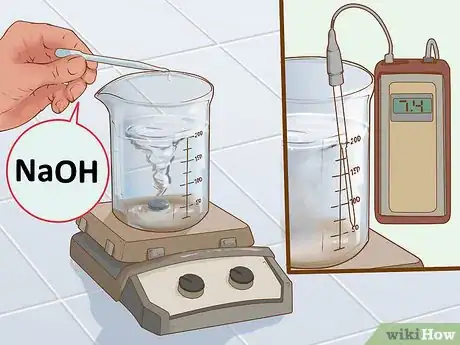
6Adjust the pH of the solution to 7.4. Once everything has fully dissolved, you need to adjust the pH of the buffer until it is 7.4.[8] Keep the stir bar on and place the pH meter into the top of the solution. Let the numbers on the pH meter stop moving. The solution will likely be too acidic so you’ll need to add sodium hydroxide (NaOH) to adjust the pH.
- Use a small pipette to add NaOH a few drops at a time. After each addition, let the solution stir for a moment and check the new reading on the pH meter.
- Adjusting the pH a solution is a process that requires patience. If you accidentally go higher than 7.4, you can add a few drops of hydrochloric acid (HCl) to bring it back down.
-
7Top off the water until the solution is 1 liter. After the pH has been properly adjusted, you can bring your solution up to the final volume of 1 liter. Carefully pour the entire solution from the beaker into the 1-L graduated cylinder. Add enough distilled water to make it 1 L.[9]
- Pour the final solution into a 1-L glass bottle safe for autoclaving.
-
8Sterilize the solution in an autoclave. Place the lid on the bottle, but do not screw it down. During autoclaving, the pressure in the bottle can build up and explode if you tighten the lid. Put a strip of autoclave tape across the lid to hold it in place while autoclaving.[10]
- Set the autoclave to sterilize for 20 minutes at 15 psi on the liquid cycle.[11]
- Autoclave tape has a temperature indicator in it. If the autoclave tape doesn’t turn black when the process is done, the autoclave did not get hot enough and you will need to re-sterilize.
-
9Store PBS at room temperature. After autoclaving, let the PBS cool to room temperature and then tighten the lid to keep the contaminants out. Tightening the lid before the bottle cools can create a vacuum and make it very difficult to remove the lid later. PBS is stable at room temperature.[12]
- You can also aliquot the PBS into smaller containers to use one at a time.
Making a 10X Stock
-
1Collect your materials and ingredients. Making the 10X (ten times the normal concentration used) stock is almost identical to making the 1X stock, you simply add 10 times the amount of chemicals. To make the 10X stock, you will need a 1-L graduated cylinder, a balance, a 2-L beaker, a stir bar, a stir plate, a pH meter, an autoclave, and a glass bottle for storage.[13] To make 1-L you will need the following amounts of each compound:[14]
- 700 mL of distilled water
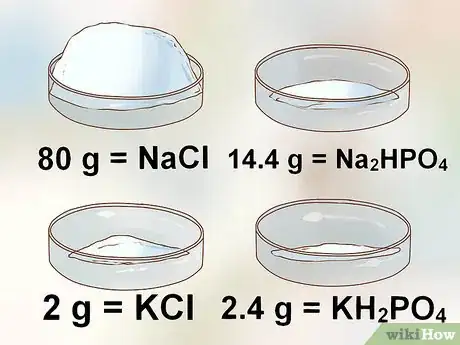
- 80 g of NaCl (sodium chloride)
- 2 g of KCl (potassium chloride)
- 14.4 g of Na2HPO4 (disodium phosphate)
- 2.4 g of KH2PO4 (monopotassium phosphate)
- You will also need small amounts of HCl and/or NaOH to pH the final solution.
- The final concentrations of each compound in the 10X solution are 1.37 M NaCl, 27 mM KCl, 100 mM Na2HPO4, and 18 mM KH2PO4.
-
2Place the beaker on top of the stir plate and add the stir bar. To help dissolve everything quickly, use a stir bar and stir plate to keep the solution constantly moving. Place the stir bar into the empty beaker before beginning to avoid splashes.[15]
- Wait until there is solution in the beaker before turning the stir plate on.
-
3Measure out 700 mL of distilled water into the 2-L beaker, then turn on the stir plate. You want to start with less than the final volume because the volume will change as you add the compounds to it. After everything has been added and dissolved, you will add the rest of the water to bring the total volume up to 1-L. Measure 700 mL of water with the graduated cylinder and pour this directly into a clean beaker.[16]
- Once water is added, turn on the stir plate to a medium speed. You should see a small whirlpool form in the water as the stir bar begins to spin.
-
4Weigh out the proper amounts of each chemical. Weigh out 80 g of NaCl, 2 g of KCl, 14.4 g of Na2HPO4, and 2.4 g of KH2PO4. Weigh out each chemical individually in their own weight boat.[17]
- If you add too much of a chemical, simply remove the excess until you have the right amount.
-
5Dissolve all of the chemicals in the solution. Add each chemical to the water while it stirs. There is no special order to add each one, so you can add everything all at once. Continue to stir until all the powder is fully dissolved.[18]
- It will take at least 5-10 minutes to dissolve, so let it stir and check on it later.
-
6Ensure the pH of the solution is 7.4. With everything fully dissolved, you need to check the pH of the buffer and adjust it to 7.4.[19] Keep the stir bar on and submerge the tip of the pH meter in the top of the solution. Let the numbers on the pH meter stop moving to measure the current pH. The solution will likely be too acidic so you’ll need to add sodium hydroxide (NaOH) to make it more basic.
- Use a small pipette to add NaOH a few drops at a time. After each addition, let the solution stir for a moment and wait until the numbers stop moving. Keep adding NaOH until you reach 7.4.
- To pH a solution is a process that requires patience. If you accidentally go higher than 7.4 you can add a few drops of hydrochloric acid (HCl) to bring it back down.
-
7Add water to make the final volume 1 liter. With the pH properly adjusted, bring the solution up to the final volume of 1 liter. Carefully pour the entire solution from the beaker into the 1-L graduated cylinder. Add enough distilled water to make it 1 L.[20]
- Pour the final solution into a 1-L glass bottle safe for autoclaving.
-
8Use the autoclave to sterilize the solution. Place the lid on the bottle, but do not screw it all the way down. The autoclave process produces steam which can cause a buildup of pressure in the bottle. If the lid is tightened, the bottle can explode. Use a strip of autoclave tape across the lid to hold it in place while autoclaving.[21]
- Set the autoclave to sterilize for 20 minutes at 15 psi on the liquid cycle.[22]
- Autoclave tape has a temperature indicator in it. If it reaches the proper temperature, the tape will turn black. If the tape does not turn black, the autoclave did not get hot enough and you will need to re-sterilize.
-
9Keep PBS stored at room temperature. After autoclaving, let the PBS cool completely before tightening the lid. If you tighten the lid while it is still hot, it can create a vacuum that will make it very difficult to remove the lid later. PBS is stable long-term at room temperature.[23]
- You can also aliquot the PBS into smaller containers to use one at a time.
Diluting the 10X Stock to 1X
-
1Determine the amount of 1X solution you will need. First, you must know how much 1X stock you want before you can calculate how much 10X stock you will need. The amount to make will be dependent on the type of experiment you are doing.
- If you just need an aliquot of 1X stock to have around, make around 500 mL. You can use this 1X stock for a while and won’t have to make it as often.
-
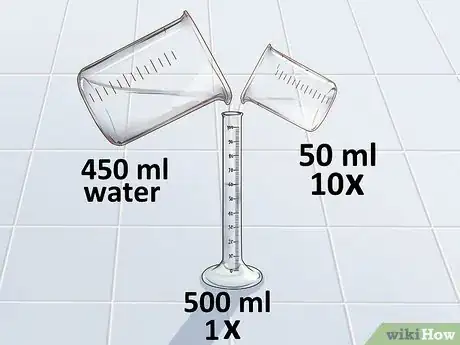
2Calculate the amount of 10X stock needed. To calculate the amount of 10X stock needed, you simply divide your total volume by 10.[24] Remember, to get the correct volume, your units must match: if the volume you are making is in milliliters, the volume you add will be in milliliters.
- For example: If you want 500 mL of solution, divide 500 by 10 and you get 50 mL. You need 50 mL of 10X stock to make 500 mL of 1X.
- For example: If you want 2 L of solution, divide 2/10 and you get 0.2 L. You can convert this to mL by multiplying by 1000. You need 200 mL of 10X stock to make 2 L of 1X.
-
3Subtract the 10X volume from the total volume. To determine the amount of water needed to dilute the 10X stock, you just need to subtract the calculated 10X volume from the total volume that you want.[25]
- For example: To make 500 mLs, you need 50 mLs of 10X stock. Since 500 – 50 = 450, you need 450 mL of distilled water to dilute the stock.
-
4Dilute the 10X stock with distilled water. Using the volumes that you have calculated, add distilled water to the 10X stock. You have now made a 1X stock of PBS.[26]
- For example, add 50 mL of 10X stock to 450 mL of distilled water to make 1X PBS stock. Swirl the container to mix the solution.
-
5Check the pH of the 1X stock. Sometimes the dilution process affects the pH of the buffer. It is prudent to check the pH of the 1X stock and make sure it is at 7.4 following dilution.[27]
- Use the pH meter to measure the pH and use HCl or NaOH to correct the buffer to a pH of 7.4.
Warnings
- The chemicals used to make PBS are all safe to handle, but it is important to wear proper protective equipment at all times when working in the lab.⧼thumbs_response⧽
- Wear gloves, a lab coat, and eye protection while in the lab.⧼thumbs_response⧽
- Always follow the safety protocols when working in a lab.⧼thumbs_response⧽
- Although you don’t need them to make PBS, always know where your emergency showers and eyewashes are.⧼thumbs_response⧽
References
- ↑ http://protocolsonline.com/recipes/phosphate-buffered-saline-pbs/
- ↑ http://cshprotocols.cshlp.org/content/2006/1/pdb.rec8247
- ↑ https://www.kjmagnetics.com/blog.asp?p=stir-bars
- ↑ http://www.orgchemboulder.com/Technique/Equipment/Benchequip/Stirbar.shtml
- ↑ http://protocolsonline.com/recipes/phosphate-buffered-saline-pbs/
- ↑ http://protocolsonline.com/recipes/phosphate-buffered-saline-pbs/
- ↑ http://protocolsonline.com/recipes/phosphate-buffered-saline-pbs/
- ↑ http://cshprotocols.cshlp.org/content/2006/1/pdb.rec8247
- ↑ http://cshprotocols.cshlp.org/content/2006/1/pdb.rec8247
- ↑ http://www.calstatela.edu/sites/default/files/groups/Environmental%20Health%20and%20Safety/autoclavefactsheet.pdf
- ↑ http://cshprotocols.cshlp.org/content/2006/1/pdb.rec8247
- ↑ http://cshprotocols.cshlp.org/content/2006/1/pdb.rec8247
- ↑ http://protocolsonline.com/recipes/phosphate-buffered-saline-pbs/
- ↑ http://cshprotocols.cshlp.org/content/2006/1/pdb.rec8247
- ↑ https://www.kjmagnetics.com/blog.asp?p=stir-bars
- ↑ http://protocolsonline.com/recipes/phosphate-buffered-saline-pbs/
- ↑ http://protocolsonline.com/recipes/phosphate-buffered-saline-pbs/
- ↑ http://protocolsonline.com/recipes/phosphate-buffered-saline-pbs/
- ↑ http://cshprotocols.cshlp.org/content/2006/1/pdb.rec8247
- ↑ http://cshprotocols.cshlp.org/content/2006/1/pdb.rec8247
- ↑ http://www.calstatela.edu/sites/default/files/groups/Environmental%20Health%20and%20Safety/autoclavefactsheet.pdf
- ↑ http://cshprotocols.cshlp.org/content/2006/1/pdb.rec8247
- ↑ http://cshprotocols.cshlp.org/content/2006/1/pdb.rec8247
- ↑ https://www.elabprotocols.com/protocols/#!protocol=234
- ↑ https://www.quansysbio.com/support/dilutions-explanations-and-examples/
- ↑ https://www.elabprotocols.com/protocols/#!protocol=234
- ↑ http://protocolsonline.com/recipes/phosphate-buffered-saline-pbs/